Efficient CuBr-Catalyzed Synthesis of Trans-Styryl Benzofuranones for Commercial Scale-Up
The landscape of fine chemical manufacturing is constantly evolving, driven by the need for more sustainable and cost-effective synthetic routes for complex organic molecules. A significant breakthrough in this domain is detailed in patent CN114835664A, which discloses a novel and efficient synthesis method for trans-styryl benzofuranone compounds. These compounds serve as critical scaffolds in the development of advanced pharmaceutical agents and agrochemical active ingredients, owing to their unique biological activity and physicochemical properties. The patented methodology leverages a copper-catalyzed alkenylation strategy that transforms simple benzofuranone and styrene derivatives into high-value olefinic products with exceptional efficiency. By utilizing a CuBr catalyst system coupled with elemental iodine as an oxidant, this process circumvents the traditional reliance on precious metal catalysts, offering a greener and more economically viable pathway for industrial application. For R&D directors and procurement specialists alike, this innovation represents a pivotal shift towards reducing the cost of goods sold (COGS) while maintaining rigorous purity standards required for regulatory compliance in the life sciences sector.
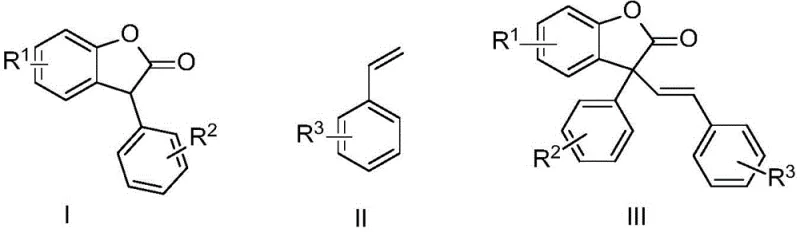
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of complex olefinic frameworks, such as those found in trans-styryl benzofuranones, has relied heavily on classical cross-coupling reactions like the Heck reaction or condensation methods like the Wittig reaction. While these methods are well-established in academic literature, they present substantial bottlenecks when translated to large-scale commercial manufacturing. The Heck reaction, for instance, typically necessitates the use of palladium-based catalysts, which are not only prohibitively expensive due to the scarcity of the metal but also pose significant challenges regarding residual metal removal in final drug substances. Furthermore, conventional methods often suffer from poor stereoselectivity, yielding mixtures of cis and trans isomers that require energy-intensive and yield-reducing separation processes. The harsh reaction conditions associated with Wittig reactions, often involving strong bases and sensitive phosphorus ylides, further complicate the operational safety profile and increase waste generation, making them less attractive for modern green chemistry initiatives aimed at minimizing environmental impact.
The Novel Approach
In stark contrast to these legacy technologies, the novel approach outlined in the patent data introduces a streamlined, one-pot synthesis that utilizes earth-abundant copper salts instead of precious metals. This method employs benzofuranone derivatives and styrene derivatives as direct starting materials, reacting them in the presence of CuBr and elemental iodine under a nitrogen atmosphere. The reaction conditions are remarkably mild, typically proceeding at temperatures between 80°C and 150°C, with optimal results observed around 120°C. This transition to a copper-catalyzed system not only drastically lowers the catalyst cost but also simplifies the reaction setup by eliminating the need for inert atmosphere gloveboxes or specialized high-pressure equipment often required for other coupling strategies. The process demonstrates high functional group tolerance, accommodating various substituents on both the benzofuranone and styrene rings, which allows for the rapid generation of diverse compound libraries for structure-activity relationship (SAR) studies without the need for extensive protecting group chemistry.
Mechanistic Insights into CuBr-Catalyzed Radical Alkenylation
Understanding the mechanistic underpinnings of this transformation is crucial for R&D teams aiming to optimize the process for specific substrates. The reaction proceeds through a radical-mediated pathway initiated by the copper catalyst and the iodine oxidant. As illustrated in the mechanistic diagram, the CuBr catalyst facilitates the generation of radical species from the benzofuranone substrate at the C3 position. This carbon-centered radical then undergoes addition to the double bond of the styrene derivative. The subsequent steps involve oxidation and elimination processes mediated by the iodine species, ultimately restoring aromaticity and forming the new carbon-carbon double bond with high trans-selectivity. This radical mechanism is distinct from the two-electron processes seen in palladium catalysis, offering a complementary reactivity profile that can access chemical space difficult to reach via traditional ionic pathways. The use of elemental iodine serves a dual purpose: it acts as a terminal oxidant to regenerate the active copper species and participates in the radical propagation cycle, ensuring the reaction proceeds to completion with high atom economy.
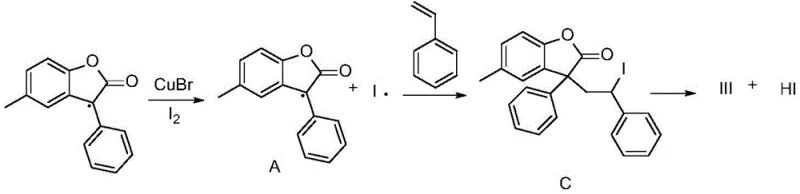
From an impurity control perspective, this mechanism offers significant advantages for pharmaceutical manufacturing. The high stereoselectivity for the trans-isomer is inherent to the thermodynamic stability of the transition state in this radical addition-elimination sequence. This means that the formation of the undesired cis-isomer is minimized at the source, rather than requiring downstream correction. For quality control laboratories, this translates to a cleaner crude reaction profile, reducing the burden on purification units such as preparative HPLC or recrystallization steps. Furthermore, the absence of phosphine ligands, which are common in palladium chemistry and can be difficult to remove completely, ensures that the final product meets stringent limits for genotoxic impurities and heavy metal residues. The robustness of the radical mechanism against moisture and oxygen (within the controlled nitrogen environment) also contributes to batch-to-batch consistency, a critical parameter for validating commercial manufacturing processes.
