Scalable Manufacturing of 4-Pentafluorosulfanylbenzoylguanidine Derivatives for Cardiovascular Drug Development
Scalable Manufacturing of 4-Pentafluorosulfanylbenzoylguanidine Derivatives for Cardiovascular Drug Development
The pharmaceutical industry's relentless pursuit of novel cardiovascular therapeutics has placed significant emphasis on the development of Na+/H+ Exchanger isoform 1 (NHE1) inhibitors. A pivotal class of compounds in this domain involves 4-pentafluorosulfanylbenzoylguanidines, which exhibit potent biological activity due to the unique lipophilic and metabolic stability properties conferred by the pentafluorosulfanyl (SF5) group. Patent CN101475519B discloses a highly efficient and robust synthetic methodology for preparing these critical intermediates, addressing longstanding challenges in yield and scalability. This technical insight report analyzes the patented process, highlighting its strategic value for R&D teams seeking reliable pathways and procurement officers aiming for cost-effective supply chains. The core innovation lies in the utilization of commercially available 4-nitrophenylsulfur pentafluoride as a starting material, bypassing the need for complex de novo construction of the fluorinated aromatic core.
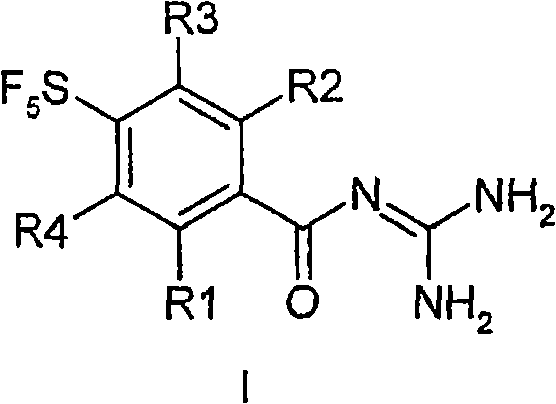
The structural versatility of Formula I allows for extensive SAR (Structure-Activity Relationship) exploration, with variable substituents at the R1 and R2 positions enabling fine-tuning of pharmacokinetic profiles. For global pharmaceutical manufacturers, securing a stable supply of such high-purity intermediates is paramount for advancing clinical candidates through the pipeline. The disclosed method not only optimizes the chemical transformations but also integrates purification strategies that are amenable to industrial scale-up, ensuring that the final active pharmaceutical ingredient (API) precursors meet stringent regulatory standards. By leveraging this technology, stakeholders can mitigate the risks associated with supply chain disruptions often caused by bespoke synthetic routes requiring exotic reagents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art, specifically referenced in German application 10222192, described the preparation of pentafluorothiobenzoylguanidines but was plagued by significant technical drawbacks that hindered commercial viability. The conventional methods often relied on reaction conditions that were excessively harsh or required reagents that introduced substantial technical complexity, leading to inconsistent batch-to-batch quality. Furthermore, the reported yields in these legacy processes were frequently low, necessitating large quantities of starting materials to produce modest amounts of the desired intermediate, which drastically inflated the cost of goods sold (COGS). The purification steps associated with these older routes often involved difficult separations of closely related impurities, requiring extensive chromatography that is impractical for multi-kilogram or ton-scale manufacturing. Consequently, the reliance on such inefficient methodologies created bottlenecks in the supply chain, delaying project timelines and increasing the financial burden on drug development programs.
The Novel Approach
In stark contrast, the methodology outlined in CN101475519B introduces a streamlined synthetic strategy that begins with the reduction of readily accessible 4-nitrophenylsulfur pentafluoride derivatives. This novel approach circumvents the need for constructing the SF5-substituted benzene ring from scratch, instead functionalizing an existing, commercially sourced scaffold. The process employs a logical sequence of transformations including ortho-halogenation, transition metal-catalyzed cross-coupling, and diazotization-cyanation, each step optimized for high conversion and ease of workup. By selecting reagents and conditions that favor crystallization over chromatography for purification, the new method significantly enhances operational simplicity. This shift from complex, low-yielding protocols to a modular, high-efficiency route represents a paradigm shift in the manufacturing of NHE1 inhibitor intermediates, offering a clear path toward cost reduction and supply security for downstream API production.
