Advanced Photocatalytic Synthesis of Cyano Carboxylic Acids for Commercial Scale-up
The pharmaceutical and fine chemical industries are constantly seeking safer, more sustainable pathways to construct complex molecular architectures, particularly those containing versatile functional groups like nitriles and carboxylic acids. Patent CN113773227A introduces a groundbreaking methodology for the preparation of ε-, ζ-, and η-cyanocarboxylic acids, utilizing carbon dioxide as a C1 building block under visible-light photocatalysis. This technology represents a significant paradigm shift from classical stoichiometric reactions to catalytic, atom-economical processes. By leveraging inexpensive and readily available raw materials such as olefins and cyclic ketoximes, this invention addresses critical pain points in the synthesis of biologically active compounds, including precursors for Vitamin B6, caffeine, and folic acid. Furthermore, the resulting aliphatic dicarboxylic acid derivatives serve as essential monomers for high-performance polyamides and polyesters, underscoring the broad industrial applicability of this innovation beyond just pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of cyano carboxylic acid scaffolds has relied heavily on nucleophilic substitution reactions involving halogenated carboxylic acids and sodium cyanide or hydrocyanic acid. While chemically straightforward, these classical routes present formidable challenges for modern manufacturing environments. The primary concern is the extreme toxicity of cyanide reagents, which necessitates rigorous safety protocols, specialized containment infrastructure, and complex waste neutralization procedures to prevent the release of lethal hydrogen cyanide gas. Additionally, alternative strategies involving the partial hydrolysis of dinitrile compounds suffer from poor selectivity; controlling reaction conditions to hydrolyze only one cyano group while leaving the other intact is notoriously difficult, often leading to mixtures of mono-acids, di-acids, and unreacted starting materials that are costly to separate. These inefficiencies translate directly into higher production costs, extended lead times, and increased environmental liability, making traditional methods less attractive for large-scale commercial operations.
The Novel Approach
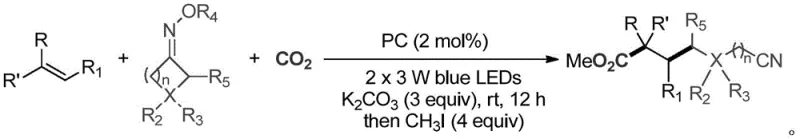
In stark contrast, the methodology disclosed in CN113773227A offers a streamlined, catalytic alternative that circumvents the use of toxic cyanide sources entirely. By employing a continuous free radical addition, single electron reduction, and carboxylation sequence, this process effectively fixes carbon dioxide into the molecular backbone under mild conditions. The reaction proceeds at room temperature using low-energy 2x3W blue LEDs, significantly reducing the thermal load and energy consumption compared to high-temperature thermal processes. The use of cyclic ketoximes as radical precursors allows for the precise installation of the cyano group via ring-opening, while the olefin component dictates the carbon chain length and substitution pattern. This modularity enables the rapid generation of structural diversity, allowing manufacturers to access a wide library of ε-, ζ-, and η-cyano carboxylic acid derivatives from a common set of building blocks without the need for hazardous reagents or harsh reaction conditions.
Mechanistic Insights into Visible-Light Photocatalytic Carboxylation
The core of this transformation lies in the sophisticated interplay between the photocatalyst, the cyclic ketoxime ester, and the olefin substrate under visible light irradiation. Upon excitation by blue LEDs, the photocatalyst—whether it be an Iridium complex like Ir[dF(CF3)ppy]2(dtbbpy)PF6, a Ruthenium salt, or the metal-free organic photocatalyst 4CzIPN—enters an excited state capable of engaging in single electron transfer (SET) events. The cyclic ketoxime ester acts as a radical precursor; upon reduction by the excited photocatalyst, it undergoes fragmentation to release a cyano-alkyl radical and a stable leaving group. This generated radical species then adds across the double bond of the olefin substrate in a regioselective manner, forming a new carbon-carbon bond and a stabilized benzylic or alkyl radical intermediate. This intermediate is subsequently reduced by the reduced form of the photocatalyst to generate a carbanion, which rapidly traps carbon dioxide to form the carboxylate species. Finally, quenching with methyl iodide yields the desired ester product. This elegant cascade ensures high atom economy and minimizes side reactions, as each step is driven by the thermodynamic favorability of radical stabilization and CO2 fixation.
From an impurity control perspective, this mechanism offers distinct advantages over traditional ionic pathways. Because the reaction proceeds through discrete radical intermediates rather than highly reactive ionic species, there is a reduced tendency for polymerization or non-specific nucleophilic attacks on sensitive functional groups. The use of mild bases like DIPEA or triethylamine further suppresses base-catalyzed degradation pathways that might otherwise compromise the integrity of the ester or nitrile moieties. Moreover, the specificity of the radical ring-opening of the ketoxime ensures that the cyano group is introduced cleanly at the terminal position of the chain, avoiding the formation of regioisomers that are common in hydrolysis-based approaches. The result is a crude reaction mixture with a simplified impurity profile, facilitating downstream purification and ensuring that the final high-purity cyano carboxylic acid meets stringent pharmaceutical specifications with minimal processing.
