Advanced Synthesis of Chiral 4-Halotryptophan Derivatives for Commercial Scale-Up
The pharmaceutical industry continuously demands high-purity chiral building blocks to ensure the efficacy and safety of complex drug molecules, particularly those containing indole alkaloid structures. A recent technological breakthrough documented in patent CN112409235B introduces a robust synthetic methodology for producing chiral 4-halogenated tryptophan derivatives, which serve as critical precursors for bioactive compounds like lysergic acid and various indole alkaloids. This innovation addresses the longstanding challenges of low enantioselectivity and harsh reaction conditions associated with traditional manufacturing routes. By leveraging a specialized rhodium-catalyzed asymmetric hydrogenation strategy, the process delivers exceptional optical purity with yields reaching 95% and enantiomeric excess values up to 99.9%. For global procurement teams and R&D directors, this represents a pivotal shift towards more sustainable and economically viable supply chains for high-value pharmaceutical intermediates. The ability to access these complex chiral structures reliably is essential for accelerating drug development pipelines and ensuring consistent quality in final API production.
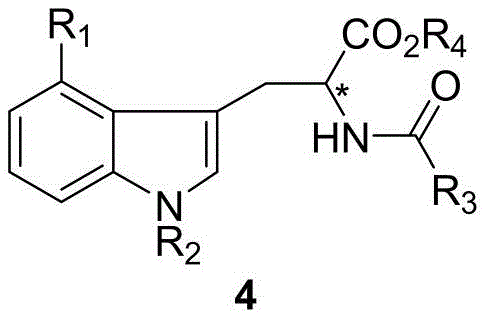
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of optically pure 4-halotryptophan derivatives has been plagued by inefficient methodologies that hinder large-scale commercial adoption. Traditional approaches often rely on palladium-catalyzed ring-closure reactions or nickel-catalyzed reductive couplings, which necessitate the use of stoichiometric amounts of expensive transition metals. These legacy processes frequently require rigorous anhydrous and oxygen-free environments, imposing severe operational constraints and increasing safety risks in a manufacturing setting. Furthermore, earlier methods utilizing chiral auxiliaries introduce additional synthetic steps for attachment and removal, thereby elongating the production timeline and generating substantial chemical waste. The cumulative effect of these inefficiencies is a significant escalation in production costs and a reduction in overall process throughput. Additionally, the enantioselectivity achieved by conventional catalytic asymmetric hydrogenation using ligands like DIPAMP often plateaus at around 94% ee, which is insufficient for stringent pharmaceutical specifications without further purification.
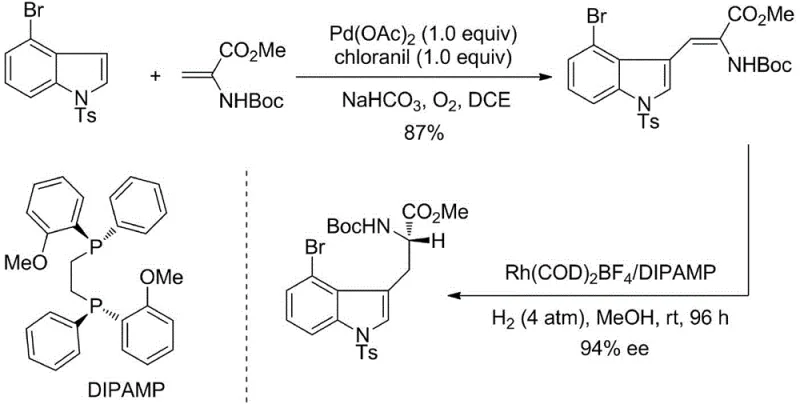
The Novel Approach
In stark contrast to these outdated techniques, the novel approach outlined in the patent data utilizes a streamlined three-step sequence that eliminates the need for stoichiometric metal reagents and complex chiral auxiliaries. This modern methodology employs a highly efficient condensation reaction followed by a mild alcoholysis ring-opening step to construct the key dehydrotryptophan intermediate. The cornerstone of this innovation is the subsequent asymmetric hydrogenation step, which utilizes a specialized rhodium catalyst coordinated with a Duanphos ligand system. This catalytic system operates effectively under relatively mild temperatures ranging from 30°C to 80°C and moderate hydrogen pressures, significantly reducing energy consumption and equipment stress. The result is a process that not only simplifies the operational workflow but also dramatically enhances the stereochemical outcome, consistently delivering products with superior optical purity. This strategic shift from stoichiometric to catalytic processes fundamentally alters the economic landscape of producing these valuable intermediates.
