Scalable Synthesis of Novel PTP1B Inhibitor Intermediates for Commercial Diabetes Drug Production
Introduction to Advanced PTP1B Inhibitor Synthesis
The global pharmaceutical landscape is constantly evolving to address the rising prevalence of metabolic disorders, particularly Type 2 Diabetes Mellitus (T2DM). A pivotal development in this sector is detailed in patent CN102020546B, which discloses a robust chemical total synthesis method for a novel Protein Tyrosine Phosphatase 1B (PTP1B) inhibitor. This specific compound, bis-(2,3,6-tribromo-4,5-dimethoxy-phenyl)-methanone, represents a significant breakthrough in the design of insulin sensitizers. Unlike traditional therapies that merely manage symptoms, this molecule targets the molecular etiology of insulin resistance by inhibiting PTP1B activity, thereby enhancing insulin receptor sensitivity. For R&D directors and procurement specialists, understanding the synthetic accessibility and structural integrity of this intermediate is crucial for securing a reliable supply chain for next-generation anti-diabetic drugs.
The significance of this patent extends beyond mere biological activity; it provides a comprehensive roadmap for the commercial production of high-purity pharmaceutical intermediates. The synthesis strategy outlined leverages cost-effective starting materials and avoids the use of exotic or prohibitively expensive catalysts, which is a critical factor for cost reduction in pharmaceutical intermediate manufacturing. By establishing a clear pathway from basic aromatic precursors to a highly functionalized brominated ketone, the technology enables manufacturers to produce this potent inhibitor with consistent quality. This report analyzes the technical nuances of this synthesis, evaluating its feasibility for scale-up and its potential to disrupt the current market for diabetes therapeutics through improved efficacy and supply chain stability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of PTP1B inhibitors has been fraught with challenges related to bioavailability, selectivity, and chemical stability. Early generations of inhibitors, such as peptide-based substrates containing phosphorylated tyrosine residues, exhibited high affinity for the enzyme but suffered from poor chemical and biological stability in vivo, rendering them impractical for oral administration. Furthermore, other classes like naphthoquinones and thiazolidinediones often faced issues with severe hepatotoxicity or poor cell membrane permeability. From a manufacturing perspective, synthesizing complex heterocyclic systems often requires multi-step sequences involving sensitive reagents and harsh conditions that are difficult to control on a large scale. These limitations result in low overall yields, high production costs, and significant difficulties in maintaining batch-to-batch consistency, which are major pain points for supply chain managers aiming to reduce lead time for high-purity pharmaceutical intermediates.
The Novel Approach
The methodology presented in the patent data introduces a streamlined, four-step synthetic route that elegantly constructs the target bis-(2,3,6-tribromo-4,5-dimethoxy-phenyl)-methanone. This approach utilizes a Friedel-Crafts acylation followed by a sequential, regioselective bromination strategy. By starting with veratric acid and veratrole, the process builds the carbon skeleton efficiently before introducing the critical halogen substituents. This stepwise functionalization allows for precise control over the substitution pattern, ensuring the correct positioning of bromine atoms which are essential for the compound's biological activity. The use of standard reagents such as polyphosphoric acid, elemental bromine, and N-bromosuccinimide (NBS) simplifies the operational complexity. This novel approach not only enhances the chemical stability of the final product compared to peptide analogs but also significantly improves the economic viability of the manufacturing process, making it an attractive candidate for commercial scale-up of complex pharmaceutical intermediates.
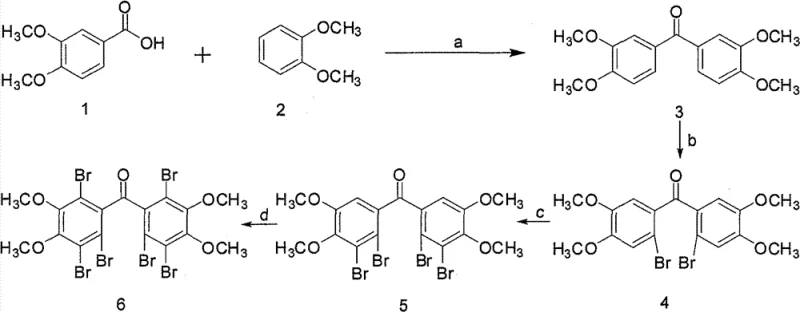
Mechanistic Insights into Sequential Bromination and Acylation
The core of this synthesis lies in the strategic application of electrophilic aromatic substitution reactions to achieve a highly substituted benzophenone derivative. The initial step involves the condensation of veratric acid and veratrole mediated by polyphosphoric acid (PPA) at temperatures between 70°C and 90°C. PPA serves a dual role here as both a dehydrating agent and a strong acid catalyst, facilitating the formation of the acylium ion which attacks the electron-rich aromatic ring of veratrole. This Friedel-Crafts acylation is highly efficient, yielding the bis-(3,4-dimethoxy-phenyl)-methanone intermediate with an impressive 85% yield. The methoxy groups on the aromatic rings act as strong activating groups, directing subsequent substitutions to specific ortho and para positions, which is fundamental for the regioselectivity observed in the following bromination steps.
Subsequent bromination steps demonstrate exquisite control over reaction conditions to achieve the desired substitution pattern without over-bromination or degradation. The first bromination uses elemental bromine in acetic acid at room temperature to install a bromine atom at the ortho position relative to the carbonyl group. The second step employs aluminum trichloride as a Lewis acid catalyst to activate the ring for further bromination at elevated temperatures (70-90°C), introducing a second bromine atom. Finally, the use of N-bromosuccinimide (NBS) in concentrated sulfuric acid allows for the introduction of the third bromine atom under mild conditions. This mechanistic progression ensures that impurities are minimized at each stage. The rigorous control of stoichiometry, such as maintaining a specific molar ratio of bromine to substrate, prevents the formation of poly-brominated byproducts that would be difficult to separate, thereby ensuring the high purity required for pharmaceutical applications.
