Advanced Palladium-Catalyzed Synthesis of O-Nitroacetophenone Intermediates for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking safer, more efficient routes to critical intermediates, and the technology disclosed in patent CN101985424A represents a significant leap forward in the synthesis of o-nitroacetophenone compounds. Traditionally, the production of these vital building blocks relied on hazardous mixed-acid nitration or multi-step oxidation processes that posed severe environmental and safety risks. This new methodology fundamentally reimagines the synthetic pathway by employing a palladium-catalyzed carbon-hydrogen (C-H) activation strategy. By converting the carbonyl group of acetophenone into a hydroxyimino directing group, the process enables highly selective mono-nitration at the ortho-position under mild oxidative conditions. For global procurement teams and R&D directors, this innovation translates directly into a reliable o-nitroacetophenone supplier capability that prioritizes both ecological compliance and operational safety, eliminating the generation of waste acid and toxic gases associated with legacy manufacturing protocols.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of ortho-nitrophenyl ethyl ketone has been plagued by significant technical and environmental hurdles that drive up costs and complicate supply chain management. The most common traditional route involves the nitration of ethylbenzene followed by oxidation, a process that requires excessive amounts of strong acids such as nitric and sulfuric acid. This exothermic reaction is difficult to control, often leading to dangerous thermal runaways and the production of large volumes of spent acid waste that requires expensive treatment before disposal. Furthermore, alternative routes starting from aniline involve high-temperature acetylation and oxidation steps which suffer from poor chemo-selectivity, resulting in complex mixtures that are notoriously difficult to purify. These inefficiencies create bottlenecks in cost reduction in pharmaceutical intermediates manufacturing, as the downstream purification and waste management costs often exceed the raw material expenses, making the final product economically volatile and environmentally unsustainable for modern green chemistry standards.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a sophisticated directing group strategy to achieve precise molecular editing. The process begins with readily available acetophenone derivatives, which are converted into oximes and subsequently methylated to form O-methyloximes. This temporary modification serves as a powerful directing group for the palladium catalyst, guiding the nitration exclusively to the ortho-position adjacent to the nitrogen atom. This method allows for the direct functionalization of the aromatic ring without the need for pre-functionalized halides or harsh electrophilic conditions. The versatility of this route is demonstrated by its ability to accommodate various substituents on the phenyl ring, including alkyl, alkoxy, and halogen groups, as illustrated in the general substrate structure below.
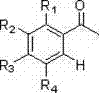
This structural flexibility ensures that a wide range of high-purity OLED material precursors or agrochemical intermediates can be accessed from a single, unified platform, drastically simplifying the inventory management for chemical manufacturers.
Mechanistic Insights into Pd-Catalyzed C-H Activation Nitration
The core of this technological breakthrough lies in the mechanistic elegance of the palladium-catalyzed C-H activation cycle. In the key nitration step, the palladium(II) catalyst, typically palladium diacetate, coordinates with the nitrogen atom of the O-methyloxime directing group. This coordination brings the metal center into close proximity with the ortho-C-H bond, facilitating its cleavage and the formation of a stable palladacycle intermediate. In the presence of an oxidant such as potassium persulfate and a nitrating agent like silver nitrite, the palladium center undergoes oxidation and subsequent reductive elimination to install the nitro group with high fidelity. This mechanism bypasses the typical electrophilic aromatic substitution rules that often lead to mixtures of isomers, ensuring that the reaction proceeds with exceptional regioselectivity. The use of silver nitrite as the nitrogen source is particularly advantageous, as it acts as a mild nitrating agent that minimizes side reactions, while the persulfate oxidant regenerates the active palladium species to sustain the catalytic cycle efficiently.
Furthermore, the robustness of this catalytic system is evidenced by its tolerance towards sensitive functional groups, which is critical for the synthesis of complex drug candidates. For instance, the patent explicitly details the successful synthesis of halogenated derivatives, which are often prone to dehalogenation or side reactions under harsh acidic conditions. As shown in the specific examples of fluorinated and chlorinated products, the method preserves the integrity of the carbon-halogen bonds while installing the nitro group.
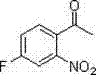
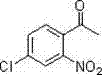
The isolation of 4-fluoro-2-nitroacetophenone and 4-chloro-2-nitroacetophenone in high yields confirms that the electronic effects of electron-withdrawing groups do not inhibit the C-H activation process. This level of impurity control is paramount for R&D directors who require materials with defined impurity profiles to accelerate drug development timelines without the burden of extensive re-crystallization or chromatographic purification.
How to Synthesize O-Nitroacetophenone Efficiently
The practical implementation of this synthesis involves a streamlined five-step sequence that transforms simple acetophenones into valuable nitro-ketones. The process begins with the formation of the oxime using hydroxylamine hydrochloride, followed by O-methylation to activate the directing group. The crucial palladium-catalyzed nitration is then performed in a sealed vessel at elevated temperatures, typically around 130°C, using 1,2-dichloroethane as the solvent. Finally, the directing group is removed via acid hydrolysis to reveal the target ketone. This standardized protocol offers a reproducible pathway for the commercial scale-up of complex pharmaceutical intermediates, ensuring consistent quality across different batch sizes.
