Advanced Radical Cyclization for High-Purity Benzimidazole Isoquinoline Ketones and Commercial Scalability
The pharmaceutical and fine chemical industries are constantly seeking efficient pathways to construct complex polycyclic scaffolds that serve as critical backbones for bioactive molecules. A significant breakthrough in this domain is detailed in patent CN114105981B, which discloses a novel method for preparing benzimidazol[2,1-a]isoquinolin-6(5H)-one compounds. These heterocyclic structures are ubiquitous drug structural motifs known for their potent applications in treating anti-diabetes, anti-tumor diseases, and inflammatory conditions. The disclosed technology leverages an efficient radical cyclization strategy of 2-arylbenzimidazoles, specifically utilizing N-methacryloyl-2-phenylbenzimidazoles as radical acceptors. This approach represents a paradigm shift from traditional methods by enabling the direct introduction of cyanoalkyl, ketone, or nitro substituents under mild thermal conditions, thereby offering a reliable pharma intermediate supplier with a robust technological foundation for producing high-value medicinal intermediates.
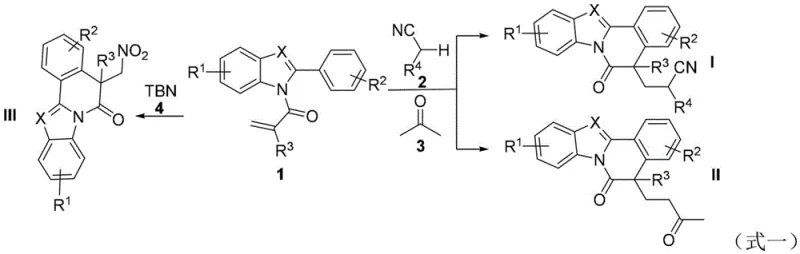
The development of this synthetic route addresses critical limitations found in conventional methodologies. Historically, the construction of benzimidazol[2,1-a]isoquinolin-6(5H)-one skeletons via radical cyclization has relied heavily on C-, S-, P-, Si-, and Ge-centered radicals. However, the functionalization using N-centered radicals or the direct C(sp3)-H functionalization of nitriles and ketones remained largely unexplored or inefficient. Prior art, such as reports in J. Org. Chem. 2020 and RSC Adv. 2021, necessitated the use of brominated raw materials as radical precursors and required expensive photocatalytic redox systems to proceed. These legacy methods impose substantial burdens on cost reduction in pharmaceutical intermediate manufacturing due to the high price of halogenated precursors and the capital expenditure associated with photoreactors. Furthermore, the reliance on noble metal photocatalysts often introduces challenges in removing trace metal impurities, which is a critical quality attribute for API intermediates.
In stark contrast, the novel approach described in the patent utilizes a thermally induced radical generation system that bypasses the need for light irradiation and halogenated starting materials. By employing simple and abundant radical sources such as nitriles, acetone, or tert-butyl nitrite, the method achieves direct C-H functionalization. The reaction proceeds under an air atmosphere, eliminating the strict requirement for inert gas protection which simplifies operational complexity. The versatility of this system allows for the synthesis of three distinct classes of products: cyanoalkyl-substituted derivatives (Type I), ketone skeleton-containing compounds (Type II), and nitro-substituted analogs (Type III). This flexibility ensures that the commercial scale-up of complex pharmaceutical intermediates can be achieved with a single platform technology, adapting easily to diverse molecular requirements without changing the core reactor setup.
Mechanistic Insights into Transition Metal-Catalyzed Radical Cyclization
The mechanistic pathway of this transformation involves a sophisticated interplay between transition metal catalysts and oxidants to generate reactive radical species. For the synthesis of cyanoalkyl-substituted products, the system likely employs a Nickel(II) catalyst which interacts with the oxidant, such as benzoyl peroxide, to generate carbon-centered radicals from the alpha-position of the nitrile. Similarly, for ketone skeleton formation, an Iron(III) catalyst facilitates the homolytic cleavage of the oxidant to abstract a hydrogen atom from acetone, generating an alpha-acetonyl radical. In the case of nitro-substitution, a Copper(II) catalyst assists in the decomposition of tert-butyl nitrite to produce nitrogen-centered radicals. These generated radicals then add to the electron-deficient alkene moiety of the N-methacryloyl group, initiating an intramolecular cyclization onto the pendant aryl ring.
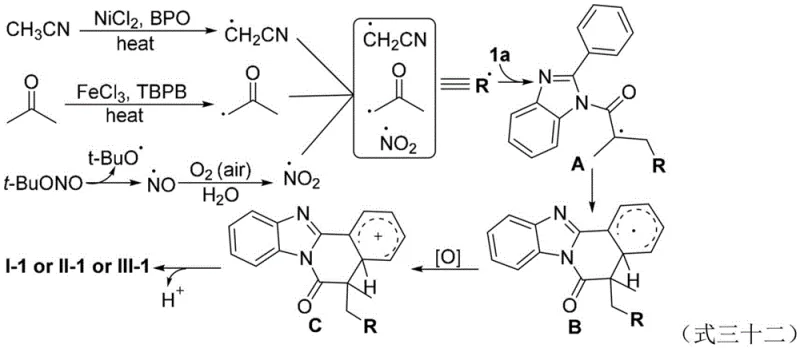
Following the initial radical addition, the resulting intermediate undergoes oxidative aromatization to restore the aromaticity of the fused ring system, yielding the final benzimidazol[2,1-a]isoquinolin-6(5H)-one structure. The control experiments utilizing radical scavengers like BHT (butylated hydroxytoluene) confirmed the radical nature of the process, as the reaction was significantly inhibited in the presence of the scavenger, and adducts between BHT and the proposed radical intermediates were detected. This mechanistic understanding is crucial for ensuring high-purity pharmaceutical intermediates, as it allows for the precise tuning of oxidant equivalents and catalyst loading to minimize side reactions. The ability to operate under air atmosphere suggests that oxygen may play a role in the re-oxidation of the metal catalyst or the final oxidative step, further streamlining the process by removing the need for stoichiometric external oxidants in some contexts.