How to Synthesize Trans-Styryl Benzofuranone Efficiently
The practical execution of this synthesis is designed for operational simplicity, making it accessible for both laboratory-scale discovery and pilot-plant production. The general protocol involves charging a reaction vessel with the benzofuranone derivative, the styrene derivative, the CuBr catalyst, elemental iodine, and a base such as disodium hydrogen phosphate in a polar solvent like acetonitrile. The mixture is then heated to the target temperature, typically 120°C, and stirred for a duration of 6 to 18 hours, with 12 hours being the preferred timeframe for maximum conversion. Following the reaction, the workup is straightforward, often requiring only standard extraction and column chromatography to isolate the pure white powder product. This simplicity reduces the training burden on operators and minimizes the risk of human error during scale-up. For detailed standard operating procedures and specific stoichiometric ratios tailored to your specific substrate variants, please refer to the standardized synthesis steps provided below.
- Charge a reaction vessel with benzofuranone derivative, styrene derivative, CuBr catalyst, elemental iodine oxidant, and base in acetonitrile solvent.
- Heat the reaction mixture to 120°C under a nitrogen atmosphere and stir continuously for approximately 12 hours to ensure complete conversion.
- Upon completion, purify the crude reaction mixture via column chromatography to isolate the high-purity trans-styryl benzofuranone target product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this CuBr-catalyzed methodology offers tangible strategic benefits that extend beyond mere technical feasibility. The shift from precious metal catalysts to base metal catalysts fundamentally alters the cost structure of the synthesis, removing exposure to the volatile pricing of palladium and rhodium. Additionally, the use of commodity chemicals like styrene and simple benzofuranones as starting materials ensures a stable and diversified supply base, reducing the risk of single-source bottlenecks. The operational simplicity of the one-pot process also implies a reduced footprint in the manufacturing facility, allowing for higher throughput in existing reactor trains without the need for capital-intensive retrofitting. These factors collectively contribute to a more resilient supply chain capable of meeting the demanding delivery schedules of the global pharmaceutical market.
- Cost Reduction in Manufacturing: The replacement of expensive palladium catalysts with inexpensive copper bromide results in substantial cost savings on raw materials. Since the catalyst loading is optimized and the oxidant (iodine) is cheap and readily available, the overall variable cost per kilogram of product is significantly lowered. Furthermore, the high yield and selectivity reduce the amount of starting material wasted, improving the overall mass balance and reducing the cost associated with raw material procurement and waste disposal.
- Enhanced Supply Chain Reliability: The reliance on widely available bulk chemicals like styrene and acetonitrile mitigates supply risks associated with specialized reagents. Unlike complex ligands or sensitive organometallic reagents that may have long lead times or limited suppliers, the inputs for this process are commodity items with robust global supply chains. This ensures that production schedules can be maintained consistently, even during periods of market volatility, providing a reliable source of high-purity pharmaceutical intermediates for downstream drug substance manufacturing.
- Scalability and Environmental Compliance: The process operates under relatively mild thermal conditions and uses standard solvents, facilitating easy scale-up from gram to ton quantities. The absence of toxic phosphorus byproducts and heavy metal residues simplifies the environmental compliance profile, reducing the cost and complexity of wastewater treatment and hazardous waste management. This alignment with green chemistry principles not only lowers operational costs but also enhances the sustainability credentials of the final product, which is increasingly important for corporate social responsibility reporting.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear understanding of the method's capabilities and limitations. Understanding these details is essential for project managers evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Why is the CuBr/I2 system preferred over traditional Palladium catalysts for this synthesis?
A: The CuBr/I2 system significantly reduces raw material costs by eliminating the need for expensive palladium catalysts. Furthermore, it operates under milder conditions with excellent stereoselectivity for the trans-isomer, simplifying downstream purification.
Q: What represents the primary advantage of this one-pot method for supply chain reliability?
A: The one-pot nature of the reaction minimizes unit operations and handling time. Combined with the use of readily available starting materials like styrene and benzofuranone, this ensures a robust and continuous supply chain with reduced lead times.
Q: Is this synthesis method scalable for industrial production of pharmaceutical intermediates?
A: Yes, the method utilizes standard solvents like acetonitrile and common heating protocols (80-150°C), making it highly amenable to scale-up from laboratory grams to multi-ton commercial production without complex equipment requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trans-Styryl Benzofuranone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in the development of next-generation therapeutics. Our team of expert chemists has thoroughly analyzed the potential of the CuBr-catalyzed alkenylation method described in CN114835664A and is prepared to leverage this technology for your specific project needs. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of trans-styryl benzofuranone intermediate delivered meets the highest international standards for pharmaceutical applications.
We invite you to collaborate with us to unlock the full potential of this cost-effective synthesis platform. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. We encourage you to reach out today to discuss your project specifics,索取 specific COA data for our reference standards, and obtain comprehensive route feasibility assessments that will accelerate your drug development pipeline. Let us be your trusted partner in delivering high-quality chemical solutions that drive innovation and efficiency in the global healthcare industry.
