Advanced Catalytic Synthesis of N-Heterocyclic Aryl Quinazoline-4-Amines for Pharmaceutical Applications
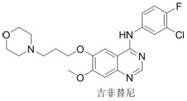
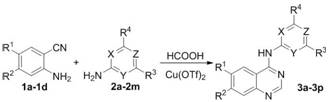
The pharmaceutical industry continuously seeks robust and sustainable pathways for constructing privileged scaffolds, particularly the quinazoline-4-amine core found in numerous FDA-approved kinase inhibitors such as gefitinib and erlotinib. Patent CN112457260A introduces a transformative methodology for synthesizing N-heterocyclic aryl quinazoline-4-amine compounds, utilizing a copper-catalyzed tandem reaction that fundamentally alters the economic and environmental landscape of producing these critical pharmaceutical intermediates. This innovative approach leverages copper trifluoromethanesulfonate (Cu(OTf)₂) to catalyze a direct three-component coupling of substituted anthranilonitriles, heterocyclic aromatic amines, and formic acid. By bypassing traditional multi-step activations, this technology offers a streamlined route that addresses long-standing challenges in impurity control and process safety, positioning it as a vital asset for any organization aiming to secure a reliable API intermediate supplier partnership.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the quinazoline-4-amine framework has relied heavily on classical synthetic strategies that are fraught with operational inefficiencies and environmental hazards. As illustrated in the traditional reaction pathways, the most common route involves the initial condensation of anthranilic acid derivatives to form quinazolin-4(3H)-ones, followed by a harsh chlorination step using phosphorus oxychloride (POCl₃) or thionyl chloride (SOCl₂) to generate reactive 4-chloroquinazolines. This chlorination stage is particularly problematic for large-scale manufacturing due to the generation of corrosive acidic waste streams and the requirement for specialized corrosion-resistant equipment. Furthermore, the subsequent nucleophilic aromatic substitution with amines often requires elevated temperatures and prolonged reaction times, leading to potential decomposition of sensitive heterocyclic moieties and the formation of difficult-to-remove side products.
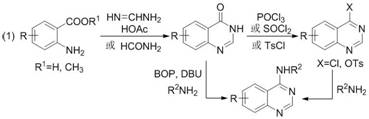
The Novel Approach
In stark contrast to these legacy methods, the novel protocol disclosed in the patent data utilizes a direct tandem addition/condensation/cyclization strategy that operates under significantly milder conditions. By employing formic acid not merely as a solvent but as an active reactant in the cyclization process, the method eliminates the need for pre-activation of the nitrile group via toxic chlorinating agents. The reaction proceeds efficiently at temperatures between 80°C and 110°C, which is substantially lower than the reflux conditions often required for SNAr reactions on chloroquinazolines. This shift in paradigm allows for the direct assembly of the target N-heterocyclic aryl quinazoline-4-amine structure in a single pot, drastically reducing the number of unit operations and minimizing the exposure of personnel to hazardous reagents, thereby enhancing the overall sustainability profile of the manufacturing process.
Mechanistic Insights into Cu(OTf)2-Catalyzed Cyclization
The efficacy of this transformation hinges on the unique Lewis acidic properties of copper trifluoromethanesulfonate, which acts as a potent catalyst for activating the nitrile group of the anthranilonitrile substrate towards nucleophilic attack by the heterocyclic amine. The mechanism likely involves the coordination of the copper center to the nitrogen of the nitrile, increasing its electrophilicity and facilitating the initial addition step to form an amidine intermediate. Subsequently, the formic acid participates in a condensation reaction, providing the necessary carbon unit for the closure of the pyrimidine ring. This cascade sequence is highly atom-economical, as the formic acid serves a dual role, and the catalyst loading can be optimized to as low as 0.05 mmol/mmol of substrate while maintaining high conversion rates. The choice of Cu(OTf)₂ over other copper salts like CuCl or CuI is critical, as the triflate anion provides a non-coordinating environment that maximizes the Lewis acidity of the copper cation, ensuring rapid turnover and high yields.
From an impurity control perspective, this mechanistic pathway offers distinct advantages over stepwise syntheses. In conventional routes, the isolation of the 4-chloroquinazoline intermediate often leads to hydrolysis byproducts if moisture is not strictly excluded, creating impurities that are structurally similar to the final product and challenging to separate. The one-pot nature of the Cu(OTf)₂ catalyzed method minimizes the handling of reactive intermediates, thereby reducing the opportunity for hydrolytic degradation or oxidative side reactions. Furthermore, the mild acidic environment provided by the formic acid solvent system helps to suppress the formation of polymeric byproducts that can occur under the strongly basic conditions sometimes required for alternative cyclization methods. This results in a cleaner crude reaction profile, which simplifies downstream purification and ensures that the final high-purity pharmaceutical intermediates meet stringent regulatory specifications with minimal processing.
