Scalable Synthesis of Chiral Purine[3,2-c]Oxazoles via Nickel-Catalyzed Cycloaddition for Advanced Drug Discovery
Introduction to Advanced Purine Scaffold Synthesis
The development of efficient synthetic routes for complex heterocyclic scaffolds remains a cornerstone of modern medicinal chemistry. Patent CN115403593A introduces a groundbreaking methodology for the asymmetric synthesis of chiral purine[3,2-c]oxazole compounds, a structurally unique class of tricyclic purine derivatives. These compounds are increasingly recognized for their potent pharmacological profiles, often surpassing their bicyclic precursors in terms of bioactivity and selectivity. The patent discloses a robust [3+2] cycloaddition strategy that constructs the oxazole ring directly onto the purine core with high stereocontrol. This innovation addresses a critical gap in the literature, as prior methods for generating such tricyclic systems were either non-existent or suffered from poor efficiency. For a reliable pharmaceutical intermediate supplier, mastering this chemistry opens doors to new libraries of anticancer agents.
![General structure of chiral purine[3,2-c]oxazole compounds showing variable substituents](/insights/img/chiral-purine-oxazole-nickel-catalysis-pharma-supplier-20260307110009-01.png)
The core innovation lies in the utilization of (E)-2,6-disubstituted-9-acrylates and aryl oxirane dicarboxylic diesters as building blocks. By leveraging a specific nickel-based catalytic system, the process achieves excellent regioselectivity and enantioselectivity. The resulting scaffolds serve as versatile intermediates that can be further derivatized into diverse drug candidates. This technology represents a significant leap forward in the cost reduction in pharmaceutical intermediate manufacturing, offering a streamlined path to high-value chemical entities that were previously difficult to access.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of tricyclic purine systems has been fraught with synthetic challenges. Traditional approaches often rely on multi-step sequences involving harsh cyclization conditions, which can lead to decomposition of sensitive functional groups or racemization of chiral centers. Furthermore, existing methods for forming five-membered oxygen-containing rings fused to purines typically lack generality, requiring substrate-specific optimization that hinders rapid library generation. The absence of reported methods for chiral purine[3,2-c]oxazoles prior to this invention underscores the difficulty of controlling stereochemistry in such congested molecular environments. These limitations result in prolonged development timelines and increased material costs for research teams seeking to explore this chemical space.
The Novel Approach
The methodology described in CN115403593A circumvents these hurdles through a direct [3+2] cycloaddition between an unsaturated dipole and an epoxide. This atom-economical transformation builds complexity rapidly from simple starting materials. The use of a chiral nickel catalyst allows for the precise installation of stereocenters during the ring-forming event, eliminating the need for resolution steps that waste half of the material. As illustrated in the reaction scheme below, the process tolerates a wide range of substituents on both the purine and the epoxide components.
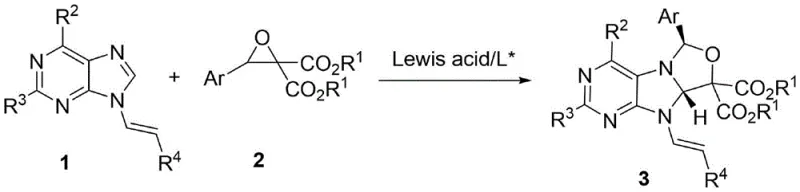
This novel approach not only simplifies the synthetic route but also enhances the overall yield and purity of the final products. By operating under mild conditions, the method preserves the integrity of sensitive ester and halogen functionalities, which are crucial for downstream diversification. For procurement managers, this translates to a more predictable supply chain for high-purity pharmaceutical intermediates, reducing the risk of batch failures associated with unstable reaction pathways.
Mechanistic Insights into Ni-Catalyzed Asymmetric Cycloaddition
The success of this transformation hinges on the synergistic interaction between the nickel center and the chiral bis-oxazoline ligand L7. Mechanistically, the Lewis acidic nickel species activates the epoxide towards nucleophilic attack by the purine acrylate. The bulky tert-butyl groups on the ligand create a well-defined chiral pocket that dictates the facial selectivity of the addition. This steric environment ensures that the reaction proceeds through a specific transition state, leading to the predominant formation of one enantiomer. Detailed screening revealed that Ni(ClO4)2·6H2O outperforms other Lewis acids like Sc(OTf)3 or Cu(ClO4)2, likely due to its optimal coordination geometry and Lewis acidity strength.