Mechanistic Insights into Rh-Catalyzed Asymmetric Hydrogenation
The core of this technological advancement lies in the sophisticated mechanistic pathway of the rhodium-catalyzed asymmetric hydrogenation, which dictates the high fidelity of the chiral center formation. The catalyst system, typically comprising a rhodium precursor such as Rh(NBD)2BF4 and the chiral bisphosphine ligand Duanphos, forms a highly active cationic complex in situ. This complex coordinates with the olefinic bond of the dehydrotryptophan precursor, facilitating the stereoselective addition of hydrogen across the double bond. The unique steric and electronic properties of the Duanphos ligand create a chiral environment that strongly favors the formation of one enantiomer over the other, thereby suppressing the generation of unwanted isomers. This precise control over the reaction trajectory is crucial for minimizing impurity profiles and reducing the burden on downstream purification processes. By optimizing the molar ratio of the catalyst to the substrate, the process ensures maximum turnover frequency while maintaining the integrity of the sensitive indole ring structure throughout the transformation.
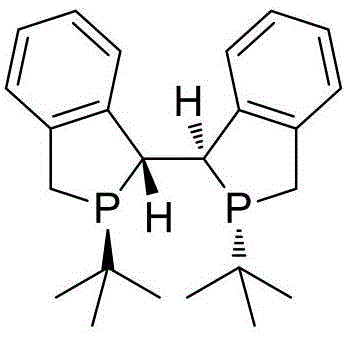
Impurity control is inherently built into the design of this synthetic route, addressing a major concern for R&D directors focused on regulatory compliance and product safety. The initial condensation step utilizes readily available reagents like hippuric acid and acetic anhydride, which react cleanly to form the oxazolone intermediate with minimal side products. Subsequent ring-opening with metal alkoxides proceeds under controlled conditions that prevent racemization or degradation of the sensitive halogenated indole moiety. The final hydrogenation step is particularly effective at avoiding the formation of over-reduced byproducts or dehalogenated impurities, which are common pitfalls in transition metal catalysis. The high selectivity of the rhodium-Duanphos system ensures that the halogen substituent at the 4-position remains intact, preserving the chemical functionality required for subsequent coupling reactions in drug synthesis. This robust impurity profile translates directly into higher quality raw materials for API manufacturers and reduces the risk of batch rejection during quality control testing.
How to Synthesize Chiral 4-Halotryptophan Efficiently
Implementing this synthesis route requires careful attention to reaction parameters to replicate the high yields and selectivity reported in the patent literature. The process begins with the preparation of the oxazolone intermediate, followed by conversion to the dehydrotryptophan ester, and concludes with the critical asymmetric hydrogenation step. Operators must maintain strict control over temperature and pressure during the hydrogenation phase to ensure optimal catalyst performance and safety. The following guide outlines the standardized operational framework derived from the patent examples, serving as a foundational reference for process chemists aiming to adopt this technology. Detailed standard operating procedures and specific parameter adjustments for different scales are essential for successful technology transfer.
- Condense 4-haloindole-3-carbaldehyde with N-acylglycine derivative using acid anhydride and carboxylate to form the oxazolone intermediate.
- Perform alcoholysis ring-opening of the oxazolone intermediate using metal alkoxides in alcohol solvent to generate the dehydrotryptophan precursor.
