Scalable Production of High-Performance 2,7-Dibromo-9-Hydroxyl Phenanthrene Derivatives for Next-Gen OLEDs
The rapid evolution of organic electroluminescent materials has positioned blue-emitting compounds as a critical bottleneck in the development of next-generation flat panel displays. While red and green emitters have achieved commercial viability, stable and efficient blue materials remain a significant challenge due to issues with thermal stability and color purity. Addressing this gap, patent CN102775279A discloses a novel series of 2,7-dibromo-9-hydroxyl phenanthrene derivatives designed specifically to overcome these limitations. By strategically introducing large substituent groups at the 2, 7, and 9 positions, and incorporating a bulky group at the 10-position, this technology effectively minimizes intermolecular accumulation. This structural innovation results in superior electrochemical performance, enhanced thermal stability, and improved blue luminescence properties, making these derivatives ideal candidates for high-end organic light-emitting diode (OLED) applications.
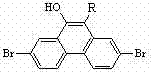
The core value of this invention lies in its ability to tailor the molecular architecture for optimal device performance. Traditional phenanthrene derivatives often suffer from shallow blue emission or complex synthesis requirements when attempting to modify the 9,10-positions. In contrast, the derivatives described in this patent utilize a streamlined synthetic pathway that allows for precise control over the substitution pattern. The variable group R, which can be a phenyl, C1-5 alkyl substituted phenyl, or biphenyl group, provides flexibility in tuning the material's properties. For procurement managers and supply chain leaders in the electronic chemicals sector, this represents a reliable source of high-purity OLED material precursors that can be manufactured with consistent quality and scalability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art in the field of blue OLED host materials has struggled to balance synthetic feasibility with performance metrics. For instance, documents such as WO04/016575 describe 3,6-dinaphthylphenanthrene compounds; however, the lack of substitution at the 9,10-positions in these molecules leads to undesirable shallow blue luminescence, rendering them suboptimal for high-quality displays. Similarly, JP2001/332384 details aryl-substituted phenanthrenes including 3,6,9,10-tetraphenyl variants, yet the 3,6-substitution pattern remains a persistent drawback affecting emission color purity. Furthermore, approaches like those in JP2003/055276 attempt to fuse bicyclic aliphatic groups at the 9,10-positions to improve stability, but this necessitates highly complex and costly synthetic routes that are difficult to scale. These conventional methods often involve multi-step sequences with low overall yields, expensive catalysts, and difficult purification processes, creating significant barriers to cost reduction in electronic chemical manufacturing.
The Novel Approach
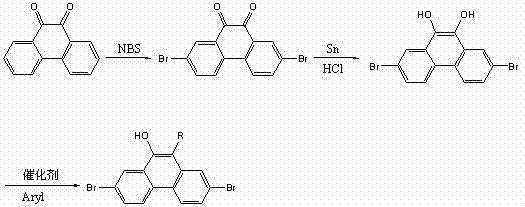
The methodology presented in CN102775279A offers a transformative solution by shifting the substitution strategy to the 2, 7, and 9 positions while introducing a large steric group at the 10-position. This approach not only resolves the emission color issues associated with 3,6-substitution but also simplifies the synthetic pathway significantly. The process begins with readily available 9,10-phenanthrenequinone and proceeds through a robust three-stage sequence: selective bromination, reduction, and acid-catalyzed condensation. This route avoids the need for complex fused ring systems and utilizes common industrial reagents. As illustrated in the reaction scheme below, the transformation is direct and efficient, enabling the production of diverse derivatives by simply varying the aromatic reactant in the final step.
This novel approach drastically simplifies the supply chain for complex OLED intermediates. By eliminating the need for exotic starting materials or harsh reaction conditions typically found in legacy processes, manufacturers can achieve higher throughput with lower operational risks. The ability to use aromatic compounds like benzene, toluene, or biphenyl directly in the final condensation step further enhances the versatility of the platform. For R&D directors evaluating new material pipelines, this chemistry offers a clear path to high-purity products with minimal impurity profiles, ensuring that the final electronic materials meet the stringent specifications required for commercial display panels.
Mechanistic Insights into Acid-Catalyzed Condensation and Functionalization
The synthesis mechanism relies on a sequence of well-controlled electrophilic substitutions and reductions that ensure high regioselectivity. The initial step involves the bromination of 9,10-phenanthrenequinone using N-bromosuccinimide (NBS) in concentrated sulfuric acid. This reaction selectively targets the 2 and 7 positions of the phenanthrene backbone, driven by the electronic activation of the quinone system. Following isolation, the 2,7-dibromophenanthrenequinone undergoes reduction using tin (Sn) and concentrated hydrochloric acid in glacial acetic acid. This step converts the quinone moiety into a 9,10-dihydroxy structure, which is crucial for the subsequent condensation reaction. The final stage is an acid-catalyzed dehydration condensation where the 2,7-dibromo-9,10-dihydroxyphenanthrene reacts with an aromatic compound in the presence of a strong acid catalyst such as trifluoromethanesulfonic acid. This reaction likely proceeds via a carbocation intermediate generated from the aromatic reactant or the dihydroxy species, leading to the formation of the C-C bond at the 10-position and the elimination of water.
Impurity control is intrinsic to this mechanistic design. The use of concentrated sulfuric acid in the bromination step ensures high conversion rates, minimizing unreacted starting material. The reduction step is performed under reflux conditions which drive the reaction to completion, yielding intermediates with purity greater than 98%. In the final condensation, the choice of catalyst and temperature (optimized between 60-120°C) allows for precise control over the reaction kinetics, preventing over-alkylation or polymerization side reactions. The workup procedure, involving simple water washing and solvent evaporation followed by filtration, effectively removes inorganic salts and catalyst residues. This robust mechanism ensures that the final 2,7-dibromo-9-hydroxyl phenanthrene derivatives possess the high thermal stability and electrochemical performance necessary for demanding optoelectronic applications, with experimental data showing decomposition temperatures around 330°C.
