Advanced Synthesis of Fluorinated Triarylamine Derivatives for High-Performance OLED Manufacturing
Advanced Synthesis of Fluorinated Triarylamine Derivatives for High-Performance OLED Manufacturing
The rapid evolution of the organic light-emitting diode (OLED) industry has created an urgent demand for high-performance blue light emitting materials that can match the efficiency and longevity of their red and green counterparts. Patent CN102115447B addresses this critical technological gap by disclosing a novel class of triarylamine derivatives containing fluorine substituents, specifically designed to function as superior blue light organic electroluminescent materials. These compounds are structurally engineered to overcome the inherent charge transport imbalances found in traditional hole-transport materials, offering a pathway to more efficient and stable full-color displays. The core innovation lies in the strategic substitution of hydrogen atoms with fluorine on the biphenyl backbone, which modulates the electronic properties without compromising the structural integrity required for thin-film formation. As a reliable OLED material supplier, understanding the nuances of this patented chemistry is essential for developing next-generation display technologies that meet the rigorous standards of modern consumer electronics.
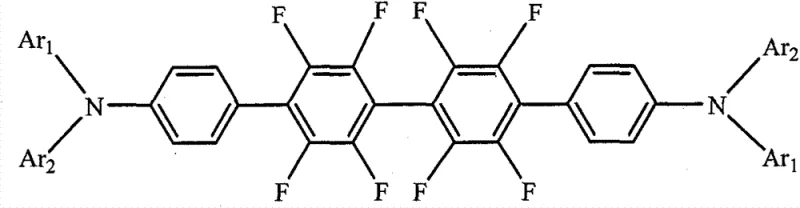
The general structure of these derivatives, as illustrated in the patent documentation, features a central octafluorobiphenyl core linked to triarylamine units, where the aryl groups can be phenyl or naphthyl, optionally substituted with methyl groups. This modular design allows for fine-tuning of the material's photophysical properties, such as emission wavelength and quantum yield, simply by varying the Ar1 and Ar2 substituents. The versatility of this molecular architecture means that manufacturers can customize the material profile to fit specific device architectures, whether for mobile displays, large-screen televisions, or lighting applications. Furthermore, the presence of multiple fluorine atoms significantly enhances the chemical and thermal stability of the molecule, a critical factor for ensuring the long operational lifetime of OLED panels. By leveraging this patented technology, producers can achieve a level of performance consistency that is often difficult to attain with non-fluorinated analogues, thereby reducing waste and improving overall manufacturing yields.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of blue OLED materials has been hindered by the difficulty in achieving balanced charge injection and transport within the emissive layer. Conventional triarylamine compounds, while excellent hole transporters, typically exhibit hole mobilities that are two orders of magnitude higher than their electron mobilities. This severe imbalance leads to the accumulation of positive charges within the device, causing efficiency roll-off at high brightness levels and accelerating device degradation over time. Additionally, many traditional blue emitters suffer from poor thermal stability, with decomposition temperatures that may not withstand the thermal stresses encountered during vacuum deposition processes. The lack of robust synthetic routes that can introduce electron-withdrawing groups without complex multi-step sequences has further limited the commercial viability of high-performance blue materials. Consequently, the industry has faced significant challenges in cost reduction in electronic chemical manufacturing, as inefficient materials require thicker layers or additional compensatory layers to function correctly, driving up material consumption and processing time.
The Novel Approach
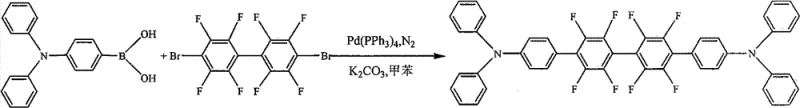
The methodology presented in CN102115447B offers a transformative solution by integrating electron-withdrawing fluorine substituents directly into the triarylamine framework through a streamlined Suzuki-Miyaura coupling reaction. This approach effectively lowers the hole mobility while simultaneously enhancing electron mobility, resulting in a much more balanced charge transport rate that is ideal for high-efficiency light emission. The use of 4,4'-dibromooctafluorobiphenyl as a key building block allows for the direct introduction of eight fluorine atoms in a single coupling step, drastically simplifying the synthetic complexity compared to post-functionalization methods. Moreover, the resulting derivatives demonstrate exceptional solubility in common organic solvents like toluene and chloroform, reaching concentrations of approximately 5% by mass, which facilitates solution-processing techniques such as inkjet printing. This dual improvement in electronic balance and processability represents a significant leap forward, enabling the production of high-purity OLED material with superior device performance characteristics and simplified fabrication protocols.
