Advanced Pyrazinoindanone Electroluminescent Materials: Scalable Synthesis and Commercial Viability
Advanced Pyrazinoindanone Electroluminescent Materials: Scalable Synthesis and Commercial Viability
The rapid evolution of the display industry towards Organic Light-Emitting Diodes (OLEDs) has necessitated the development of third-generation luminescent materials that overcome the theoretical efficiency limits of traditional fluorescent emitters. Patent CN110615783B introduces a novel class of electroluminescent materials utilizing a pyrazinoindanone core as a robust electron acceptor, coupled with various electron-donating groups to achieve Thermally Activated Delayed Fluorescence (TADF). This technological breakthrough addresses the critical industry demand for materials that offer high luminous efficiency, excellent color purity, and simplified molecular design compared to complex phosphorescent systems. By strategically modifying the 2 and 8 positions of the pyrazinoindanone scaffold, researchers have created a versatile platform for tuning emission wavelengths and energy levels, positioning these derivatives as prime candidates for next-generation full-color displays and solid-state lighting applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the OLED industry has relied heavily on first-generation fluorescent materials and second-generation phosphorescent materials, both of which present significant drawbacks for mass manufacturing and performance optimization. Fluorescent materials are fundamentally limited by spin statistics, where only 25% of excitons (singlets) contribute to light emission, capping the internal quantum efficiency at a theoretical maximum of 25%. On the other hand, while phosphorescent materials can harvest both singlet and triplet excitons to reach 100% efficiency, they depend on scarce and prohibitively expensive noble metals like iridium and platinum. Furthermore, phosphorescent emitters often suffer from severe efficiency roll-off at high brightness due to triplet-triplet annihilation, and their long emission lifetimes can lead to device instability. These factors create substantial supply chain vulnerabilities and cost barriers for large-area panel production, driving the urgent need for metal-free alternatives that do not compromise on performance.
The Novel Approach
The synthetic strategy outlined in the patent data offers a transformative solution by employing a metal-free TADF architecture centered on a pyrazinoindanone acceptor. The core innovation lies in the modular construction of the molecule, where the electron-deficient pyrazinoindanone unit is linked to electron-rich donors via a robust carbon-nitrogen bond formation. 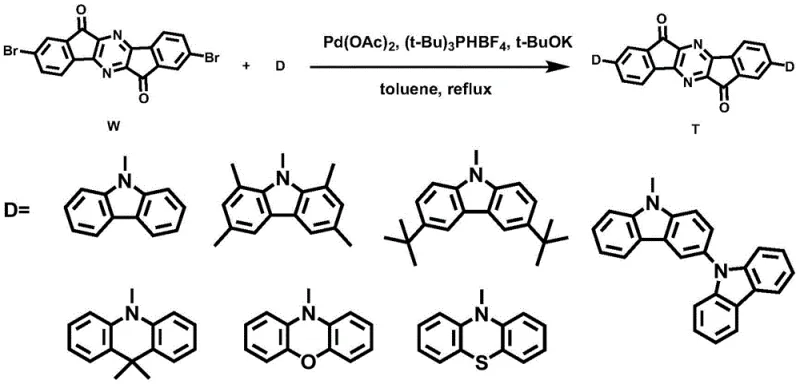 As illustrated in the reaction scheme, the final step involves a palladium-catalyzed Buchwald-Hartwig amination, a highly reliable method for constructing aryl amines. This approach allows for the precise tuning of the Highest Occupied Molecular Orbital (HOMO) and Lowest Unoccupied Molecular Orbital (LUMO) distribution. By spatially separating these orbitals onto the donor and acceptor moieties respectively, the exchange energy is minimized, facilitating efficient reverse intersystem crossing (RISC) from triplet to singlet states. This mechanism effectively harvests triplet excitons for fluorescence without the need for heavy metals, offering a pathway to 100% internal quantum efficiency with significantly reduced material costs.
As illustrated in the reaction scheme, the final step involves a palladium-catalyzed Buchwald-Hartwig amination, a highly reliable method for constructing aryl amines. This approach allows for the precise tuning of the Highest Occupied Molecular Orbital (HOMO) and Lowest Unoccupied Molecular Orbital (LUMO) distribution. By spatially separating these orbitals onto the donor and acceptor moieties respectively, the exchange energy is minimized, facilitating efficient reverse intersystem crossing (RISC) from triplet to singlet states. This mechanism effectively harvests triplet excitons for fluorescence without the need for heavy metals, offering a pathway to 100% internal quantum efficiency with significantly reduced material costs.
