Advanced Visible-Light Synthesis of 1-Iodo-3-Perfluoroalkyl Alkenes for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to introduce fluorine-containing motifs into complex molecular architectures, driven by the unique physicochemical properties these groups impart. Patent CN112047839B discloses a groundbreaking preparation method for 1-iodo-3-perfluoroalkyl alkene compounds, a class of highly reactive intermediates essential for modern drug discovery and material science. These compounds, characterized by the general structure shown in Formula (I), serve as versatile building blocks capable of participating in diverse coupling reactions such as Kumada, Negishi, and Sonogashira couplings, as well as radical cycloadditions. The innovation lies not just in the molecule itself, but in the sustainable and efficient pathway to access it, addressing long-standing challenges in fluoroalkylation chemistry.
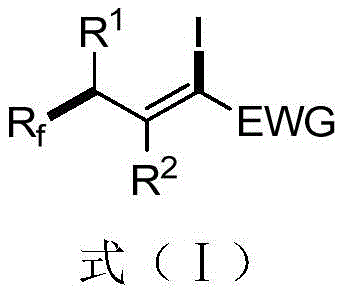
Traditionally, the synthesis of alkenyl iodides and perfluoroalkylated olefins has been plagued by significant operational hurdles that hinder efficient manufacturing. Conventional methods often rely on the use of expensive transition metal photocatalysts like Ruthenium or Iridium complexes, or require harsh thermal conditions that can degrade sensitive functional groups. Furthermore, existing techniques, such as those disclosed in prior art like CN106832256A, frequently necessitate the introduction of fluorine-containing nonionic surfactants and multiple additions of initiators. These additives complicate the reaction matrix, making product isolation tedious and generating substantial chemical waste. The reliance on such complex additive systems limits the substrate scope and increases the overall cost of goods, creating a bottleneck for procurement teams aiming to source high-purity intermediates reliably.
In stark contrast, the novel approach detailed in this patent leverages a visible-light promoted strategy that drastically simplifies the synthetic landscape. By utilizing an Atom Transfer Radical Addition (ATRA) reaction between an alkenyl diazo compound and a perfluoroalkyl iodide, the method achieves high conversion under mild irradiation from common light sources like CFL or LEDs. Crucially, this is followed by a manganese-catalyzed isomerization step that ensures high stereoselectivity. This dual-step process eliminates the need for exotic photocatalysts or surfactants, relying instead on inexpensive manganese carbonyl complexes. The result is a green synthesis concept where nitrogen is the sole byproduct, aligning perfectly with modern environmental compliance standards and offering a streamlined route for the commercial scale-up of complex fluorine-containing intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of perfluoroalkyl groups into organic molecules has required rigorous exclusion of air and moisture, alongside the use of stoichiometric amounts of hazardous reagents. Traditional thermal radical initiators often lack selectivity, leading to broad distributions of byproducts that are difficult to separate, thereby reducing the overall yield and purity of the final API intermediate. The necessity for specialized surfactants in aqueous or biphasic systems further complicates the workup procedure, requiring extensive extraction and purification steps that increase solvent consumption and processing time. For supply chain managers, these inefficiencies translate into longer lead times and higher vulnerability to raw material price fluctuations, as the process becomes dependent on a wider array of specialized chemicals that may face supply constraints.
The Novel Approach
The methodology presented in CN112047839B represents a paradigm shift towards operational simplicity and efficiency. By employing visible light as the energy source, the reaction proceeds at temperatures ranging from 0°C to 80°C, significantly reducing energy consumption compared to high-temperature thermal processes. The use of a manganese catalyst for the subsequent isomerization step is particularly advantageous; manganese is abundant and cost-effective compared to noble metals. This approach demonstrates exceptional substrate adaptability, accommodating various perfluoroalkyl chains from trifluoromethyl to longer perfluorodecyl groups, as well as diverse electron-withdrawing groups. The ability to achieve Z/E selectivity ratios greater than 30:1 directly through catalysis minimizes the need for resource-intensive chromatographic purification, directly impacting the bottom line through cost reduction in pharma intermediate manufacturing.
Mechanistic Insights into Visible-Light Promoted ATRA and Mn-Catalyzed Isomerization
The core of this synthetic breakthrough involves a sophisticated interplay between radical chemistry and transition metal catalysis. Initially, the perfluoroalkyl iodide interacts with the alkenyl diazo compound under visible light irradiation. This interaction can proceed via the formation of an Electron Donor-Acceptor (EDA) complex or through direct homolytic cleavage facilitated by the light source, generating a perfluoroalkyl radical. This radical adds across the double bond of the diazo compound, followed by iodine atom transfer, resulting in the formation of the 1-iodo-3-perfluoroalkyl alkene skeleton. This initial step typically yields a mixture of Z and E isomers, dictated by the kinetics of the radical addition and the steric bulk of the substituents involved in the transition state.