How to Synthesize N-Heterocyclic Aryl Quinazoline-4-Amine Efficiently
Implementing this synthesis requires careful attention to the stoichiometry of the formic acid and the selection of the appropriate catalyst loading to maximize throughput. The process is designed to be operationally simple, involving the mixing of the substituted anthranilonitrile, the heterocyclic aromatic amine, and the catalyst in formic acid, followed by heating and stirring. Detailed standardized synthesis steps see the guide below.
- Mix substituted anthranilonitrile, heterocyclic aromatic amine, formic acid, and Cu(OTf)2 catalyst in a reactor.
- Heat the mixture to 80-110°C and stir for 8-16 hours to complete the tandem addition/condensation/cyclization.
- Cool, remove excess formic acid, neutralize with sodium bicarbonate, extract with ethyl acetate, and recrystallize from ethanol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this catalytic methodology translates into tangible strategic benefits regarding cost structure and supply continuity. The elimination of phosphorus oxychloride and thionyl chloride from the supply chain removes a significant logistical burden, as these reagents are subject to strict regulatory controls and require specialized storage and disposal protocols. By replacing them with formic acid, a commodity chemical with a stable global supply, manufacturers can mitigate the risk of production stoppages caused by the unavailability of controlled precursors. Additionally, the reduction in reaction steps from a multi-stage process to a one-pot operation inherently lowers the capital expenditure required for reactor occupancy time, allowing for increased production capacity within existing infrastructure without the need for significant facility upgrades.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the drastic simplification of the workflow and the removal of expensive waste treatment requirements associated with halogenated byproducts. Conventional chlorination routes generate substantial amounts of acidic wastewater that require neutralization and treatment before discharge, incurring high environmental compliance costs. In contrast, the formic acid-based system generates a much cleaner waste stream, significantly reducing the operational expenditure related to effluent management. Furthermore, the high yield and selectivity of the copper-catalyzed reaction minimize the loss of valuable starting materials, ensuring that the cost of goods sold (COGS) is optimized through superior material efficiency rather than just bulk purchasing power.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials such as substituted anthranilonitriles and simple heterocyclic amines ensures a robust supply chain that is less susceptible to geopolitical disruptions or raw material shortages. Unlike complex activated intermediates that may have limited suppliers, the precursors for this reaction are commoditized and produced by multiple vendors globally. This diversification of the supply base provides procurement teams with greater leverage in negotiations and ensures business continuity. The mild reaction conditions also mean that the process can be easily transferred between different manufacturing sites or contract manufacturing organizations (CMOs) without requiring specialized corrosion-resistant reactors, further enhancing the flexibility and resilience of the supply network.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces thermal management challenges, but the moderate temperature range of 80°C to 110°C for this reaction makes it highly amenable to scale-up in standard stainless steel reactors. The absence of exothermic hazards associated with the addition of chlorinating agents reduces the risk profile during large-batch production, facilitating smoother technology transfer from pilot plant to commercial scale. From an environmental standpoint, the alignment with green chemistry principles—specifically the prevention of waste and the use of safer solvents—positions manufacturers to meet increasingly rigorous corporate sustainability goals and regulatory standards. This proactive approach to environmental compliance future-proofs the production asset against tightening regulations on volatile organic compounds and hazardous waste disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical application of the technology for industrial partners seeking to optimize their production of kinase inhibitor scaffolds.
Q: What are the advantages of using Cu(OTf)2 over traditional chlorinating agents?
A: Using Cu(OTf)2 eliminates the need for toxic reagents like POCl3 and SOCl2, significantly reducing hazardous waste and simplifying post-treatment purification.
Q: Can this method be scaled for industrial production of kinase inhibitors?
A: Yes, the mild reaction conditions (80-110°C) and use of formic acid as both solvent and reagent make the process highly suitable for large-scale commercial manufacturing.
Q: What is the typical purity profile of the synthesized intermediates?
A: The method yields high-purity products through simple recrystallization, avoiding complex chromatographic separations often required in multi-step conventional routes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Heterocyclic Aryl Quinazoline-4-Amine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and sustainable synthesis routes in the development of next-generation oncology therapeutics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to market supply is seamless and reliable. We are committed to delivering stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of intermediate meets the exacting standards required for pharmaceutical drug substance manufacturing. Our capability to implement advanced catalytic technologies like the Cu(OTf)₂ mediated cyclization allows us to offer competitive pricing without compromising on quality or delivery performance.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis method can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the potential economic benefits specific to your project volume. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your project moves forward with the most efficient and commercially viable chemical strategy available.