Mechanistic Insights into Buchwald-Hartwig Coupling and TADF Design
The chemical elegance of this system is rooted in the specific electronic properties of the pyrazinoindanone core and its interaction with diverse donor groups. 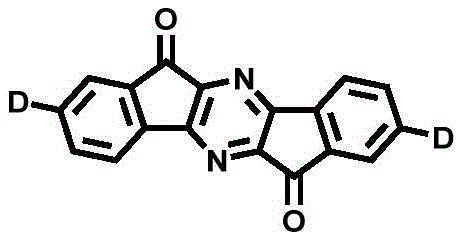 The general structure reveals a rigid, planar acceptor core that ensures good thermal stability and film-forming properties, which are essential for the vacuum deposition processes used in OLED manufacturing. The patent details the use of seven distinct donor groups, ranging from simple carbazole to more complex phenoxazine and phenothiazine derivatives. This structural diversity allows chemists to finely adjust the bandgap and emission color. For instance, the incorporation of bulky tert-butyl groups on the carbazole donor (as seen in derivative D3) not only modulates the electronic properties but also enhances solubility and suppresses aggregation-caused quenching in the solid state. The mechanistic pathway ensures that the HOMO is localized on the donor while the LUMO resides on the pyrazinoindanone acceptor, creating the necessary charge-transfer character for TADF activity.
The general structure reveals a rigid, planar acceptor core that ensures good thermal stability and film-forming properties, which are essential for the vacuum deposition processes used in OLED manufacturing. The patent details the use of seven distinct donor groups, ranging from simple carbazole to more complex phenoxazine and phenothiazine derivatives. This structural diversity allows chemists to finely adjust the bandgap and emission color. For instance, the incorporation of bulky tert-butyl groups on the carbazole donor (as seen in derivative D3) not only modulates the electronic properties but also enhances solubility and suppresses aggregation-caused quenching in the solid state. The mechanistic pathway ensures that the HOMO is localized on the donor while the LUMO resides on the pyrazinoindanone acceptor, creating the necessary charge-transfer character for TADF activity.
From a process chemistry perspective, the synthesis of the key intermediate, brominated pyrazinoindenone (Intermediate W), is a critical determinant of final product quality. The route begins with 5-bromo-1-indanone, undergoing a condensation with isoamyl nitrite followed by reduction and oxidation steps. This multi-step sequence requires precise control over reaction conditions, such as maintaining temperatures around 40°C for the initial condensation and utilizing strong oxidants like sodium dichromate for the final ring closure. The successful execution of these steps ensures the formation of the rigid conjugated system required for efficient electron transport. Any impurities generated during the formation of the pyrazinoindanone core could act as trap sites in the final OLED device, leading to reduced efficiency or shortened operational lifetime. Therefore, the rigorous purification protocols described, including column chromatography and recrystallization, are vital for achieving the high-purity standards demanded by the electronics industry.
How to Synthesize Pyrazinoindanone Derivatives Efficiently
The preparation of these high-performance electroluminescent materials follows a logical, step-wise progression that balances yield with purity. The process initiates with the functionalization of the indanone scaffold, proceeds through the construction of the heterocyclic core, and culminates in the coupling of the donor units. This standardized workflow allows for the systematic exploration of structure-property relationships by simply swapping the donor component while keeping the acceptor synthesis constant. For R&D teams looking to replicate or scale this chemistry, understanding the nuances of the catalytic cycle and workup procedures is paramount to success. The detailed standardized synthesis steps are provided in the guide below to facilitate immediate laboratory implementation.
- Synthesize intermediate N by reacting 5-bromo-1-indanone with isoamyl nitrite and concentrated hydrochloric acid in toluene at 40°C.
- Convert intermediate N to intermediate M using sodium hydrosulfite and ammonia solution in ethanol under nitrogen protection at 95°C.
- Oxidize intermediate M with sodium dichromate dihydrate in acetic acid/acetic anhydride to obtain the brominated pyrazinoindenone core (Intermediate W).