How to Synthesize Bis-(2,3,6-tribromo-4,5-dimethoxy-phenyl)-methanone Efficiently
The synthesis of this potent PTP1B inhibitor is achieved through a logical sequence of acylation and halogenation reactions that are well-suited for industrial adaptation. The process begins with the coupling of two methoxylated aromatic precursors to form the central ketone linkage, followed by the systematic introduction of bromine atoms to enhance biological activity. Each step has been optimized for yield and ease of workup, utilizing common organic solvents and straightforward purification techniques like recrystallization and column chromatography. The detailed standardized synthesis steps provided below outline the specific conditions, reagents, and isolation procedures necessary to replicate this high-value intermediate with consistency and precision.
- Perform Friedel-Crafts acylation of veratric acid and veratrole using polyphosphoric acid at 70-90°C to form the dimethoxy ketone core.
- Conduct mono-bromination using elemental bromine in acetic acid at room temperature to introduce the first bromine atom at the ortho position.
- Execute di-bromination using bromine and aluminum trichloride in acetic acid at elevated temperatures to add the second bromine atom.
- Finalize the synthesis via tribromination using N-bromosuccinimide (NBS) in concentrated sulfuric acid to achieve the target hexa-substituted structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from laboratory-scale discovery to commercial manufacturing is often hindered by cost volatility and raw material scarcity. This patented synthesis route addresses these concerns directly by utilizing veratric acid and veratrole as starting materials, which are commodity chemicals available in bulk quantities at competitive prices. The elimination of precious metal catalysts, such as palladium or platinum, which are common in cross-coupling reactions, removes a significant cost driver and supply chain bottleneck. Furthermore, the reliance on standard mineral acids and halogens simplifies the sourcing of reagents, ensuring that production schedules are not disrupted by the lead times associated with specialized fine chemicals. This robustness in raw material sourcing translates directly into enhanced supply chain reliability for downstream drug manufacturers.
- Cost Reduction in Manufacturing: The process achieves cost efficiency not through unverified claims but through tangible chemical engineering principles. By avoiding expensive transition metal catalysts and ligands, the bill of materials is significantly lowered. Additionally, the high yield of the final step (95%) minimizes waste and maximizes the output per batch, effectively reducing the cost per kilogram of the active intermediate. The use of recyclable solvents like acetic acid and the ability to purify intermediates via simple crystallization further contribute to substantial cost savings in the overall production lifecycle.
- Enhanced Supply Chain Reliability: The synthetic route is designed with scalability in mind, utilizing reagents that are stable and easy to transport. Unlike moisture-sensitive organometallic reagents that require special handling and storage, the chemicals used here (PPA, Bromine, NBS) are standard inventory items for most fine chemical facilities. This reduces the risk of production delays caused by reagent degradation or shipping complications. The robustness of the reaction conditions, which tolerate a reasonable range of temperatures and mixing rates, ensures that the process can be transferred between different manufacturing sites without loss of quality, securing a continuous supply of the critical PTP1B inhibitor.
- Scalability and Environmental Compliance: The process generates waste streams that are manageable within standard environmental compliance frameworks. The primary byproducts are inorganic salts and acidic aqueous layers which can be neutralized and treated using conventional wastewater treatment protocols. The absence of heavy metal residues simplifies the purification of the final API, reducing the burden on quality control labs to perform extensive heavy metal testing. This environmental profile facilitates easier regulatory approval and supports sustainable manufacturing practices, which are increasingly important for corporate social responsibility goals in the pharmaceutical industry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this PTP1B inhibitor intermediate. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these details is essential for making informed decisions about integrating this technology into your existing product pipeline or sourcing strategy.
Q: What are the key advantages of this synthesis route for PTP1B inhibitors?
A: The route utilizes cheap and readily available starting materials like veratric acid and veratrole. It avoids complex transition metal catalysts, relying on standard reagents like polyphosphoric acid and NBS, which simplifies purification and reduces heavy metal contamination risks.
Q: How does the final compound compare to existing diabetes treatments?
A: Unlike peptide-based inhibitors which suffer from poor stability, this brominated methanone derivative offers superior chemical stability. Bioassays indicate significant PTP1B inhibitory activity, suggesting potential as a robust insulin sensitizer for Type 2 diabetes therapy.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process demonstrates good scalability with a total yield of approximately 33.8%. The reactions use common solvents like acetic acid and dichloromethane, and the workup procedures involve standard filtration and extraction, making it highly adaptable for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bis-(2,3,6-tribromo-4,5-dimethoxy-phenyl)-methanone Supplier
As the demand for effective Type 2 diabetes treatments continues to grow, the need for high-quality, scalable intermediates like bis-(2,3,6-tribromo-4,5-dimethoxy-phenyl)-methanone becomes paramount. NINGBO INNO PHARMCHEM stands ready to support your development goals with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific reaction conditions required for this synthesis, including the safe handling of brominating agents and strong acids. We adhere to stringent purity specifications and operate rigorous QC labs to ensure that every batch meets the exacting standards required for pharmaceutical grade intermediates, providing you with a partner you can trust for long-term supply continuity.
We invite you to collaborate with us to optimize this synthesis for your specific commercial needs. Our technical team is prepared to provide a Customized Cost-Saving Analysis tailored to your volume requirements, identifying further opportunities for efficiency gains. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. By partnering with NINGBO INNO PHARMCHEM, you secure not just a supplier, but a strategic ally committed to advancing the availability of life-saving diabetes medications through superior chemical manufacturing excellence.
