Advanced TCCA-Mediated Oxidation for Commercial Scale-Up of Complex Opioid Intermediates
The pharmaceutical industry continuously seeks robust synthetic pathways for critical opioid intermediates, and patent CN1918168B introduces a transformative approach to preparing 3-O-protected morphinones and their dienol carboxylate derivatives. This technology addresses long-standing challenges in the oxidation of secondary alcohols within complex morphine alkaloid scaffolds, offering a viable alternative to traditional methods that often suffer from harsh conditions or problematic by-products. By leveraging trichloroisocyanuric acid (TCCA) in conjunction with specific sulfide compounds, the disclosed process achieves efficient conversion of 3-O-protected morphines into valuable ketone intermediates under remarkably mild conditions. For a reliable pharmaceutical intermediate supplier, adopting such methodologies signifies a commitment to process safety and environmental compliance while maintaining high chemical fidelity. The strategic implementation of this oxidation protocol allows manufacturers to bypass the limitations of cryogenic temperatures and noxious emissions typically associated with legacy oxidation technologies, thereby streamlining the production of analgesic precursors like codeinone and its derivatives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the oxidation of morphine alkaloids to their corresponding ketones has relied heavily on methods such as the Swern oxidation or manganese dioxide mediated processes, both of which present significant operational hurdles for large-scale manufacturing. The Swern oxidation, while effective in laboratory settings, necessitates the use of dimethyl sulfoxide (DMSO) and oxalyl chloride at cryogenic temperatures, often below -60°C, which imposes substantial energy costs and equipment requirements on production facilities. Furthermore, a critical drawback of the Swern protocol is the generation of dimethyl sulfide, a volatile compound with an extremely offensive odor that requires specialized scrubbing systems and rigorous containment measures to protect worker safety and community relations. Alternative methods using manganese dioxide often suffer from inconsistent reactivity, requiring large excesses of the oxidant and generating significant solid waste that complicates downstream processing and filtration steps. These conventional techniques frequently result in variable yields and purity profiles, creating bottlenecks in the supply chain for high-purity opioid intermediates and increasing the overall cost of goods sold for finished pharmaceutical products.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a combination of trichloroisocyanuric acid and organic sulfides to effect oxidation under much more manageable thermal conditions, typically ranging from -40°C to 50°C. This methodology eliminates the need for extreme cryogenic cooling, allowing reactions to proceed at temperatures that are easily maintainable with standard industrial chilling systems, thus drastically simplifying the engineering controls required for commercial production. The use of TCCA, a stable solid chlorinating agent, provides a consistent source of active chlorine that reacts predictably with the sulfide to generate the active oxidizing species in situ, avoiding the handling hazards associated with gaseous chlorine or unstable liquid reagents. Moreover, this system is designed to minimize the formation of malodorous by-products, significantly improving the working environment and reducing the burden on waste treatment infrastructure. By enabling the direct conversion of protected morphines to ketones with high selectivity, this approach supports cost reduction in opioid manufacturing by reducing reagent consumption and simplifying the purification workflow required to meet stringent pharmacopeial standards.
Mechanistic Insights into TCCA-Sulfide Mediated Oxidation
The core of this technological advancement lies in the generation of a reactive sulfonium cation intermediate, which serves as the primary oxidizing agent for the secondary alcohol functionality on the morphine scaffold. As illustrated in the mechanistic data, the reaction initiates when the chlorine-containing reagent, specifically trichloroisocyanuric acid, interacts with the sulfide compound (R1SR2) to form a chlorosulfonium species. This electrophilic sulfur center then attacks the hydroxyl group of the 3-O-protected morphine, forming an alkoxysulfonium ion that is primed for elimination. The presence of a base, such as triethylamine or diisopropylethylamine, facilitates the deprotonation of the alpha-carbon, driving the collapse of the intermediate to release the desired ketone and a reduced sulfur by-product. This mechanism is highly advantageous because it proceeds through a well-defined pathway that minimizes side reactions such as over-oxidation or degradation of the sensitive morphinan skeleton. Understanding this catalytic cycle is crucial for R&D teams aiming to optimize reaction parameters, as the stoichiometry of the sulfide and the choice of base can be tuned to maximize conversion efficiency while maintaining the integrity of the chiral centers essential for biological activity.