Mechanistic Insights into the Multi-Step Synthetic Pathway
The chemical elegance of this process is best understood through a detailed mechanistic dissection of the key transformation steps, particularly the installation of the R2 substituent and the conversion of the amine to the acylguanidine moiety. The synthesis initiates with the reduction of the nitro group to an amine, typically using tin(II) chloride in concentrated hydrochloric acid, a robust and scalable reduction method. Following this, the amine undergoes ortho-halogenation, where the amino group directs the electrophilic substitution to the adjacent position, setting the stage for subsequent functionalization. The introduction of diverse R2 groups is achieved via palladium-catalyzed cross-coupling reactions, such as the Suzuki-Miyaura coupling, utilizing organoboron reagents like trimethylboroxine. This catalytic cycle involves oxidative addition of the aryl halide to the Pd(0) species, transmetallation with the boronate, and reductive elimination to forge the new carbon-carbon bond, allowing for the precise installation of alkyl or aryl groups with high fidelity.
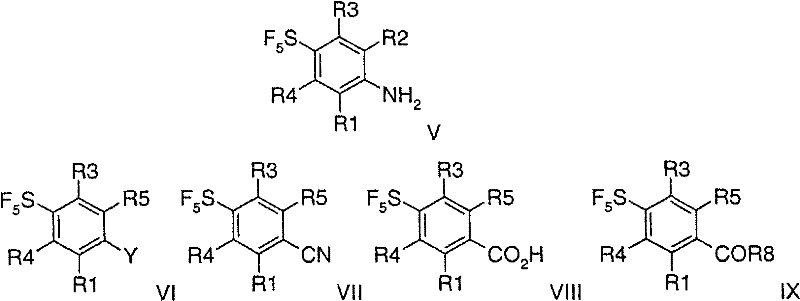
Following the establishment of the carbon skeleton, the amino functionality is strategically replaced to facilitate the formation of the carboxylic acid precursor. This is accomplished through a diazotization-halogenation sequence, effectively converting the amine into a halide (Formula VI), which is subsequently subjected to Rosenmund-von Braun type cyanation using zinc cyanide and a palladium catalyst. The resulting nitrile (Formula VII) is then hydrolyzed under basic conditions to yield the corresponding benzoic acid (Formula VIII). This sequence is critical as it allows for the orthogonal manipulation of the aromatic ring, ensuring that the sensitive SF5 group remains intact throughout the harsh acidic and basic conditions employed. Finally, the carboxylic acid is activated, typically via conversion to an acid chloride using thionyl chloride, and reacted with guanidine to form the target acylguanidine. The rigorous control of reaction parameters, such as temperature and stoichiometry, ensures minimal formation of side products, thereby simplifying the impurity profile and enhancing the overall purity of the final intermediate.
How to Synthesize 2-Methyl-4-Pentafluorothiobenzoylguanidine Efficiently
The practical implementation of this synthetic route requires careful attention to reaction conditions and safety protocols, particularly when handling reactive halogenating agents and cyanide sources. The patent provides explicit guidance on solvent selection, recommending aprotic polar solvents like DMF or DMAc for the cyanation step to ensure solubility and reaction kinetics, while non-protic solvents like dichloromethane are preferred for the initial halogenation to control exotherms. The detailed standardized synthesis steps provided below outline the specific molar ratios, temperature ranges, and workup procedures necessary to replicate the high yields reported in the experimental examples. Adhering to these protocols allows manufacturing teams to achieve consistent results, minimizing the risk of batch failures and ensuring that the produced intermediates meet the strict quality specifications required for pharmaceutical applications.
- Reduce 4-nitrophenylsulfur pentafluoride to the corresponding amine using tin(II) chloride in acidic conditions.
- Perform ortho-halogenation followed by palladium-catalyzed cross-coupling to introduce the R2 substituent.
- Convert the amine to a nitrile via diazotization-halogenation and subsequent cyanation, then hydrolyze to the carboxylic acid.