How to Synthesize 2,7-Dibromo-9-Hydroxyl Phenanthrene Derivatives Efficiently
The synthesis of these high-value OLED intermediates is designed for operational simplicity and scalability. The process integrates three distinct chemical transformations that can be executed in standard glass-lined or stainless steel reactors commonly found in fine chemical facilities. The protocol emphasizes the use of stoichiometric control and temperature management to maximize yield and purity. For example, the molar ratio of the dihydroxy intermediate to the aromatic reactant is optimized at approximately 1:1.2 to ensure complete consumption of the valuable phenanthrene core while minimizing excess reagent waste. The following guide outlines the standardized operational framework derived from the patent examples, providing a clear roadmap for technical teams aiming to implement this technology.
- Bromination of 9,10-phenanthrenequinone using N-bromosuccinimide (NBS) in concentrated sulfuric acid to yield 2,7-dibromophenanthrenequinone.
- Reduction of the quinone intermediate using tin (Sn) and concentrated hydrochloric acid in glacial acetic acid under reflux to form 2,7-dibromo-9,10-dihydroxyphenanthrene.
- Condensation of the dihydroxy intermediate with aromatic compounds (benzene, toluene, or biphenyl) using trifluoromethanesulfonic acid catalyst at 60-120°C.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route offers substantial strategic benefits beyond mere technical performance. The primary advantage lies in the significant simplification of the raw material portfolio. Unlike legacy methods that require custom-synthesized fused ring systems or rare organometallic reagents, this process utilizes commodity chemicals such as 9,10-phenanthrenequinone, N-bromosuccinimide, tin, and common aromatic solvents. This shift drastically reduces the risk of supply chain disruptions and mitigates price volatility associated with specialty reagents. Furthermore, the high yields reported in the patent examples, such as 96% for the reduction step and up to 90% for the final condensation, translate directly into improved material efficiency and lower cost of goods sold (COGS). The elimination of complex purification steps like column chromatography in favor of recrystallization and filtration further drives down processing costs and waste generation.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the use of inexpensive and widely available starting materials. By avoiding the need for precious metal catalysts or complex ligand systems often required in cross-coupling reactions for similar structures, the direct material costs are significantly lowered. Additionally, the high atom economy of the condensation step, which primarily eliminates water as a byproduct, minimizes waste disposal costs. The streamlined workflow reduces the number of unit operations required, leading to lower energy consumption and labor costs per kilogram of finished product. This efficiency allows for competitive pricing strategies in the highly sensitive electronic materials market without compromising on quality.
- Enhanced Supply Chain Reliability: Sourcing reliability is a critical metric for any long-term supply agreement. Since the key reagents like NBS, tin, and hydrochloric acid are produced on a massive global scale, the risk of shortage is negligible. The process does not rely on single-source suppliers for exotic intermediates, providing procurement teams with the flexibility to negotiate better terms and ensure continuity of supply. The robustness of the reaction conditions, which tolerate standard industrial equipment and do not require extreme pressures or cryogenic temperatures, further ensures that production can be maintained consistently across different manufacturing sites. This resilience is essential for meeting the rigorous delivery schedules of major display panel manufacturers.
- Scalability and Environmental Compliance: Scaling this chemistry from laboratory to commercial production is straightforward due to the absence of hazardous exotherms or unstable intermediates. The reactions are conducted in common solvents like glacial acetic acid, toluene, or tetrahydrofuran, which are easily recovered and recycled, supporting sustainable manufacturing practices. The solid byproducts, primarily inorganic salts from the reduction step, are easy to separate and dispose of in compliance with environmental regulations. The high purity of the crude product reduces the need for extensive solvent-intensive purification, thereby lowering the overall environmental footprint. This alignment with green chemistry principles facilitates smoother regulatory approvals and enhances the corporate sustainability profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 2,7-dibromo-9-hydroxyl phenanthrene derivatives. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity for stakeholders evaluating this technology for integration into their supply chains. Understanding these details is crucial for assessing the feasibility of adopting this material for next-generation blue OLED projects.
Q: How does the 2,7,9-substitution pattern improve OLED performance?
A: The specific substitution at the 2, 7, and 9 positions, combined with a bulky group at the 10-position, effectively reduces intermolecular stacking interactions. This structural modification enhances thermal stability, improves blue luminescence efficiency, and increases molecular solubility compared to traditional 3,6-substituted phenanthrenes.
Q: What are the typical purity levels achievable with this synthesis route?
A: The patented process utilizes straightforward purification methods such as recrystallization and filtration. Experimental data indicates that final products can consistently achieve purity levels greater than 98%, with some examples reaching over 99% purity, which is critical for high-performance electronic applications.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the synthesis relies on readily available raw materials like 9,10-phenanthrenequinone and common reagents such as NBS and tin. The reaction conditions are moderate (30-150°C), and the workup involves standard unit operations like filtration and solvent evaporation, making it highly amenable to commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,7-Dibromo-9-Hydroxyl Phenanthrene Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance intermediates play in the advancement of organic electronics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and reliability. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Whether you require custom derivatives with specific R groups or bulk quantities of the standard 2,7-dibromo-9-hydroxyl phenanthrene core, our flexible manufacturing capabilities allow us to adapt quickly to your evolving requirements while maintaining the highest standards of quality and safety.
We invite you to collaborate with us to optimize your material sourcing strategy. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced synthesis platform can enhance your supply chain efficiency and reduce your overall manufacturing costs. Let us be your partner in bringing the next generation of blue light emitting materials to market.
