Advanced Synthesis of Functionalized Dicyanoethylene Compounds for High-Performance Optoelectronics
Introduction to Next-Generation Dicyanoethylene Synthesis
The landscape of organic optoelectronic material synthesis is undergoing a significant transformation, driven by the urgent need for greener, more efficient, and scalable manufacturing processes. As detailed in the groundbreaking patent CN106966922B, a novel methodology has emerged for the production of functionalized substituted dicyanoethylene compounds, which serve as critical precursors for red and near-red light emitting materials, phthalocyanines, and porphyrins. This technology represents a paradigm shift away from hazardous traditional routes, offering a robust pathway for generating high-value intermediates essential for the electronic chemical industry. By leveraging a unique desulfurization strategy using 2,5-dinitrothiophene derivatives and triphenylphosphine, this approach addresses long-standing challenges regarding toxicity, yield variability, and environmental compliance that have plagued the sector for decades.
For R&D directors and procurement strategists alike, the implications of this innovation are profound. The ability to synthesize these complex structures without relying on volatile halogenating agents or highly toxic nitriles opens new doors for cost-effective scale-up. The patent outlines a versatile protocol that accommodates a wide array of substituents, from simple alkyl chains to complex polycyclic aromatic hydrocarbons, ensuring that manufacturers can tailor material properties for specific OLED material applications without compromising on process safety or efficiency. This report delves deep into the mechanistic advantages and commercial viability of this superior synthetic route.
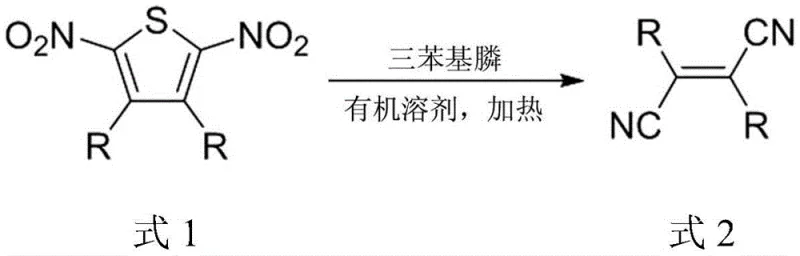
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of dicyanoethylene compounds has been fraught with significant operational and environmental hurdles. Traditional methods often rely on the oxidative coupling of phenylacetonitrile in the presence of halogens such as bromine or iodine, typically within alkaline or hydroxide aqueous solutions. These processes are notoriously sensitive to reaction conditions and substrate structure, leading to unpredictable yields that can fluctuate wildly between 20% and 80%. Furthermore, the reliance on phenylacetonitrile introduces severe safety concerns due to its high toxicity, posing risks to worker health and complicating waste disposal protocols. The use of stoichiometric amounts of halogens not only increases raw material costs but also generates substantial halogenated waste streams that require expensive treatment before discharge, thereby inflating the overall cost of production and limiting the feasibility of large-scale manufacturing.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN106966922B utilizes a completely different mechanistic pathway that circumvents these legacy issues. By employing 2,5-dinitrothiophene derivatives as the core scaffold and triphenylphosphine as the reducing agent, the reaction proceeds through a clean thermal desulfurization mechanism. This approach operates effectively at temperatures ranging from 90°C to 180°C in common organic solvents like o-dichlorobenzene or DMF. The elimination of halogenating agents and toxic nitrile starting materials drastically simplifies the safety profile of the operation. Moreover, the reaction demonstrates exceptional universality, successfully converting both aromatic and alkyl-substituted precursors into the desired dicyanoethylene products with reliable consistency. This shift not only enhances the environmental friendliness of the process but also stabilizes the supply chain by reducing dependency on regulated hazardous chemicals.
Mechanistic Insights into Triphenylphosphine-Mediated Desulfurization
The core of this technological breakthrough lies in the elegant simplicity of the triphenylphosphine-mediated desulfurization of the dinitrothiophene ring. In this transformation, the thiophene ring acts as a temporary template that holds the two substituent groups (R) in the correct cis-configuration prior to the formation of the central double bond. Upon heating with excess triphenylphosphine, the nitro groups and the sulfur atom are extruded from the ring system, likely forming triphenylphosphine oxide and other sulfur-phosphorus byproducts, while the carbon backbone collapses to form the stable fumaronitrile derivative. This mechanism is particularly advantageous because it avoids the use of transition metal catalysts in the final bond-forming step, which is a critical consideration for high-purity electronic chemical manufacturing where trace metal contamination can degrade device performance.
From an impurity control perspective, this route offers distinct advantages over oxidative coupling. Traditional halogen-based methods often leave behind difficult-to-remove halogenated byproducts or require rigorous purification to eliminate residual metal catalysts used in preceding steps. The thermal reduction method described here generates byproducts that are generally easier to separate via standard chromatographic techniques or crystallization. The use of boronic acids in the precursor synthesis (via Suzuki coupling) allows for precise control over the substitution pattern on the aromatic rings, ensuring that the final dicyanoethylene product possesses the exact electronic properties required for specific optoelectronic applications. This level of structural precision is vital for tuning the bandgap and emission characteristics of the final OLED materials.
