Revolutionizing Organosilicon Synthesis: Scalable Production of Exo-Gem-Disilyl Alkanes via Earth-Abundant Cobalt Catalysis
Revolutionizing Organosilicon Synthesis: Scalable Production of Exo-Gem-Disilyl Alkanes via Earth-Abundant Cobalt Catalysis
The landscape of organosilicon chemistry is undergoing a significant transformation driven by the urgent need for sustainable, cost-effective, and highly selective synthetic methodologies. Patent CN109705154B introduces a groundbreaking approach to synthesizing exo-gem-disilyl alkane compounds containing four silicon-hydrogen bonds, addressing critical bottlenecks in the production of high-value pharmaceutical intermediates and fine chemicals. This technology leverages a chiral CoX2-IIP complex to catalyze the tandem hydrosilylation of terminal alkynes with trihydrosilanes, achieving moderate to excellent yields ranging from 51% to 99%. By shifting away from expensive and toxic precious metal catalysts towards earth-abundant cobalt, this innovation not only enhances atom economy but also aligns with the rigorous environmental and safety standards demanded by modern global supply chains. The ability to introduce two silyl groups onto the same carbon atom with high regioselectivity opens new avenues for constructing complex molecular architectures essential for drug discovery and advanced material science.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of gem-disilylalkanes has been plagued by significant technical and economic challenges that hinder their widespread adoption in industrial settings. Traditional methods often rely on multi-step sequences involving unstable intermediates or require the use of prohibitively expensive transition metal catalysts such as palladium, platinum, rhodium, and ruthenium. For instance, earlier strategies reported by research groups involved [1,4]-reverse Brook reactions or palladium-carbene insertions, which frequently suffer from poor atom economy and limited substrate scope. A major drawback of these conventional pathways is the difficulty in controlling regioselectivity during tandem reactions, often leading to complex mixtures of isomers that are arduous and costly to separate. Furthermore, the reliance on precious metals introduces severe supply chain vulnerabilities, as the availability and price volatility of these resources can drastically impact production timelines and overall manufacturing costs. The presence of toxic heavy metal residues in the final product also necessitates extensive and expensive purification protocols to meet the stringent purity specifications required for pharmaceutical applications, thereby adding unnecessary complexity to the process.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in patent CN109705154B presents a streamlined, single-pot solution that utilizes a chiral cobalt catalyst to drive the efficient conversion of alkynes and trihydrosilanes into exo-gem-disilyl alkanes. This novel approach capitalizes on the unique reactivity of earth-abundant cobalt complexes coordinated with imidazopyridinylimidazoline (IIP) ligands, enabling a highly selective tandem hydrosilylation reaction. The process operates under mild conditions, typically between -30°C and 80°C, and can be conducted in common organic solvents like toluene or even under solvent-free conditions, offering remarkable operational flexibility. By employing sodium triethylborohydride as a reducing agent, the system activates the cobalt catalyst in situ, facilitating the successive addition of silicon atoms to the terminal alkyne with exceptional precision. This methodology not only circumvents the need for toxic precious metal salts but also achieves regioselectivity ratios exceeding 19:1 in most cases, ensuring the production of high-purity intermediates with minimal byproduct formation. The simplicity of the operation, combined with the high atom economy of the reaction, represents a paradigm shift in how organosilicon building blocks are manufactured for commercial applications.

Mechanistic Insights into Cobalt-Catalyzed Tandem Hydrosilylation
The success of this synthetic route lies in the sophisticated design of the chiral CoX2-IIP catalyst system, which orchestrates the reaction through a well-defined mechanistic pathway. The catalyst, represented structurally in the patent as Formula III or its enantiomer, features a robust coordination environment that stabilizes the active cobalt species while imparting the necessary steric and electronic properties to control selectivity. During the reaction, the cobalt center facilitates the activation of the silicon-hydrogen bond in the trihydrosilane reagent, generating a reactive cobalt-hydride species that inserts into the carbon-carbon triple bond of the alkyne. This initial hydrometalation step is critical, as it sets the stage for the subsequent silylation event. The unique architecture of the IIP ligand ensures that the reaction proceeds through a specific trajectory that favors the formation of the gem-disilyl product over other potential isomers. The presence of the reducing agent, such as sodium triethylborohydride, plays a pivotal role in regenerating the active catalytic species, allowing the cycle to continue efficiently with low catalyst loading, often as low as 1 mol% to 5 mol%. This mechanistic efficiency translates directly into lower operational costs and reduced waste generation, making the process highly attractive for large-scale manufacturing.
