Scalable Synthesis of Spiro-chroman-4,3'-oxindoles via Metal-Free Dearomatization for Pharmaceutical Applications
Introduction to Next-Generation Spiro-Oxindole Synthesis
The landscape of pharmaceutical intermediate manufacturing is undergoing a significant transformation driven by the need for sustainable, cost-effective, and high-purity synthetic routes. A pivotal advancement in this domain is detailed in patent CN108976243B, which discloses a groundbreaking method for synthesizing spiro-chroman-4,3'-oxindole compounds. This technology leverages the power of biomass-derived platform molecules, specifically 2,5-dimethylfuran (2,5-DMF), reacting with oxyindole-containing o-hydroxybenzyl alcohols. Unlike traditional methodologies that rely on scarce and costly transition metals, this innovation utilizes a Bronsted acid-catalyzed intermolecular dearomatization strategy. For R&D directors and procurement specialists alike, this represents a paradigm shift towards greener chemistry that does not compromise on efficiency or structural complexity. The ability to construct these privileged scaffolds in a single step with high atom economy addresses critical bottlenecks in the supply chain of complex heterocyclic intermediates used in drug discovery.
The significance of this patent extends beyond mere academic interest; it offers a tangible solution for the commercial scale-up of complex pharmaceutical intermediates. By utilizing 2,5-DMF, a renewable resource, the process aligns with global sustainability goals while simultaneously reducing dependency on petrochemical feedstocks. The reaction proceeds under remarkably mild conditions, typically at room temperature, which drastically lowers energy consumption compared to thermal cycloadditions. Furthermore, the methodology demonstrates exceptional substrate universality, tolerating a wide range of electronic and steric environments on both the oxindole and the furan components. This robustness is essential for process chemists aiming to diversify their compound libraries without re-optimizing reaction parameters for every new analog, thereby accelerating the timeline from benchtop synthesis to pilot plant production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
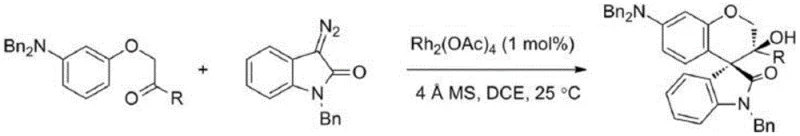
Prior to this innovation, the construction of the spiro-chroman-4,3'-oxindole core was predominantly achieved through transition metal-catalyzed pathways, most notably those involving rhodium complexes. As illustrated in the background art, earlier methods developed by research groups such as Hu Wenhao's utilized Rh2(OAc)4 to facilitate the reaction between α-phenol ketones and diazo oxindoles. While scientifically valid, these approaches present severe limitations for industrial application. The primary drawback is the reliance on rhodium, a precious metal with volatile pricing and limited global supply, which directly inflates the Cost of Goods Sold (COGS). Moreover, the presence of heavy metals in the final active pharmaceutical ingredient (API) or intermediate is strictly regulated. Removing trace rhodium to meet ppm-level specifications requires additional purification steps, such as scavenging resins or extensive recrystallization, which reduces overall yield and increases waste generation. Additionally, the synthesis of diazo precursors often involves hazardous reagents and multi-step sequences, lowering the overall atom economy and introducing safety risks associated with potentially explosive intermediates.
The Novel Approach
In stark contrast, the method disclosed in CN108976243B circumvents these issues by employing a metal-free organocatalytic system. The core of this novelty lies in the activation of oxyindole-containing o-hydroxybenzyl alcohols using simple Bronsted acids like p-toluenesulfonic acid (p-TSA). This activation generates an reactive ortho-quinone methide (o-QM) intermediate in situ, which then undergoes a [4+2] cycloaddition with 2,5-dimethylfuran. This approach effectively transforms the furan from a stable aromatic compound into a reactive dienophile through a dearomatization process. The elimination of transition metals not only simplifies the workup procedure—often requiring only standard aqueous washes and column chromatography—but also ensures the final product is free from toxic metal contaminants. This "green" attribute is increasingly becoming a prerequisite for suppliers aiming to partner with top-tier multinational pharmaceutical companies who prioritize environmental, social, and governance (ESG) criteria in their vendor selection processes.
Mechanistic Insights into Bronsted Acid-Catalyzed Dearomatization
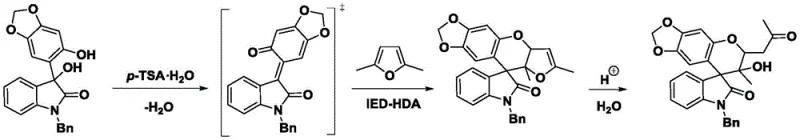
Understanding the mechanistic underpinnings of this transformation is crucial for R&D teams looking to adapt this chemistry for proprietary pipelines. The reaction initiates with the protonation of the hydroxyl group on the o-hydroxybenzyl alcohol substrate by the Bronsted acid catalyst. This facilitates the elimination of a water molecule, generating a highly electrophilic ortho-quinone methide (o-QM) species. This o-QM intermediate serves as the electron-deficient component in the subsequent cycloaddition. Uniquely, 2,5-dimethylfuran, typically considered an electron-rich diene, acts here as a dienophile in an Inverse Electron Demand Hetero-Diels-Alder (IED-HDA) reaction. This reversal of roles is thermodynamically challenging due to the aromatic stability of the furan ring, but the high reactivity of the o-QM drives the process forward. The resulting bicyclic intermediate subsequently undergoes acid-catalyzed hydrolysis and ring-opening to yield the final spiro-chroman-4,3'-oxindole structure. This intricate cascade occurs seamlessly in one pot, showcasing the elegance of the design.