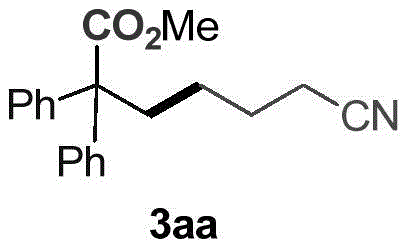
How to Synthesize Methyl 6-cyano-2,2-diphenylhexanoate Efficiently
To implement this synthesis in a laboratory or pilot setting, operators must adhere to precise stoichiometric ratios and atmospheric controls to maximize yield and reproducibility. The process begins with the careful degassing of the reaction vessel to remove oxygen, which can quench radical intermediates and inhibit the photocatalytic cycle. A typical protocol involves charging a Schlenk tube with the olefin, cyclic ketoxime, photocatalyst (2 mol%), and amine base in DMSO solvent, followed by multiple evacuation and CO2 backfill cycles to ensure a saturated atmosphere of the C1 source. The reaction is then subjected to blue LED irradiation at room temperature for 12 to 24 hours, allowing sufficient time for the radical cascade to reach completion. Following the carboxylation step, the addition of methyl iodide and heating to 50°C facilitates the final esterification. Detailed standardized synthesis steps are provided below to guide technical teams in replicating these results.
- Combine 1,1-stilbene, cyclobutanone O-(4-(trifluoromethyl)benzoyl)oxime, Ir photocatalyst (2 mol%), DIPEA, and DMSO in a Schlenk tube.
- Evacuate and backfill with CO2 three times, then stir at room temperature under 2x3W blue LED illumination for 12 hours.
- Add methyl iodide, stir at 50°C for 1 hour, then extract with ethyl acetate and purify via flash column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this photocatalytic technology offers compelling strategic benefits that extend beyond simple yield improvements. By eliminating the need for regulated toxic cyanide salts, facilities can drastically reduce the costs associated with hazardous material handling, storage, and disposal compliance. The shift from stoichiometric reagents to catalytic amounts of photocatalyst (as low as 2 mol%) significantly lowers the raw material cost per kilogram of product, especially when utilizing the metal-free 4CzIPN option which avoids the price volatility associated with precious metals like Iridium and Ruthenium. Furthermore, the mild reaction conditions (room temperature, ambient pressure) reduce the energy footprint of the manufacturing process, aligning with corporate sustainability goals and potentially lowering utility expenses. The simplicity of the post-reaction workup, which often requires only standard extraction and column chromatography, streamlines the production timeline and reduces the demand for specialized purification equipment.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous cyanide reagents removes a major cost center related to safety infrastructure and waste neutralization. Additionally, the high atom efficiency of using CO2 as a feedstock means that a greater proportion of the input mass is incorporated into the final product, reducing raw material waste. The ability to use inexpensive organic photocatalysts like 4CzIPN further decouples production costs from the fluctuating market prices of precious metals, providing a more stable and predictable cost structure for long-term manufacturing contracts.
- Enhanced Supply Chain Reliability: The starting materials for this process, such as substituted stilbenes and cyclic ketoximes, are commercially available or easily synthesized from commodity chemicals, ensuring a robust and resilient supply chain. Unlike processes dependent on scarce or geopolitically sensitive reagents, the inputs for this method are widely sourced, minimizing the risk of supply disruptions. The operational simplicity of the reaction, which does not require high-pressure reactors or cryogenic cooling, allows for flexible manufacturing across various facility types, enhancing the overall agility of the supply network to respond to market demand fluctuations.
- Scalability and Environmental Compliance: Scaling this photochemical process is facilitated by the availability of modern flow chemistry reactors and high-power LED arrays, which can overcome light penetration limitations inherent in batch processing. The absence of heavy metal waste streams (when using organic photocatalysts) and toxic cyanide byproducts simplifies environmental permitting and reduces the regulatory burden on manufacturing sites. This cleaner profile not only accelerates the timeline for regulatory approval of new drug substances but also future-proofs the manufacturing process against increasingly stringent environmental regulations regarding chemical emissions and waste disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photocatalytic carboxylation technology. These insights are derived directly from the experimental data and scope defined in the patent literature, providing a clear understanding of the method's capabilities and limitations for potential partners and licensees.
Q: Why is this photocatalytic method superior to traditional cyanide substitution?
A: Traditional methods rely on toxic sodium cyanide or hydrocyanic acid, posing severe safety and environmental hazards. This patent utilizes benign carbon dioxide and visible light, eliminating the need for hazardous cyanide reagents and simplifying waste treatment.
Q: What catalysts are compatible with this synthesis route?
A: The process supports a range of photocatalysts including Iridium complexes (e.g., Ir[dF(CF3)ppy]2(dtbbpy)PF6), Ruthenium complexes, and metal-free organic photocatalysts like 4CzIPN, offering flexibility for cost and purity requirements.
Q: What is the substrate scope for the olefin and ketoxime components?
A: The method tolerates a wide variety of substituted stilbenes (including fluoro, chloro, bromo, methoxy, and ester groups) and cyclic ketoximes (four to six-membered rings), enabling the synthesis of diverse epsilon-, zeta-, and eta-cyano carboxylic acids.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cyano Carboxylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic route described in CN113773227A for the production of high-value pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from bench-scale discovery to industrial manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with advanced photochemical reactors and rigorous QC labs capable of meeting stringent purity specifications required by global regulatory bodies. We are committed to delivering high-purity cyano carboxylic acids that consistently exceed quality expectations, leveraging our technical expertise to optimize every step of the synthesis for maximum yield and minimal impurity formation.
We invite you to collaborate with us to unlock the full commercial potential of this innovative chemistry. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and target specifications. Please contact us today to request specific COA data for our existing catalog of intermediates or to discuss route feasibility assessments for your proprietary molecules. Let us help you secure a sustainable, cost-effective supply chain for your next-generation therapeutic candidates.