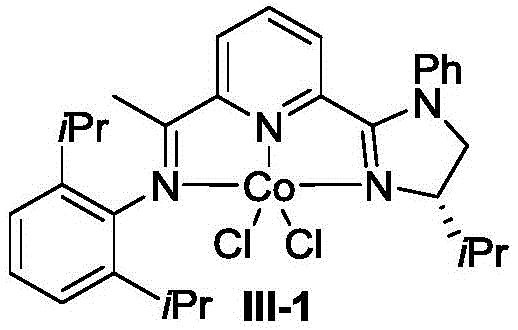
Impurity control is another critical aspect where this cobalt-catalyzed system excels, primarily due to its high regioselectivity and chemoselectivity. In traditional hydrosilylation reactions, competing pathways such as anti-Markovnikov addition or over-reduction can lead to a plethora of side products that complicate downstream processing. However, the chiral environment provided by the IIP ligand effectively suppresses these undesired pathways, channeling the reactants almost exclusively towards the desired exo-gem-disilyl alkane structure. The patent data indicates that regioselectivity can reach levels greater than 19:1, which implies that the ratio of the desired product to any regioisomeric impurities is exceptionally high. This level of purity is paramount for pharmaceutical intermediates, where even trace amounts of structural analogs can affect the efficacy and safety of the final drug substance. By minimizing the formation of impurities at the source, this technology significantly reduces the burden on purification units, allowing for simpler workup procedures such as column chromatography or distillation. The robustness of the catalyst also means that it tolerates a wide range of functional groups on the alkyne substrate, including halogens, esters, and heterocycles, without compromising the integrity of the reaction or generating degradation products.
How to Synthesize Exo-Gem-Disilyl Alkanes Efficiently
The practical implementation of this synthesis method is designed to be straightforward and adaptable to various production scales, from laboratory discovery to commercial manufacturing. The process begins with the preparation of the reaction vessel under an inert atmosphere, typically nitrogen or argon, to prevent catalyst deactivation by oxygen or moisture. Key reagents, including the terminal alkyne, trihydrosilane, and the chiral cobalt catalyst, are introduced in precise stoichiometric ratios optimized for maximum yield and selectivity. The reaction mixture is then stirred at controlled temperatures, allowing the tandem hydrosilylation to proceed to completion within a timeframe of 0.5 to 60 hours, depending on the specific substrate and conditions employed. Following the reaction, standard workup techniques are applied to isolate the crude product, which is subsequently purified to meet the required quality standards. For a detailed breakdown of the specific operational parameters, reagent quantities, and safety precautions required for this synthesis, please refer to the standardized guide below.
- Prepare the reaction system under inert gas protection, utilizing a chiral CoX2-IIP complex as the catalyst and sodium triethylborohydride as the reducing agent.
- React terminal alkynes with trihydrosilanes in an organic solvent such as toluene at temperatures ranging from -30°C to 80°C to achieve high regioselectivity.
- Perform post-treatment purification via column chromatography or distillation to isolate the high-purity exo-gem-disilyl alkane product with yields up to 99%.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this cobalt-catalyzed technology offers substantial strategic benefits that extend far beyond simple chemical transformation. The most immediate impact is seen in cost reduction, driven by the replacement of scarce and volatile precious metal catalysts with abundant and affordable cobalt salts. This shift eliminates the financial risks associated with fluctuating prices of palladium or platinum and removes the need for expensive metal scavenging processes often required to meet residual metal limits in pharmaceutical products. Additionally, the high atom economy of the reaction ensures that a greater proportion of raw materials are converted into the final product, minimizing waste disposal costs and maximizing resource efficiency. The mild reaction conditions further contribute to energy savings, as the process does not require extreme heating or cooling, thereby lowering the overall utility consumption of the manufacturing facility. These factors combine to create a more resilient and cost-effective supply chain capable of delivering high-quality intermediates at competitive prices.