From an impurity control perspective, this mechanism offers distinct advantages. The high diastereoselectivity observed (dr > 20:1) suggests a highly ordered transition state, likely governed by hydrogen bonding interactions between the catalyst, the o-QM, and the approaching furan molecule. This stereocontrol minimizes the formation of unwanted diastereomers, which are often difficult to separate and can complicate regulatory filings. Furthermore, because the reaction avoids radical pathways often associated with metal catalysis, the impurity profile is cleaner, consisting mainly of unreacted starting materials or simple hydrolysis byproducts rather than complex metal-ligand complexes or oligomerization products. For quality assurance teams, this predictability translates to more robust analytical methods and higher confidence in batch-to-batch consistency, which is paramount when supplying high-purity pharmaceutical intermediates for clinical trials.
How to Synthesize Spiro-chroman-4,3'-oxindole Efficiently
Implementing this synthesis in a laboratory or pilot setting requires attention to solvent choice and catalyst loading to maximize efficiency. The patent data indicates that 1,2-dichloroethane (DCE) is the optimal solvent, providing the best balance of solubility and reaction rate. The process is operationally simple: the substrates are mixed at room temperature, and the reaction progress can be easily monitored via thin-layer chromatography (TLC). The mild conditions mean that specialized pressure vessels or heating mantles are unnecessary, reducing capital expenditure for equipment. Below is the standardized protocol derived from the patent examples, which serves as a baseline for further process optimization.
- Dissolve the oxyindole-containing o-hydroxybenzyl alcohol substrate in a suitable solvent such as 1,2-dichloroethane (DCE) within a reaction vessel.
- Add 2,5-dimethylfuran to the solution at a molar ratio of 1: 3 relative to the alcohol substrate, followed by the addition of a Bronsted acid catalyst like p-TSA·H2O (10 mol%).
- Stir the reaction mixture at 25°C until completion monitored by TLC, then purify the crude product using silica gel column chromatography to isolate the target spiro-compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology offers compelling strategic benefits that extend beyond the laboratory. The shift from precious metal catalysis to organocatalysis fundamentally alters the cost structure of manufacturing these complex scaffolds. By removing rhodium from the equation, the direct material costs are significantly reduced, and the supply chain becomes more resilient against fluctuations in the precious metals market. Additionally, the use of 2,5-dimethylfuran, a biomass-derived platform chemical, ensures a sustainable and potentially lower-cost feedstock compared to petroleum-derived synthons. This alignment with bio-based economies future-proofs the supply chain against regulatory pressures regarding carbon footprints and fossil fuel dependency.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts such as rhodium removes a major cost driver from the bill of materials. Furthermore, the simplified downstream processing required to remove metal residues reduces solvent consumption and waste disposal costs. The high yields reported (up to 85%) mean less raw material is wasted, improving the overall mass balance and economic efficiency of the production line. This cumulative effect leads to substantial cost savings per kilogram of the final intermediate, allowing for more competitive pricing in the global market.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals like p-TSA and biomass-derived furans mitigates the risk of supply disruptions often associated with specialized catalysts. These reagents are widely available from multiple global suppliers, ensuring continuity of supply even during geopolitical or logistical disturbances. The robustness of the reaction conditions (room temperature, ambient pressure) also means that production can be easily transferred between different manufacturing sites without requiring extensive re-validation of specialized equipment, thereby enhancing the flexibility and reliability of the supply network.
- Scalability and Environmental Compliance: The metal-free nature of this process simplifies environmental compliance, as there is no need for rigorous testing and certification regarding heavy metal leaching. This accelerates the regulatory approval process for new drug applications containing these intermediates. Moreover, the mild reaction conditions and high atom economy align with green chemistry principles, reducing the generation of hazardous waste. This makes the process easier to scale from gram to ton quantities while maintaining a favorable environmental profile, which is increasingly critical for maintaining a social license to operate in the fine chemical industry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis method. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity for potential partners evaluating this technology for their own development programs.
Q: What are the advantages of this metal-free synthesis over traditional rhodium-catalyzed methods?
A: This novel method eliminates the need for expensive transition metal catalysts like rhodium, significantly reducing raw material costs and removing the complex downstream processing required to eliminate toxic heavy metal residues from the final pharmaceutical intermediate.
Q: What is the typical yield and stereoselectivity achieved in this process?
A: Under optimized conditions using p-TSA·H2O in DCE at room temperature, the reaction achieves isolated yields up to 85% with excellent diastereoselectivity (dr > 20:1), ensuring high purity of the desired spiro-chroman scaffold.
Q: Is this synthesis method suitable for large-scale commercial production?
A: Yes, the process operates at ambient temperature (25°C) using commercially available biomass-derived feedstocks and simple organic acids, making it highly amenable to scale-up without requiring specialized high-pressure or cryogenic equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Spiro-chroman-4,3'-oxindole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic route described in CN108976243B for the production of high-value spiro-oxindole derivatives. As a leading CDMO partner, we possess the technical expertise to translate this academic breakthrough into a robust, GMP-compliant manufacturing process. Our facilities are equipped to handle diverse synthetic pathways, ranging from early-stage milligram synthesis for drug discovery to extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We understand that the integrity of your supply chain depends on consistent quality; therefore, our stringent purity specifications and rigorous QC labs ensure that every batch of spiro-chroman-4,3'-oxindole meets the exacting standards required for pharmaceutical applications.
We invite you to collaborate with us to leverage this metal-free technology for your next project. Whether you require custom synthesis of specific analogs or bulk manufacturing of the core scaffold, our team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and discuss route feasibility assessments. By partnering with us, you secure a reliable source of advanced pharmaceutical intermediates that combine cutting-edge science with commercial viability.