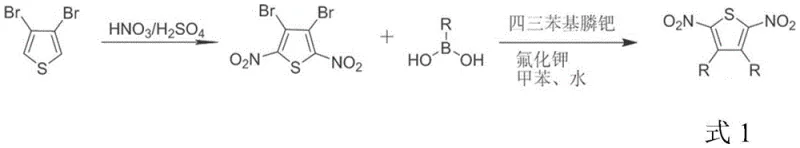
How to Synthesize Functionalized Dicyanoethylene Efficiently
The practical implementation of this synthesis involves a two-stage strategy that maximizes flexibility and yield. First, the requisite 2,5-dinitrothiophene intermediate is prepared, either through direct nitration of 3,4-disubstituted thiophenes for alkyl variants or via palladium-catalyzed Suzuki coupling of 3,4-dibromo-2,5-dinitrothiophene for aromatic variants. Once the intermediate is secured, the key transformation is executed by mixing the dinitro compound with triphenylphosphine in a molar ratio of 1:4 to 1:8. The reaction is heated in a high-boiling solvent such as o-dichlorobenzene to drive the desulfurization to completion. Detailed standardized operating procedures for scaling this reaction from gram to kilogram quantities are provided in the technical documentation below, ensuring reproducibility across different production batches.
- Prepare the 2,5-dinitrothiophene intermediate via nitration of 3,4-disubstituted thiophenes or Suzuki coupling of 3,4-dibromo-2,5-dinitrothiophene.
- Mix the dinitrothiophene intermediate with triphenylphosphine in a molar ratio of 1: 4 to 1:8 in a high-boiling organic solvent.
- Heat the reaction mixture to temperatures between 90°C and 180°C under inert atmosphere, then purify the resulting dicyanoethylene product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route translates into tangible strategic benefits that extend beyond mere chemical curiosity. The primary advantage lies in the drastic simplification of the raw material portfolio. By eliminating the need for elemental bromine, iodine, and highly toxic phenylacetonitrile, facilities can reduce their regulatory burden and insurance costs associated with handling hazardous substances. This shift also mitigates the risk of supply disruptions caused by strict environmental regulations on halogen production. Furthermore, the reaction conditions are robust and tolerant of various functional groups, meaning that a single production line can be adapted to manufacture a wide range of derivatives simply by changing the boronic acid feedstock in the precursor step, thereby enhancing asset utilization and operational flexibility.
- Cost Reduction in Manufacturing: The economic argument for this technology is compelling when analyzed through the lens of total cost of ownership. Although triphenylphosphine is a reagent of some cost, the elimination of expensive halogenating agents and the reduction in waste treatment costs associated with halogenated effluents lead to significant overall savings. Additionally, the higher consistency of yields reduces the cost per kilogram of good product by minimizing batch failures and rework. The process also avoids the need for specialized corrosion-resistant equipment often required for handling strong acids and halogens, allowing for the use of standard glass-lined or stainless steel reactors, which further lowers capital expenditure requirements for new production capacity.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available and stable starting materials. Boronic acids, used in the precursor synthesis, are commodity chemicals produced at massive scales for the pharmaceutical industry, ensuring a steady and competitive supply. Unlike niche halogenating reagents that may be subject to geopolitical supply constraints or seasonal production quotas, the feedstocks for this method are globally sourced. This diversity in sourcing options reduces the risk of single-supplier dependency. Moreover, the stability of the intermediates allows for potential stockpiling strategies, enabling manufacturers to buffer against market volatility and ensure continuous delivery to downstream clients in the fast-paced display and lighting sectors.
- Scalability and Environmental Compliance: Scaling chemical processes often exposes hidden bottlenecks related to heat transfer and waste management, but this method is inherently designed for scalability. The thermal nature of the reaction is straightforward to manage in large vessels, and the absence of exothermic halogenation steps reduces the risk of thermal runaways. From an environmental standpoint, the process aligns perfectly with modern green chemistry principles by reducing the E-factor (mass of waste per mass of product). The byproducts, primarily phosphine oxides, are less environmentally persistent than halogenated organic waste, simplifying the permitting process for facility expansion. This compliance advantage is increasingly critical as global regulations on industrial emissions tighten, future-proofing the manufacturing asset against stricter environmental mandates.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of integrating this technology into their existing workflows, we have compiled answers to the most common inquiries regarding the process parameters and material compatibility. These insights are derived directly from the experimental data and scope defined in the patent literature, providing a realistic expectation of performance. Understanding these nuances is essential for project planning and risk assessment when transitioning from legacy synthetic routes to this advanced methodology.
Q: What are the primary advantages of this synthesis method over traditional oxidative coupling?
A: This method eliminates the use of toxic halogens like bromine or iodine and avoids highly toxic phenylacetonitrile raw materials. It offers a more environmentally friendly profile with consistent yields and simpler waste treatment compared to traditional oxidative coupling methods.
Q: What is the substrate scope for the R groups in this reaction?
A: The process demonstrates broad universality, accommodating both aromatic ring substituents (such as phenyl, naphthyl, and various substituted benzenes) and alkane substituents (like hexyl chains), making it versatile for diverse optoelectronic applications.
Q: How does this method impact the purity required for electronic grade materials?
A: By avoiding transition metal catalysts in the final desulfurization step and utilizing a clean thermal reduction with triphenylphosphine, the method minimizes metal impurities, which is critical for achieving the high purity standards demanded by OLED and semiconductor manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dicyanoethylene Compounds Supplier
As the demand for high-performance organic semiconductors continues to surge, partnering with a manufacturer who possesses deep technical expertise in advanced synthetic methodologies is crucial for maintaining a competitive edge. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging cutting-edge processes like the triphenylphosphine-mediated desulfurization to deliver superior quality intermediates. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements without compromising on the stringent purity specifications required for electronic applications. Our rigorous QC labs employ state-of-the-art analytical techniques to verify the absence of metal impurities and halogenated residues, guaranteeing material consistency for your OLED or photovoltaic projects.
We invite you to collaborate with us to optimize your supply chain for next-generation optoelectronic materials. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume needs and target specifications. We encourage potential partners to reach out for specific COA data and route feasibility assessments to understand how our advanced manufacturing capabilities can accelerate your product development timelines. Let us help you navigate the complexities of fine chemical synthesis and secure a reliable supply of high-quality dicyanoethylene derivatives for your future innovations.