How to Synthesize Benzimidazol[2,1-a]isoquinolin-6(5H)-ones Efficiently
The synthesis protocol outlined in the patent provides a standardized procedure for accessing these valuable heterocycles with high reproducibility. The process is designed to be operationally simple, requiring standard laboratory glassware such as Schlenk bottles and basic heating equipment. The key to success lies in the selection of the appropriate catalyst-oxidant pair matched to the specific radical source being employed. For instance, when targeting cyanoalkyl derivatives, the use of NiCl2 and benzoyl peroxide at 120°C yields optimal results, whereas ketone derivatives require FeCl3 and tert-butyl peroxybenzoate at 90°C. The detailed standardized synthesis steps see the guide below ensure that research and development teams can replicate the high yields reported in the examples, facilitating rapid process development.
- Combine N-methacryloyl-2-phenylbenzimidazole substrate with a radical source (nitrile, acetone, or tert-butyl nitrite), a transition metal catalyst (Ni, Fe, or Cu), and an oxidant in a Schlenk bottle.
- Heat the reaction mixture under an air atmosphere at optimized temperatures ranging from 80°C to 120°C for 10 to 12 hours to facilitate radical cyclization.
- Upon completion, extract the reaction mixture with ethyl acetate, dry the organic phase, concentrate under reduced pressure, and purify the residue via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this technology offers transformative benefits that directly impact the bottom line and operational resilience. The elimination of expensive photocatalytic systems and halogenated precursors translates into significant cost optimization in fine chemical manufacturing. By utilizing commodity chemicals like acetonitrile and acetone which serve dual roles as both reagents and solvents, the material costs are drastically reduced compared to specialized radical precursors. Furthermore, the absence of noble metal photocatalysts removes the necessity for costly metal scavenging steps during downstream processing, thereby simplifying the purification workflow and reducing waste generation. This streamlined approach enhances the overall economic viability of producing these complex intermediates at scale.
- Cost Reduction in Manufacturing: The process utilizes earth-abundant transition metal catalysts such as Nickel, Iron, and Copper instead of precious metals like Iridium or Ruthenium often found in photocatalysis. This substitution leads to substantial cost savings in raw material procurement. Additionally, the reaction conditions are mild and do not require energy-intensive cooling or specialized light sources, resulting in lower utility costs. The simplified workup procedure, involving standard extraction and chromatography, reduces labor and solvent consumption, contributing to a more lean and cost-effective manufacturing process.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, including N-methacryloyl-2-phenylbenzimidazoles, nitriles, acetone, and tert-butyl nitrite, are commercially available in bulk quantities from multiple global suppliers. This diversity in sourcing mitigates the risk of supply chain disruptions associated with single-source specialty reagents. The robustness of the reaction under air atmosphere also means that production is less susceptible to failures in inert gas supply systems, ensuring consistent batch-to-batch reliability and reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The methodology is inherently scalable, having been demonstrated in Schlenk bottles which can be directly translated to larger stirred-tank reactors. The use of air as an oxidant component and the avoidance of toxic halogenated waste streams align with green chemistry principles. This environmental compatibility simplifies regulatory compliance and waste disposal management, making it easier to obtain necessary permits for commercial production. The high atom economy of incorporating simple alkyl groups directly into the scaffold minimizes waste generation, supporting sustainable manufacturing goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this radical cyclization technology. These answers are derived directly from the experimental data and mechanistic studies presented in the patent documentation. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing production pipelines. The clarity provided here aims to reduce uncertainty and accelerate the decision-making process for adopting this innovative synthetic method.
Q: What are the optimal catalyst systems for different radical sources in this synthesis?
A: The patent specifies distinct catalyst systems for optimal yield: Nickel chloride (NiCl2) with benzoyl peroxide is preferred for nitrile sources; Ferric chloride (FeCl3) with tert-butyl peroxybenzoate is optimal for acetone; and Copper acetate (Cu(OAc)2) with TEMPO is best for tert-butyl nitrite reactions.
Q: How does this method improve upon existing photocatalytic routes?
A: Unlike prior art requiring expensive photocatalytic redox systems and brominated precursors, this method utilizes thermally induced radical generation under air atmosphere. This eliminates the need for specialized light equipment and costly halogenated starting materials, significantly simplifying the process infrastructure.
Q: Is this radical cyclization process suitable for large-scale manufacturing?
A: Yes, the process is highly scalable as it operates under standard thermal conditions (80-120°C) in an air atmosphere without stringent inert gas requirements. The use of commercially available solvents like acetonitrile or acetone as both reagents and solvents further enhances its viability for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzimidazol[2,1-a]isoquinolin-6(5H)-one Supplier
NINGBO INNO PHARMCHEM stands at the forefront of implementing advanced synthetic methodologies like the one described in CN114105981B to deliver superior chemical solutions. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from laboratory discovery to industrial reality. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify the identity and purity of every batch. We understand that consistency is key in the pharmaceutical supply chain, and our processes are designed to meet the exacting standards required for clinical and commercial stage materials.
We invite you to collaborate with us to leverage this cutting-edge radical cyclization technology for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific molecular targets, demonstrating how this route can optimize your budget without compromising quality. Please contact our technical procurement team to request specific COA data and route feasibility assessments. By partnering with NINGBO INNO PHARMCHEM, you gain access to a reliable supply chain partner dedicated to driving innovation and efficiency in the production of complex pharmaceutical intermediates.