- Perform Buchwald-Hartwig coupling between Intermediate W and electron donors (e.g., carbazole) using Pd(OAc)2, t-BuOK, and (t-Bu)3PHBF4 in toluene at 110-140°C.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to pyrazinoindanone-based TADF materials represents a strategic opportunity to optimize cost structures and mitigate supply risks associated with precious metal dependency. The elimination of iridium and platinum from the emitter layer drastically reduces the raw material cost baseline, as these noble metals are subject to volatile market pricing and geopolitical supply constraints. Furthermore, the synthetic route utilizes commodity chemicals such as toluene, acetic acid, and commercially available carbazole derivatives, ensuring a stable and diversified supply base. The reliance on palladium catalysis, while requiring careful management, is a well-established industrial process with mature protocols for catalyst recovery and recycling, further enhancing the economic viability of the manufacturing process.
- Cost Reduction in Manufacturing: The primary economic driver for adopting this technology is the substantial reduction in material costs achieved by replacing rare-earth phosphors with organic small molecules. The synthesis avoids the complex ligand design and low-yield coordination chemistry often associated with phosphorescent emitters. Instead, it leverages high-yielding cross-coupling reactions that are amenable to large-scale batch processing. By removing the need for expensive heavy metal precursors, manufacturers can achieve significant margin improvements without sacrificing device performance. Additionally, the use of standard organic solvents and reagents simplifies waste management and reduces the regulatory burden associated with heavy metal disposal.
- Enhanced Supply Chain Reliability: Sourcing consistency is critical for display panel manufacturers who operate on tight production schedules. The starting material, 5-bromo-1-indanone, is readily accessible from multiple global suppliers, reducing the risk of single-source bottlenecks. The modular nature of the synthesis means that if a specific donor group faces supply issues, the platform technology allows for the rapid qualification of alternative donors with similar electronic properties. This flexibility ensures business continuity and allows supply chain heads to negotiate better terms with vendors due to the interchangeability of certain raw materials within the chemical class.
- Scalability and Environmental Compliance: The reaction conditions described, such as refluxing in toluene at 110-140°C, are fully compatible with existing stainless steel reactor infrastructure found in most fine chemical plants. There is no requirement for cryogenic conditions or ultra-high pressure equipment, which lowers the capital expenditure barrier for scale-up. From an environmental standpoint, the metal-free nature of the final product aligns with increasingly stringent global regulations regarding hazardous substances in electronics (such as RoHS). The ability to produce high-efficiency emitters without heavy metals simplifies the end-of-life recycling process for electronic devices, adding a layer of sustainability value that is increasingly important to downstream consumers and brand owners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these pyrazinoindanone derivatives. These insights are derived directly from the experimental data and performance metrics reported in the patent literature, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating these materials into their existing product lines.
Q: What is the primary advantage of pyrazinoindanone-based TADF materials?
A: These materials utilize a donor-acceptor architecture that separates HOMO and LUMO orbitals, enabling efficient reverse intersystem crossing (RISC) and achieving high internal quantum efficiency without expensive heavy metals.
Q: How does the synthesis ensure high purity for OLED applications?
A: The process utilizes robust purification steps including column chromatography and recrystallization after the final coupling reaction, ensuring the removal of palladium residues and unreacted precursors critical for device longevity.
Q: Is this manufacturing process scalable for industrial production?
A: Yes, the synthesis relies on standard organic transformations and commercially available reagents like 5-bromo-1-indanone, making it highly suitable for scale-up from kilogram to tonnage levels.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrazinoindanone Derivatives Supplier
As the global demand for high-resolution and energy-efficient displays continues to surge, the ability to source advanced OLED materials with consistent quality is a competitive differentiator. NINGBO INNO PHARMCHEM stands ready to support your development and production needs with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the precise temperature controls and inert atmosphere conditions required for the Buchwald-Hartwig coupling steps, ensuring that every batch meets stringent purity specifications. With rigorous QC labs employing HPLC, GC-MS, and NMR analysis, we guarantee the structural integrity and optical performance of every gram of material delivered, minimizing the risk of device failure in your final applications.
We invite you to collaborate with our technical team to explore how these cost-effective TADF materials can enhance your product portfolio. Whether you require custom synthesis of specific donor-acceptor combinations or large-scale supply of the core intermediates, we are prepared to provide a Customized Cost-Saving Analysis tailored to your volume requirements. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you accelerate your transition to next-generation electroluminescent technologies.