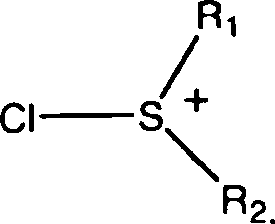
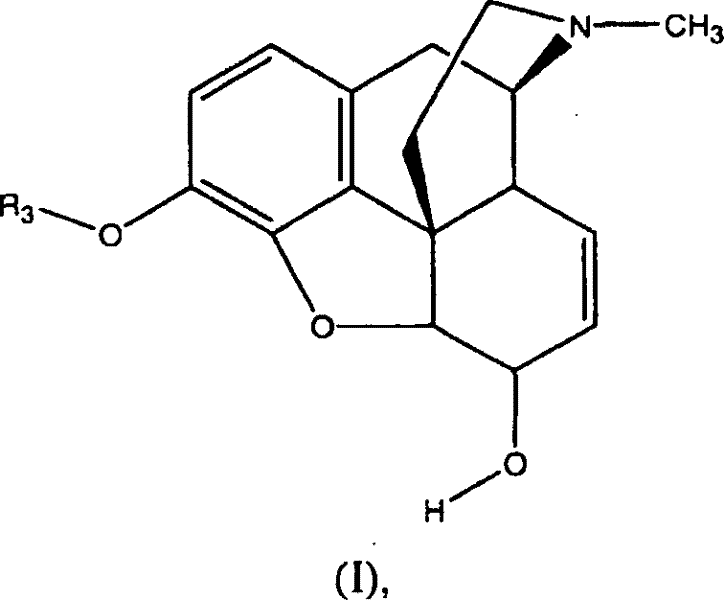
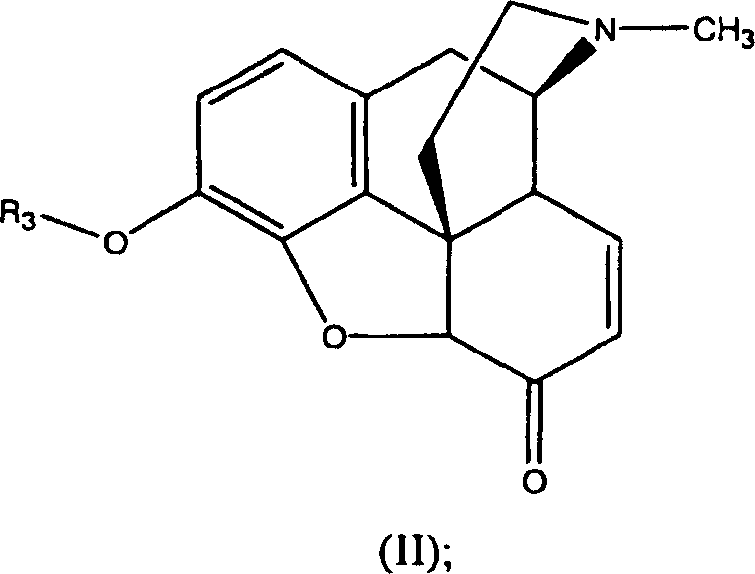
Controlling impurity profiles is paramount in the synthesis of pharmaceutical intermediates, and this oxidation method offers distinct advantages in managing by-product formation compared to traditional routes. The specific selection of the R1 and R2 groups on the sulfide compound allows chemists to modulate the reactivity and solubility of the oxidizing species, ensuring that the reaction proceeds cleanly without generating complex mixtures that are difficult to separate. For instance, using sulfides with longer alkyl chains can reduce volatility and odor, further enhancing the safety profile of the process. Additionally, the reaction conditions are compatible with a wide range of protecting groups at the 3-O position, including silyl ethers and carbonates, providing flexibility in synthetic design for downstream derivatization. The ability to conduct the reaction in common organic solvents like dichloromethane or toluene ensures that workup procedures remain straightforward, typically involving aqueous washes to remove inorganic salts followed by crystallization or chromatography. This level of control over the chemical environment ensures that the final 3-O-protected morphinone meets the rigorous purity specifications required for subsequent transformation into active pharmaceutical ingredients like naloxone or oxycodone.
How to Synthesize 3-O-Protected Morphinones Efficiently
Implementing this synthesis route requires careful attention to reagent quality and process parameters to ensure consistent results across different batch sizes. The general procedure involves preparing a mixture of the sulfide compound and trichloroisocyanuric acid in an anhydrous solvent, cooling the solution to the specified temperature range, and then introducing the 3-O-protected morphine substrate. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction mixture by combining trichloroisocyanuric acid and a sulfide compound (R1SR2) in an anhydrous organic solvent such as dichloromethane under inert atmosphere.
