Advanced Silver-Catalyzed Synthesis of Alpha-Hydroxy Alkenyl Azides for Commercial Scale-Up
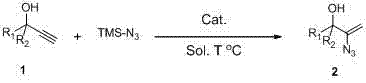
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to access nitrogen-containing heterocycles, which serve as critical scaffolds in modern drug design. Patent CN103664686A introduces a transformative approach to synthesizing α-hydroxyalkenyl azide compounds, addressing long-standing challenges in safety and efficiency. This technology leverages a silver-catalyzed hydroazidation strategy that converts readily available propargylic alcohols directly into valuable azide intermediates in a single operational step. By utilizing stable reagents like trimethylsilyl azide (TMS-N3) instead of hazardous hydrazoic acid, this method significantly lowers the barrier for entry regarding safety infrastructure while maintaining high stereoselectivity. For R&D teams focused on developing novel bioactive molecules, this pathway offers a reliable entry point into complex nitrogenous architectures without the logistical burdens associated with traditional azide chemistry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of alkenyl azides has been plagued by significant safety hazards and operational complexities that hinder large-scale adoption. Traditional routes often rely on the condensation of aldehydes with ethyl α-azidoacetate under alkaline conditions, a method severely restricted to substrates bearing electron-withdrawing groups, thus limiting its utility in diverse medicinal chemistry campaigns. Alternatively, the addition-elimination reaction involving hydrazoic acid or its equivalents presents extreme safety risks due to the explosive nature of hydrazoic acid, requiring specialized containment facilities and rigorous safety protocols that drive up capital expenditure. Furthermore, these conventional pathways frequently involve multi-step sequences with harsh reaction conditions, leading to lower overall yields and generating substantial waste streams that complicate environmental compliance. The inability to efficiently process non-activated alkynes or those lacking specific electronic activation has created a bottleneck in the supply chain for high-purity alkenyl azide intermediates.
The Novel Approach
The methodology disclosed in the patent represents a paradigm shift by enabling the direct transformation of non-activated propargylic alcohols into α-hydroxyalkenyl azides using a simple silver catalytic system. This one-pot procedure eliminates the need for pre-functionalized starting materials or dangerous gaseous reagents, streamlining the workflow from raw material to isolated product. The use of trimethylsilyl azide as a stable nitrogen source allows reactions to proceed under relatively mild thermal conditions, ranging from ambient temperature to 120°C depending on the substrate, which preserves sensitive functional groups often found in advanced intermediates. Moreover, the reaction exhibits remarkable stereospecificity, driven by the directing effect of the α-hydroxyl group, ensuring the production of structurally defined products essential for consistent biological activity. This approach not only simplifies the synthetic route but also enhances the economic viability of producing these high-value building blocks for commercial applications.
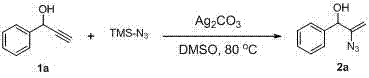
Mechanistic Insights into Silver-Catalyzed Hydroazidation
The core of this innovation lies in the activation of the alkyne moiety by silver species, which facilitates the nucleophilic attack of the azide source in a highly controlled manner. The silver catalyst, whether in the form of carbonate, nitrate, or fluoride, coordinates with the triple bond of the propargylic alcohol, increasing its electrophilicity and lowering the energy barrier for azide addition. Crucially, the adjacent hydroxyl group participates in the transition state, likely through hydrogen bonding or coordination, which directs the regioselectivity and ensures the formation of the specific vinyl azide isomer rather than a mixture of regioisomers. This mechanistic feature is vital for maintaining product purity, as it minimizes the formation of side products that would otherwise require costly and yield-loss-inducing purification steps. The tolerance of the catalytic system to various solvents, including DMSO, DMF, and toluene, further indicates a robust mechanism that is not overly sensitive to minor variations in reaction media, making it adaptable to different manufacturing environments.
Impurity control is inherently built into this catalytic cycle due to the specificity of the silver-alkyne interaction and the stability of the TMS-N3 reagent. Unlike free hydrazoic acid which can lead to uncontrolled polymerization or explosive decomposition, the silylated azide reacts cleanly under the catalytic influence, resulting in a cleaner crude reaction profile. The absence of strong acids or bases in the primary transformation step prevents the degradation of acid- or base-sensitive substituents on the aromatic or heteroaromatic rings of the substrate. This high level of chemoselectivity means that downstream processing is simplified, often requiring only standard extraction and chromatography to achieve analytical purity. For process chemists, understanding this mechanism allows for the rational optimization of catalyst loading and temperature to maximize throughput while minimizing the generation of hazardous waste, aligning with green chemistry principles.
How to Synthesize Alpha-Hydroxy Alkenyl Azides Efficiently
Implementing this synthesis requires careful attention to reagent stoichiometry and thermal management to ensure optimal conversion rates. The general protocol involves dissolving the propargylic alcohol substrate and the azide source in a suitable polar aprotic solvent, followed by the introduction of the silver catalyst under controlled heating. Detailed procedural parameters, including specific molar ratios and workup techniques, are critical for reproducing the high yields reported in the patent literature. Operators should note that reaction times can vary significantly based on the electronic nature of the substrate, necessitating real-time monitoring via thin-layer chromatography to prevent over-reaction. The following guide outlines the standardized operational framework derived from the patent examples to assist technical teams in process validation.