Impurity control is inherently built into this catalytic cycle. The high regioselectivity prevents the formation of unwanted isomers, while the mild reaction temperature of 0°C suppresses side reactions such as polymerization or hydrolysis. The patent data indicates that the diastereomeric ratio (dr) often exceeds 20:1, simplifying the purification process significantly. This level of control is essential for meeting the stringent purity specifications required in API synthesis. Understanding these mechanistic nuances allows process chemists to fine-tune reaction parameters for maximum efficiency and minimal waste generation.
How to Synthesize Chiral Purine[3,2-c]Oxazoles Efficiently
Executing this synthesis requires careful attention to catalyst preparation and reaction atmosphere. The protocol involves generating the active nickel-ligand complex in situ under an inert nitrogen atmosphere to prevent catalyst deactivation. Subsequent addition of substrates at controlled low temperatures ensures optimal stereoinduction. While the standard procedure yields excellent results, further enrichment of enantiomeric excess can be achieved through simple crystallization techniques, as demonstrated in the patent examples. The detailed standardized synthesis steps are provided in the guide below.
- Prepare the catalytic system by mixing Ni(ClO4)2·6H2O and chiral bis-oxazoline ligand L7 in dichloromethane under nitrogen.
- Add the purine acrylate substrate and aryl epoxide dicarboxylate at 0°C and stir for 72 hours.
- Purify the crude product via silica gel chromatography and optionally enrich enantiomeric excess through crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented technology offers substantial benefits for supply chain reliability and cost management. The ability to produce complex chiral scaffolds in fewer steps directly impacts the bottom line by reducing raw material consumption and processing time. Moreover, the scalability of the reaction has been experimentally verified, providing confidence for large-scale manufacturing campaigns. This reduces lead time for high-purity pharmaceutical intermediates, allowing drug developers to accelerate their preclinical programs without waiting for process optimization.
- Cost Reduction in Manufacturing: The elimination of resolution steps and the use of commercially available starting materials significantly lower the cost of goods. The high atom economy of the cycloaddition minimizes waste disposal costs, contributing to a more sustainable and economical production process. By avoiding expensive chiral auxiliaries or resolving agents, manufacturers can achieve substantial cost savings while maintaining high product quality.
- Enhanced Supply Chain Reliability: The robustness of the nickel catalytic system ensures consistent batch-to-batch reproducibility. The reaction tolerates various functional groups, meaning a single platform technology can be used to produce a diverse array of analogues. This flexibility mitigates supply risks associated with specialized reagents, ensuring a steady flow of materials for clinical trial demands.
- Scalability and Environmental Compliance: The patent explicitly demonstrates gram-scale synthesis with maintained efficiency, proving the feasibility of commercial scale-up of complex pharmaceutical intermediates. The use of dichloromethane as a solvent is standard in the industry, and the mild conditions reduce energy consumption. Furthermore, the high selectivity reduces the burden on downstream purification, aligning with green chemistry principles by minimizing solvent usage and waste generation.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this synthesis method. These answers are derived directly from the experimental data and claims within the patent documentation, providing clarity for technical teams evaluating this route for their specific projects.
Q: What is the optimal catalyst system for this cycloaddition?
A: The patent identifies Ni(ClO4)2·6H2O combined with the chiral bis-oxazoline ligand L7 as the optimal system, providing superior enantioselectivity compared to other Lewis acids.
Q: Can this reaction be scaled up for commercial production?
A: Yes, the patent demonstrates successful gram-scale synthesis (up to 5 mmol scale) with maintained high yields and enantiomeric excess, indicating strong potential for commercial scale-up.
Q: What are the biological applications of these compounds?
A: The synthesized tricyclic purine derivatives exhibit significant inhibitory activity against human colon cancer cell lines HCT-116 and SW480, highlighting their value in oncology drug discovery.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Purine[3,2-c]Oxazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this nickel-catalyzed cycloaddition technology for oncology drug discovery. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications and enantiomeric excess values, ensuring that every batch meets the highest international standards. We are committed to translating complex academic innovations into robust industrial processes.
We invite you to collaborate with us to leverage this advanced synthetic route for your pipeline. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to support your next breakthrough in anticancer therapy development.