- Add the 3-O-protected morphine substrate (Formula I) to the cooled mixture, maintaining temperatures between -40°C and 50°C to control exothermic activity.
- Introduce an organic base such as triethylamine to facilitate the formation of the sulfonium cation intermediate and drive the oxidation to the ketone (Formula II).
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this TCCA-mediated oxidation technology offers substantial strategic benefits that extend beyond mere chemical efficiency. The shift away from cryogenic-dependent processes reduces the energy footprint of manufacturing operations, leading to significant cost savings in utilities and infrastructure maintenance over the lifecycle of the product. Furthermore, the stability of trichloroisocyanuric acid simplifies inventory management and logistics, as it does not require the specialized storage conditions associated with unstable liquid oxidants or hazardous gases. This reliability enhances supply chain continuity by minimizing the risk of production delays caused by reagent degradation or delivery complications, ensuring that critical opioid intermediates are available to meet market demand without interruption. For procurement managers, this translates into a more predictable cost structure and reduced exposure to volatile raw material markets, allowing for better long-term planning and budget allocation.
- Cost Reduction in Manufacturing: The elimination of expensive cryogenic cooling systems and the reduction in waste treatment costs associated with malodorous by-products contribute to a leaner manufacturing model. By utilizing stable, solid reagents that are easily handled and dosed, facilities can reduce labor costs related to hazardous material handling and minimize the need for complex engineering controls. The high conversion efficiency of the reaction means that less starting material is wasted, improving the overall atom economy and reducing the cost per kilogram of the final intermediate. These factors combine to create a more economically viable production process that can withstand competitive pricing pressures in the global pharmaceutical market.
- Enhanced Supply Chain Reliability: The use of commercially available and stable reagents like TCCA ensures that the supply chain is less vulnerable to disruptions caused by the scarcity of specialized chemicals. Since the process does not rely on custom-synthesized catalysts or rare metals, sourcing is straightforward and can be diversified across multiple vendors to mitigate risk. The robustness of the reaction conditions also means that the process is more forgiving of minor variations in raw material quality, reducing the rate of batch failures and ensuring a steady flow of product to downstream customers. This reliability is critical for maintaining trust with partners who depend on timely delivery of high-quality intermediates for their own production schedules.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the absence of extreme operating conditions and the use of standard unit operations familiar to chemical engineers. The reduced generation of hazardous waste and odorous emissions simplifies regulatory compliance and permitting, allowing facilities to operate with a smaller environmental footprint. This alignment with green chemistry principles not only meets current regulatory standards but also future-proofs the manufacturing site against increasingly stringent environmental laws. The ability to scale efficiently ensures that supply can be ramped up quickly to meet surges in demand without compromising on quality or safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this oxidation technology in pharmaceutical manufacturing. These answers are derived directly from the patent specifications and are intended to provide clarity on the operational benefits and chemical scope of the method. Understanding these details helps stakeholders make informed decisions about integrating this process into their existing production workflows.
Q: How does this TCCA method improve upon traditional Swern oxidation?
A: Unlike Swern oxidation which generates malodorous dimethyl sulfide by-products requiring complex scrubbing systems, this TCCA-mediated process utilizes stable reagents that significantly reduce odor and simplify waste management protocols.
Q: What are the scalability advantages of using trichloroisocyanuric acid?
A: Trichloroisocyanuric acid is a solid, stable chlorinating agent that allows for precise dosing and safer handling compared to gaseous chlorine or unstable liquid oxidants, facilitating safer commercial scale-up of complex opioid intermediates.
Q: Can this method be adapted for one-pot synthesis of dienol carboxylates?
A: Yes, the patent discloses embodiments where the intermediate ketone is not isolated but directly reacted with acylating agents in the same vessel, reducing lead time for high-purity pharmaceutical intermediates and minimizing material loss.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-O-Protected Morphinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in the production of high-value pharmaceutical intermediates. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the TCCA-mediated oxidation are successfully translated into efficient manufacturing processes. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 3-O-protected morphinone meets the exacting standards required by global regulatory bodies. Our commitment to quality and consistency makes us a trusted partner for companies seeking to secure their supply of complex opioid intermediates.
We invite you to collaborate with us to explore how this advanced oxidation technology can optimize your production costs and enhance your supply chain resilience. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities align with your strategic goals for reliable pharmaceutical intermediate supplier partnerships.