- Combine propargylic alcohol substrate and trimethylsilyl azide (TMS-N3) in a polar aprotic solvent such as DMSO or DMF within a reaction vessel equipped with magnetic stirring.
- Heat the mixture to a temperature range between 25°C and 120°C depending on substrate reactivity, then introduce the silver catalyst (e.g., Ag2CO3, AgNO3, or AgF).
- Monitor reaction progress via TLC until substrate consumption is complete, typically within 1 to 8 hours, followed by aqueous workup and silica gel chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this technology offers substantial strategic benefits by decoupling production from volatile and hazardous supply chains associated with traditional azide reagents. The reliance on stable, commercially available starting materials like propargylic alcohols and TMS-N3 ensures a consistent supply flow, mitigating the risk of production stoppages due to raw material shortages or regulatory restrictions on explosive precursors. Additionally, the simplification of the synthetic route from multiple steps to a single catalytic transformation drastically reduces the consumption of solvents and energy, leading to a leaner manufacturing footprint. This efficiency translates directly into improved margin potential, as fewer unit operations mean lower labor costs and reduced equipment occupancy time, allowing for higher throughput in existing facilities without the need for major capital investment.
- Cost Reduction in Manufacturing: The elimination of hazardous hydrazoic acid removes the necessity for expensive explosion-proof reactors and specialized safety containment systems, resulting in significant capital expenditure savings. Furthermore, the high atom economy of the direct addition reaction minimizes raw material waste, ensuring that a greater proportion of input costs are converted into saleable product value. By avoiding multi-step sequences, the process reduces the cumulative loss of yield typically seen in longer synthetic routes, effectively lowering the cost per kilogram of the final intermediate. The use of common silver salts as catalysts, which can potentially be recovered or used in low loadings, further optimizes the cost structure compared to precious metal catalysts that require complex recycling protocols.
- Enhanced Supply Chain Reliability: Sourcing stable reagents like trimethylsilyl azide is far less logistically challenging than managing the transport and storage of explosive gases, thereby simplifying vendor qualification and inventory management. The robustness of the reaction conditions allows for flexibility in manufacturing locations, as the process does not demand extreme cryogenic conditions or ultra-high pressure equipment that might be unavailable in certain regions. This adaptability strengthens the resilience of the supply chain against geopolitical or logistical disruptions, ensuring continuous availability of critical intermediates for downstream API production. Moreover, the broad substrate scope means that a single manufacturing platform can produce a wide variety of derivatives, reducing the need for dedicated production lines for each specific molecule.
- Scalability and Environmental Compliance: The reaction operates in standard organic solvents such as DMSO and DMF, which are well-understood in industrial waste treatment processes, facilitating easier compliance with environmental regulations. The absence of heavy metal contaminants often associated with other catalytic systems simplifies the purification process, reducing the burden on wastewater treatment facilities and lowering disposal costs. Scaling this process from laboratory to commercial tonnage is straightforward due to the homogeneous nature of the catalysis and the lack of exothermic runaway risks typical of azide chemistry. This scalability ensures that the technology can meet the demands of both clinical trial material production and full-scale commercial manufacturing without requiring fundamental process redesigns.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this silver-catalyzed synthesis platform. These insights are derived directly from the experimental data and scope defined in the patent documentation to provide clarity for potential partners. Understanding these nuances is essential for evaluating the fit of this technology within your specific development pipeline.
Q: What are the safety advantages of using TMS-N3 over hydrazoic acid in this synthesis?
A: The patented method utilizes trimethylsilyl azide (TMS-N3), which is significantly more stable and safer to handle than explosive hydrazoic acid, thereby reducing facility safety requirements and operational risks.
Q: Does this silver-catalyzed method support diverse substrate scopes for drug discovery?
A: Yes, the process demonstrates excellent compatibility with various R1 and R2 groups including aryl, heteroaryl, alkyl, and cycloalkenyl moieties, allowing for the rapid generation of diverse chemical libraries.
Q: How does the alpha-hydroxyl group influence the stereochemistry of the product?
A: The presence of the alpha-hydroxyl group is critical for directing the addition reaction, ensuring the formation of stereospecific alkenyl azide derivatives which are essential for downstream heterocycle synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Hydroxy Alkenyl Azide Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating innovative academic research into commercial reality, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is adept at optimizing the silver-catalyzed hydroazidation process to meet stringent purity specifications required by global regulatory bodies, ensuring that every batch delivers consistent quality for your critical applications. We maintain rigorous QC labs equipped with advanced analytical instrumentation to verify the stereospecificity and purity of our alpha-hydroxy alkenyl azide intermediates, providing you with the confidence needed to advance your drug candidates. Our commitment to excellence extends beyond mere synthesis, as we offer comprehensive support in process safety assessment and regulatory documentation to streamline your path to market.
We invite you to engage with our technical procurement team to discuss how this advanced synthetic methodology can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this more efficient route for your target molecules. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions based on hard data and proven capability. Let us collaborate to accelerate your development timelines and secure a competitive advantage in the marketplace through superior chemical manufacturing solutions.