- Execute catalytic asymmetric hydrogenation using a Rhodium-Duanphos complex under hydrogen pressure to achieve high enantioselectivity.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthesis method offers transformative advantages that directly impact the bottom line and supply chain resilience for pharmaceutical manufacturers. By transitioning from stoichiometric palladium methods to a catalytic rhodium system, the process eliminates the need for expensive metal scavenging steps and reduces the overall consumption of precious metal resources. This shift results in substantial cost savings in raw material procurement and waste disposal, making the production of these intermediates more economically sustainable in the long term. Furthermore, the simplified three-step sequence reduces the total manufacturing cycle time, allowing suppliers to respond more rapidly to fluctuating market demands and urgent procurement requests. The robustness of the reaction conditions also enhances supply chain reliability by minimizing the risk of batch failures due to sensitive environmental requirements.
- Cost Reduction in Manufacturing: The elimination of stoichiometric metal reagents and chiral auxiliaries significantly lowers the direct material costs associated with producing chiral 4-halotryptophan derivatives. Traditional methods often incur high expenses related to the purchase and recovery of palladium or nickel, as well as the multi-step synthesis of chiral starting materials. In contrast, the catalytic nature of this new approach allows for the reuse of catalyst systems and reduces the volume of hazardous waste generated per kilogram of product. These efficiencies compound over large production volumes, leading to drastic improvements in profit margins for contract manufacturing organizations. Additionally, the avoidance of complex chromatographic purification steps further reduces solvent consumption and labor costs, contributing to a leaner and more cost-effective manufacturing model.
- Enhanced Supply Chain Reliability: The use of commercially available and stable starting materials ensures a consistent supply of raw inputs, mitigating the risk of production delays caused by sourcing bottlenecks. Conventional routes often depend on specialized chiral pools or custom-synthesized precursors that may have limited availability or long lead times. This new methodology relies on bulk chemicals like hippuric acid and simple halogenated indoles, which are widely accessible from multiple global suppliers. The operational simplicity of the process also means that it can be easily replicated across different manufacturing sites, providing redundancy and flexibility in the supply network. This reliability is critical for maintaining continuous production schedules for downstream API manufacturing and meeting strict delivery commitments to pharmaceutical clients.
- Scalability and Environmental Compliance: The mild reaction conditions and high atom economy of this synthesis make it exceptionally well-suited for scaling from laboratory benchtop to multi-ton commercial production. Unlike processes requiring cryogenic temperatures or high-pressure reactors beyond standard capabilities, this method operates within the range of typical industrial chemical equipment. The reduction in hazardous waste and the use of greener solvents align with increasingly stringent environmental regulations and corporate sustainability goals. This compliance reduces the regulatory burden on manufacturers and minimizes the risk of shutdowns due to environmental violations. Consequently, companies adopting this technology can scale their operations confidently, knowing that the process is both environmentally responsible and industrially robust.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this patented synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation. Understanding these details helps stakeholders evaluate the feasibility of integrating this route into their existing manufacturing portfolios. The answers reflect the consensus on performance metrics and operational advantages observed during the development of this method.
Q: What is the enantiomeric excess achieved in this synthesis?
A: The patented method achieves an enantiomeric excess (ee) of up to 99.9%, which significantly exceeds the 94% ee limit of conventional palladium-catalyzed methods.
Q: Does this process require stoichiometric amounts of expensive metals?
A: No, unlike prior art requiring stoichiometric palladium, this method utilizes a catalytic amount of Rhodium complex, drastically reducing raw material costs and heavy metal residue risks.
Q: Is this synthesis suitable for industrial scale-up?
A: Yes, the process operates under mild conditions (30-80°C) and avoids complex column chromatography, making it highly suitable for commercial scale-up from kilograms to tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral 4-Halotryptophan Derivative Supplier
At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate this advanced synthetic methodology into commercial reality for our global partners. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high purity and yield demonstrated in the lab are maintained at an industrial level. We operate stringent purity specifications and utilize rigorous QC labs to verify that every batch meets the exacting standards required for pharmaceutical intermediates. Our commitment to quality assurance means that clients can rely on us for consistent supply of high-enantiomeric excess materials essential for their drug development programs.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can optimize your supply chain and reduce costs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your volume requirements. We encourage potential partners to contact us for specific COA data and route feasibility assessments to validate the suitability of this technology for your projects. Let us collaborate to bring high-quality chiral intermediates to market faster and more efficiently.