Mechanistic Insights into Suzuki-Miyaura Coupling for Fluorinated Systems
The synthesis of these advanced materials relies on the robust mechanism of the palladium-catalyzed cross-coupling reaction, specifically utilizing tetrakis(triphenylphosphine)palladium(0) as the catalyst. The reaction initiates with the oxidative addition of the palladium center into the carbon-bromine bonds of the 4,4'-dibromooctafluorobiphenyl, a step that is facilitated by the electron-deficient nature of the fluorinated ring system. Following this, the triarylaminoboronic acid undergoes transmetallation with the palladium complex, a process that is accelerated by the presence of a base such as potassium carbonate or sodium carbonate in the aqueous phase. The final reductive elimination step releases the coupled product and regenerates the active palladium(0) species, allowing the catalytic cycle to continue efficiently. The choice of solvent, typically toluene or xylene, plays a crucial role in solubilizing both the organic reactants and the catalyst, ensuring homogeneous reaction conditions that maximize yield and minimize side reactions. Understanding this mechanistic pathway is vital for optimizing reaction parameters such as temperature and time to ensure consistent quality across different batches.
Beyond the primary coupling mechanism, the presence of fluorine atoms exerts a profound influence on the electronic distribution within the final molecule, which is key to its function as an electroluminescent material. The strong electronegativity of fluorine pulls electron density away from the nitrogen centers and the conjugated pi-system, effectively lowering the energy levels of both the Highest Occupied Molecular Orbital (HOMO) and the Lowest Unoccupied Molecular Orbital (LUMO). This adjustment is critical for aligning the energy levels of the emissive material with those of the adjacent charge transport layers in an OLED device, thereby reducing the energy barrier for charge injection. Furthermore, the C-F bonds formed are among the strongest in organic chemistry, imparting remarkable resistance to oxidative degradation and thermal breakdown. This intrinsic stability ensures that the material maintains its structural integrity and optical properties even under the high current densities required for bright display applications. For R&D teams, controlling the purity of the boronic acid precursor and the stoichiometry of the base is essential to prevent the formation of homocoupling byproducts, which could act as quenching sites and reduce the overall luminous efficiency of the device.
How to Synthesize N,N,N',N'-tetraphenyl-octafluoro-p-quaterphenyl-diamine Efficiently
The practical implementation of this synthesis involves a straightforward procedure that can be adapted for both laboratory-scale optimization and larger production runs. The process begins by charging a reaction vessel with the dibromo-fluorinated core and the appropriate triarylamine boronic acid derivative in a dry organic solvent under an inert atmosphere. Aqueous base is then added to facilitate the activation of the boronic acid, followed by the introduction of the palladium catalyst to initiate the coupling. The mixture is heated to temperatures ranging from 60°C to 150°C, typically around 100°C for toluene systems, and stirred for a period of 12 to 72 hours depending on the specific reactivity of the substrates. Upon completion, the reaction mixture is cooled and subjected to a workup procedure involving washing with water to remove inorganic salts and catalyst residues, followed by extraction of the organic phase. The crude product is then purified using standard techniques such as silica gel column chromatography or recrystallization to achieve the high purity levels required for electronic applications. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety.
- Prepare the reaction mixture by combining 4,4'-dibromooctafluorobiphenyl and triarylaminoboronic acid in an organic solvent like toluene.
- Add the palladium catalyst Pd(PPh3)4 and an aqueous base solution such as K2CO3 under an inert nitrogen atmosphere.
- Heat the mixture to 60-150°C for 12-72 hours, then cool, extract with water, and purify the product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this fluorinated triarylamine synthesis route offers substantial strategic benefits that extend beyond mere technical performance. The reliance on widely available commodity chemicals such as toluene, potassium carbonate, and standard palladium catalysts means that the supply chain is less vulnerable to the bottlenecks often associated with exotic or proprietary reagents. This accessibility translates directly into enhanced supply chain reliability, as manufacturers can source raw materials from multiple vendors without compromising on the quality or consistency of the final product. Furthermore, the simplicity of the one-pot reaction design eliminates the need for complex intermediate isolation steps, which significantly reduces the overall processing time and labor costs associated with production. By streamlining the manufacturing workflow, companies can achieve significant cost savings in terms of both operational expenditure and capital investment in specialized equipment. The robustness of the reaction conditions also implies a lower risk of batch failure, ensuring a steady and predictable output of high-value electronic chemicals.