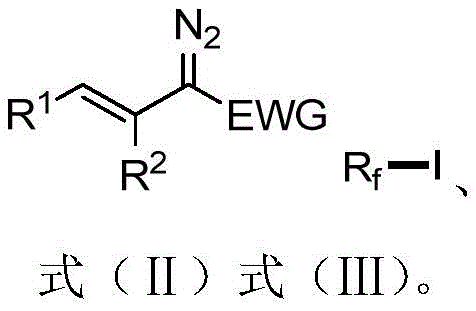
Following the initial ATRA reaction, the process employs a manganese catalyst, specifically decacarbonyl dimanganese (Mn2(CO)10), to drive the isomerization towards the thermodynamically stable or kinetically favored Z-isomer. Under continued visible light irradiation, the manganese catalyst likely facilitates a reversible radical process or a metal-hydride insertion-elimination mechanism that allows the double bond to equilibrate. This step is critical for ensuring high stereochemical purity, which is often a stringent requirement for biological activity in pharmaceutical candidates. The mechanism is robust enough to tolerate various functional groups, including esters, sulfones, and ketones, without compromising the integrity of the perfluoroalkyl chain. This mechanistic elegance ensures that impurities are minimized, providing R&D directors with a reliable pathway to generate high-purity OLED material or pharmaceutical precursors with consistent quality.
How to Synthesize 1-Iodo-3-Perfluoroalkyl Alkene Efficiently
The synthesis protocol outlined in the patent offers a straightforward, two-stage procedure that can be readily adapted for batch processing in standard laboratory or pilot plant reactors. The process begins with the mixing of the alkenyl diazoacetate and the perfluoroalkyl iodide in a suitable organic solvent such as acetonitrile or dichloromethane. Upon exposure to visible light, the ATRA reaction proceeds to completion, generating the crude isomer mixture. After removing excess starting material via distillation, the residue is subjected to the isomerization conditions using the manganese catalyst. Detailed standardized synthesis steps see the guide below.
- Perform an Atom Transfer Radical Addition (ATRA) reaction between an alkenyl diazo compound and a perfluoroalkyl iodide under visible light irradiation to generate a Z/E isomer mixture.
- Remove excess perfluoroalkyl iodide via reduced pressure distillation to purify the crude reaction mixture before the second step.
- Subject the Z/E mixture to a manganese-catalyzed isomerization reaction under continued visible light irradiation to selectively convert the mixture into the desired cis (Z) isomer.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement specialists and supply chain leaders, the adoption of this technology offers tangible strategic benefits beyond mere chemical novelty. The elimination of expensive noble metal photocatalysts and complex surfactant systems directly correlates to a significant reduction in raw material costs. Furthermore, the insensitivity of the reaction to air and moisture reduces the need for specialized inert atmosphere equipment, lowering capital expenditure requirements for manufacturing facilities. The simplified purification process, driven by high selectivity, reduces solvent usage and waste disposal costs, contributing to a more sustainable and economically viable production model.
- Cost Reduction in Manufacturing: The replacement of precious metal catalysts with abundant manganese complexes results in substantial cost savings on reagent procurement. Additionally, the high atom economy of the reaction, where nitrogen is the only byproduct, minimizes waste treatment expenses. The ability to operate under mild conditions also reduces energy consumption associated with heating and cooling, further optimizing the operational expenditure profile for large-scale production runs.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, including alkenyl diazo compounds and perfluoroalkyl iodides, are commercially available and chemically stable. The robustness of the reaction conditions means that production schedules are less likely to be disrupted by environmental factors or equipment failures related to strict anhydrous requirements. This reliability ensures a steady flow of high-purity pharmaceutical intermediates, mitigating the risk of stockouts for downstream drug manufacturing processes.
- Scalability and Environmental Compliance: The green synthesis concept inherent in this method aligns with increasingly stringent global environmental regulations. By avoiding toxic additives and minimizing waste generation, facilities can maintain compliance with lower regulatory overhead. The simplicity of the process facilitates easy scale-up from gram to kilogram scales without the need for complex process re-engineering, allowing for rapid response to market demand fluctuations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the process capabilities and limitations for potential partners.
Q: What are the primary advantages of this visible-light synthesis method over traditional thermal initiation?
A: The visible-light promoted method described in patent CN112047839B eliminates the need for harsh thermal initiators and complex additive systems. It operates under mild conditions (0-80°C), is insensitive to air and moisture, and produces nitrogen as the only byproduct, significantly simplifying downstream purification and reducing environmental impact compared to conventional surfactant-based methods.
Q: How does the process ensure high stereoselectivity for the Z-isomer?
A: The process utilizes a unique two-step strategy. First, a radical addition creates a mixture of isomers. Second, a specific manganese catalyst (such as decacarbonyl dimanganese) facilitates a photo-induced isomerization that thermodynamically favors the formation of the cis (Z) configuration, achieving Z/E ratios often exceeding 30:1 without requiring difficult chromatographic separation.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the method is explicitly designed for scalability. The reaction uses cheap and readily available raw materials, requires simple operational procedures, and avoids sensitive conditions. The high yields and straightforward product separation make it highly viable for commercial scale-up of complex fluorine-containing intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Iodo-3-Perfluoroalkyl Alkene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced fluorination technologies play in the development of next-generation therapeutics and high-performance materials. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising laboratory results of patent CN112047839B can be translated into industrial reality. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 1-iodo-3-perfluoroalkyl alkene meets the exacting standards required for pharmaceutical applications.
We invite you to collaborate with us to leverage this cutting-edge synthesis method for your specific project needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your supply chain efficiency and reduce your overall time to market.