- Convert acetophenone raw material into acetophenone oxime using hydroxylamine hydrochloride and alkali in alcohol solvent under reflux.
- Methylate the oxime intermediate using methyl iodide and inorganic strong alkali in a DMSO/water mixture to form O-methyloxime.
- Perform regioselective ortho-nitration using palladium diacetate catalyst, silver nitrite, and potassium persulfate oxidant in 1,2-dichloroethane.
- Hydrolyze the nitrated O-methyloxime intermediate using strong protonic acid to regenerate the ketone carbonyl group.
- Isolate the final o-nitroacetophenone product through neutralization, extraction, and purification via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers transformative benefits that extend far beyond simple yield improvements. By eliminating the need for mixed acid nitration, the process removes the substantial logistical and financial burdens associated with handling, storing, and disposing of corrosive hazardous wastes. This shift not only reduces the regulatory compliance overhead but also mitigates the risk of production stoppages due to environmental violations. The simplified workup procedure, which relies on standard extraction and chromatography rather than complex distillation or neutralization of bulk acids, allows for faster turnaround times between batches. Consequently, this leads to a drastic simplification of the manufacturing workflow, enabling suppliers to respond more agilely to market demand fluctuations without compromising on safety or quality standards.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven primarily by the elimination of expensive waste treatment protocols and the reduction of raw material consumption. Traditional nitration generates vast quantities of spent acid that must be neutralized and treated, incurring significant operational expenditures. By replacing this with a catalytic system that utilizes stoichiometric amounts of benign oxidants and nitrating agents, the overall cost of goods sold is substantially lowered. Additionally, the high regioselectivity of the reaction minimizes the formation of isomeric byproducts, which reduces the loss of material during purification and increases the overall throughput of the facility. This efficiency gain translates directly into competitive pricing for downstream customers seeking cost-effective sourcing solutions.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the reliance on specialized reagents or hazardous processing conditions that limit the number of qualified manufacturers. This method utilizes widely available starting materials such as acetophenone and common reagents like methyl iodide and potassium persulfate, which are easily sourced from multiple global vendors. The robustness of the reaction conditions means that production is less susceptible to interruptions caused by minor variations in temperature or reagent quality. This resilience ensures a stable supply of high-purity pharmaceutical intermediates, allowing partners to maintain lean inventory levels with confidence that their supply lines will remain uninterrupted even during periods of market volatility.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to industrial production often introduces new safety and environmental challenges, but this pathway is inherently designed for scalability. The absence of gas evolution and the use of liquid-phase reagents in a closed system make it ideal for large-scale reactor operations. Furthermore, the process aligns perfectly with modern green chemistry principles by avoiding the generation of toxic emissions and wastewater. This environmental compatibility facilitates easier permitting and regulatory approval in jurisdictions with strict environmental laws, thereby future-proofing the supply chain against tightening regulations and ensuring long-term operational viability for sustainable manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this synthesis route. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on the operational parameters and expected outcomes. Understanding these details is essential for technical teams evaluating the feasibility of integrating this technology into their existing production pipelines.
Q: How does this method improve safety compared to traditional nitration?
A: Traditional methods utilize mixed acid (nitric and sulfuric acid) which generates excessive heat and hazardous waste gas. This patent utilizes a palladium-catalyzed C-H activation pathway that operates under controlled heating without generating waste acid or toxic gases, significantly enhancing operational safety.
Q: What is the regioselectivity of this nitration process?
A: The process exhibits excellent regioselectivity for the ortho-position relative to the carbonyl group. By converting the ketone to an oxime ether directing group, the palladium catalyst selectively activates the adjacent C-H bond, preventing the formation of meta- or para- isomers common in electrophilic aromatic substitution.
Q: Can this synthesis tolerate halogen substituents on the phenyl ring?
A: Yes, the method demonstrates broad substrate adaptability. Specific embodiments in the patent successfully synthesize 4-fluoro-2-nitroacetophenone and 4-chloro-2-nitroacetophenone with high yields, proving compatibility with halogenated acetophenones which are critical for downstream pharmaceutical coupling reactions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable O-Nitroacetophenone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic methodologies requires a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in patent literature can be reliably translated into industrial reality. We are committed to maintaining stringent purity specifications through our rigorous QC labs, utilizing state-of-the-art analytical equipment to verify the identity and purity of every batch of o-nitroacetophenone derivatives we produce. Our dedication to quality assurance means that every shipment meets the exacting standards required by the global pharmaceutical industry.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced palladium-catalyzed synthesis can enhance your product portfolio and drive your business forward.