- Cost Reduction in Manufacturing: The elimination of complex multi-step functionalization sequences and the use of cost-effective catalysts significantly lower the bill of materials. By avoiding the need for expensive protecting group strategies or harsh reaction conditions that require specialized containment, the overall production cost is drastically reduced. This economic efficiency allows for more competitive pricing structures in the highly contested OLED materials market, enabling downstream device manufacturers to improve their margins. Additionally, the high solubility of the product facilitates easier purification, reducing solvent consumption and waste disposal costs associated with extensive chromatographic separations. These factors combined create a compelling economic case for adopting this technology over legacy methods that are more resource-intensive.
- Enhanced Supply Chain Reliability: The use of stable and shelf-stable reagents like 4,4'-dibromooctafluorobiphenyl ensures that inventory management is straightforward and less prone to degradation issues. Since the reaction tolerates a range of temperatures and solvent systems, production scheduling becomes more flexible, allowing manufacturers to respond quickly to fluctuations in market demand. The ability to scale the reaction from gram to kilogram quantities without fundamental changes to the process chemistry further strengthens the supply chain resilience. This scalability ensures that reducing lead time for high-purity display materials is achievable, as production capacity can be ramped up rapidly to meet the needs of new product launches or unexpected surges in orders.
- Scalability and Environmental Compliance: The reaction operates under relatively mild conditions compared to other cross-coupling methodologies, which reduces energy consumption and the carbon footprint of the manufacturing process. The aqueous workup generates waste streams that are easier to treat and manage, aligning with increasingly stringent environmental regulations governing chemical production. The high atom economy of the Suzuki coupling, particularly with the efficient incorporation of the fluorinated core, minimizes the generation of hazardous byproducts. This alignment with green chemistry principles not only mitigates regulatory risks but also enhances the corporate sustainability profile of the manufacturer, which is becoming a key differentiator in global supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these fluorinated triarylamine derivatives. These answers are derived directly from the experimental data and technical specifications outlined in the patent literature to provide accurate guidance for potential partners. Understanding these details is crucial for evaluating the feasibility of integrating these materials into existing production lines or new product developments. The information covers aspects ranging from material properties to process scalability, ensuring a comprehensive overview for decision-makers.
Q: What is the primary advantage of introducing fluorine substituents in triarylamine derivatives?
A: Introducing electron-withdrawing fluorine atoms balances the hole and electron mobility, which is typically skewed in standard triarylamines, thereby significantly improving the luminous efficiency of the resulting OLED devices.
Q: What are the thermal stability characteristics of these fluorinated derivatives?
A: The synthesized derivatives exhibit excellent thermal properties with an initial thermal decomposition temperature greater than 300°C, ensuring robust performance during device fabrication and operation.
Q: Is the synthesis process scalable for industrial production?
A: Yes, the method utilizes standard Suzuki coupling conditions with commercially available reagents and solvents like toluene, making it highly suitable for commercial scale-up of complex organic intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Triarylamine Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance organic intermediates play in the advancement of the optoelectronics industry. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous volume requirements of global display manufacturers. We are committed to delivering materials with stringent purity specifications, utilizing our rigorous QC labs to verify every batch against the highest industry standards. Our expertise in handling fluorinated compounds and palladium-catalyzed reactions allows us to optimize the synthesis of these triarylamine derivatives for maximum yield and minimal impurity profiles. By partnering with us, you gain access to a supply chain that is both robust and responsive, capable of supporting your long-term technology roadmaps.
We invite you to contact our technical procurement team to discuss how we can support your specific material needs with a Customized Cost-Saving Analysis. Whether you require specific COA data for qualification purposes or detailed route feasibility assessments for new derivative structures, we are equipped to provide the technical depth and commercial flexibility you require. Let us help you accelerate your development timeline and secure a competitive advantage in the rapidly evolving OLED market through our dedicated partnership and manufacturing excellence.