- Activate the carboxylic acid to an acid chloride and react with guanidine to form the final acylguanidine product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis offers tangible strategic benefits that extend beyond mere chemical efficiency. The primary advantage lies in the utilization of commercially available starting materials, specifically 4-nitrophenylsulfur pentafluoride, which eliminates the dependency on custom-synthesized building blocks that often carry long lead times and high price volatility. By anchoring the supply chain on commodity chemicals, organizations can significantly reduce the risk of production stoppages caused by raw material shortages. Furthermore, the process design inherently supports cost reduction in pharmaceutical intermediate manufacturing by minimizing the number of purification steps; the reliance on crystallization and distillation rather than preparative chromatography lowers solvent consumption and waste disposal costs, contributing to a more sustainable and economically viable production model.
- Cost Reduction in Manufacturing: The elimination of complex, low-yielding steps found in prior art directly translates to lower operational expenditures. By avoiding the need for exotic reagents and reducing the consumption of precious metal catalysts through optimized loading, the overall cost per kilogram of the intermediate is substantially decreased. Additionally, the ability to telescope certain steps or perform them without intermediate isolation reduces labor hours and equipment occupancy time, further driving down the manufacturing overhead. This economic efficiency allows for more competitive pricing strategies when sourcing these critical building blocks for large-scale API production.
- Enhanced Supply Chain Reliability: The robustness of the synthetic route ensures high supply continuity, a critical factor for maintaining uninterrupted drug production schedules. The use of standard industrial solvents and reagents means that sourcing is not limited to niche suppliers, providing procurement teams with multiple vendor options and greater negotiating leverage. Moreover, the scalability of the process, demonstrated from gram to multi-hundred gram scales in the patent data, assures supply chain heads that the technology can be seamlessly transferred to pilot plants and commercial manufacturing facilities without significant re-engineering, thereby securing long-term supply stability.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, the process is designed to minimize waste generation and hazardous emissions. The efficient atom economy of the cross-coupling and cyanation steps, combined with effective recovery of solvents, aligns with modern green chemistry principles. This compliance reduces the burden of environmental permitting and waste treatment costs, facilitating smoother regulatory approvals for manufacturing sites. The ability to scale this process to 100 MT annual production capacities without compromising safety or quality makes it an ideal candidate for integration into existing fine chemical manufacturing infrastructures.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and supply of 4-pentafluorosulfanylbenzoylguanidine derivatives. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these nuances is essential for making informed decisions regarding technology transfer and vendor selection.
Q: What is the primary starting material for this synthesis?
A: The process utilizes commercially available 4-nitrophenylsulfur pentafluoride as the key starting block, which significantly simplifies the supply chain compared to custom synthesis of the SF5 core.
Q: How does this method improve upon previous German patent applications?
A: Unlike prior art which suffered from low yields and technical complexity, this method employs robust catalytic steps like Suzuki coupling and standard diazotization, allowing for easier purification and higher overall throughput.
Q: Is the process suitable for large-scale production?
A: Yes, the patent explicitly demonstrates scalability through examples ranging from gram to multi-hundred gram scales, utilizing standard industrial solvents like dichloromethane and toluene.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Pentafluorosulfanylbenzoylguanidine Supplier
As the global demand for cardiovascular therapeutics continues to rise, the need for high-quality, reliably sourced intermediates has never been more critical. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging advanced synthetic technologies like those described in CN101475519B to deliver superior chemical solutions. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volumetric requirements of even the largest pharmaceutical programs. With our stringent purity specifications and rigorous QC labs, we guarantee that every batch of 4-pentafluorosulfanylbenzoylguanidine delivered meets the exacting standards required for clinical and commercial API synthesis, providing our partners with peace of mind and regulatory confidence.
We invite forward-thinking R&D and procurement leaders to collaborate with us to optimize their supply chains and accelerate their drug development timelines. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume needs and project milestones. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing you to evaluate how our manufacturing capabilities can support your next breakthrough in cardiovascular medicine. Let us be your trusted partner in transforming complex chemical challenges into commercial successes.