- Cost Reduction in Manufacturing: The transition from precious metal catalysts to earth-abundant cobalt systems fundamentally alters the cost structure of organosilicon production. By eliminating the dependency on expensive metals like rhodium and ruthenium, manufacturers can achieve significant savings on raw material procurement. Furthermore, the high selectivity of the reaction reduces the loss of valuable starting materials to byproducts, enhancing the overall yield and reducing the cost per kilogram of the final active ingredient. The simplified purification process also lowers labor and consumable costs associated with chromatography and filtration, contributing to a leaner and more profitable manufacturing operation.
- Enhanced Supply Chain Reliability: Relying on cobalt, a metal with a more stable and diversified supply base compared to platinum group metals, mitigates the risk of supply disruptions. The reagents used in this process, such as terminal alkynes and trihydrosilanes, are commercially available from multiple sources, ensuring continuity of supply even in volatile market conditions. The robustness of the catalyst system allows for consistent batch-to-batch reproducibility, which is critical for maintaining long-term contracts with pharmaceutical clients. This reliability enables supply chain planners to forecast production schedules with greater accuracy and reduce the need for excessive safety stock, optimizing inventory management and working capital.
- Scalability and Environmental Compliance: The scalability of this process is evidenced by its ability to operate under solvent-free conditions or in common industrial solvents like toluene, facilitating easy scale-up from pilot plants to multi-ton production facilities. The absence of toxic heavy metals simplifies regulatory compliance, particularly regarding environmental discharge and worker safety standards. Waste streams generated from this process are less hazardous and easier to treat, aligning with the increasing global emphasis on green chemistry and sustainable manufacturing practices. This environmental advantage not only reduces liability but also enhances the corporate reputation of manufacturers as responsible stewards of the environment, a key factor in securing partnerships with sustainability-focused multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of exo-gem-disilyl alkanes. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on the capabilities and limitations of this innovative technology. Understanding these details is essential for R&D teams evaluating the feasibility of incorporating these intermediates into their synthesis routes and for procurement professionals assessing the viability of long-term sourcing strategies.
Q: What are the primary advantages of using cobalt catalysts over traditional palladium or platinum systems for hydrosilylation?
A: The use of earth-abundant cobalt catalysts significantly reduces raw material costs compared to precious metals like palladium, platinum, or rhodium. Furthermore, the chiral CoX2-IIP complex described in patent CN109705154B eliminates the need for toxic transition metal salts, simplifying downstream purification and ensuring higher biocompatibility for pharmaceutical applications.
Q: How does this synthesis method improve regioselectivity in gem-disilyl compound formation?
A: The novel method utilizes a specialized chiral imidazopyridinylimidazoline (IIP) ligand system that directs the tandem hydrosilylation with exceptional precision. The patent reports regioselectivity ratios ranging from 10:1 to greater than 19:1, effectively minimizing the formation of unwanted isomers and reducing the burden on purification processes.
Q: Can the resulting exo-gem-disilyl alkanes be further functionalized for diverse chemical applications?
A: Yes, the presence of four silicon-hydrogen bonds in the product structure provides multiple sites for subsequent modification. These compounds can be oxidized to synthesize aliphatic aldehydes or reacted with alkynyl compounds to form polysubstituted alkenyl silanes, offering significant versatility for complex molecule construction in medicinal chemistry.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Exo-Gem-Disilyl Alkane Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the cobalt-catalyzed synthesis of exo-gem-disilyl alkanes in advancing pharmaceutical and material science applications. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of high-purity intermediates. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including the precise determination of regioselectivity and residual metal content. We are committed to leveraging this cutting-edge technology to deliver cost-effective solutions that accelerate our partners' drug development timelines while adhering to the highest standards of quality and safety.
We invite you to collaborate with us to explore how this efficient synthesis method can optimize your specific project requirements. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating the tangible economic benefits of switching to this cobalt-catalyzed route. Please contact us today to request specific COA data for our exo-gem-disilyl alkane portfolio and to discuss route feasibility assessments for your next-generation therapeutic candidates. Together, we can build a more sustainable and efficient supply chain for the future of fine chemical manufacturing.
