Advanced One-Step Synthesis of 2,3-Dihydrospiro[imidazole-4,1'-indene] Derivatives for Commercial Scale-up
The pharmaceutical and fine chemical industries are constantly seeking efficient routes to complex nitrogen-containing heterocycles, particularly spirocyclic scaffolds which are prevalent in bioactive molecules. Patent CN111732545B discloses a groundbreaking methodology for the synthesis of 2,3-dihydrospiro[imidazole-4,1'-indene] compounds, addressing a significant gap in current organic synthesis capabilities. Traditionally, constructing such spiro-frameworks has been challenging due to the lack of direct methods to functionalize the imidazole core without extensive protecting group manipulations. This invention leverages the unique reactivity of the C=N bond within 2H-imidazole derivatives, treating it as a carbon-electrophile to drive an intramolecular cyclization. ![General structural formula of 2,3-dihydrospiro[imidazole-4,1'-indene] compounds showing variable R groups](/insights/img/spiro-imidazole-synthesis-pharma-supplier-20260303150058-01.png) This strategic approach allows for the incorporation of directing groups directly into the target product in a single operational step. For R&D directors and process chemists, this represents a paradigm shift from laborious multi-step sequences to a streamlined, atom-economical process. The structural versatility is evident, with substituents R1 through R5 accommodating halogens, alkyls, trifluoromethyl groups, and various ester functionalities, making this platform highly adaptable for generating diverse libraries of potential drug candidates or functional materials.
This strategic approach allows for the incorporation of directing groups directly into the target product in a single operational step. For R&D directors and process chemists, this represents a paradigm shift from laborious multi-step sequences to a streamlined, atom-economical process. The structural versatility is evident, with substituents R1 through R5 accommodating halogens, alkyls, trifluoromethyl groups, and various ester functionalities, making this platform highly adaptable for generating diverse libraries of potential drug candidates or functional materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of related spirocyclic structures, specifically 2',3'-dihydrospiro[imidazolidine-4,1'-indene]-2,5-dione derivatives, was fraught with inefficiencies. Conventional routes typically necessitated stepwise construction, involving multiple isolation and purification stages that inherently degraded overall throughput. These legacy methods often suffered from low reaction selectivity, leading to complex mixtures of regioisomers that were difficult and costly to separate on a large scale. Furthermore, the harsh conditions sometimes required for traditional cyclizations could compromise sensitive functional groups, limiting the substrate scope and requiring additional protection-deprotection cycles. From a supply chain perspective, these inefficiencies translate to longer lead times, higher waste generation, and increased raw material consumption, creating bottlenecks for the reliable production of high-purity pharmaceutical intermediates needed for clinical trials and commercial manufacturing.
The Novel Approach
The methodology described in CN111732545B offers a robust solution by enabling a direct, one-pot synthesis of the target 2,3-dihydrospiro[imidazole-4,1'-indene] skeleton. By utilizing 2H-imidazole derivatives and 2-alkynoate esters as starting materials, the process bypasses the need for pre-functionalized precursors. 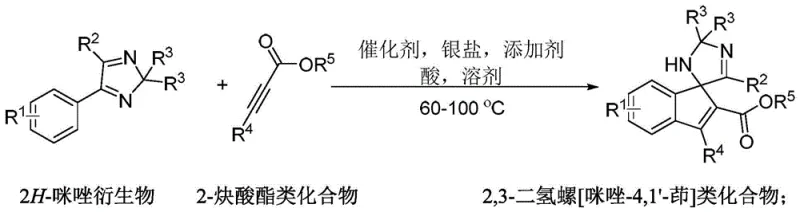 The reaction proceeds via a ruthenium-catalyzed mechanism that activates the C=N bond, facilitating a smooth cyclization under relatively mild thermal conditions (60-100°C). This novel approach not only drastically reduces the number of unit operations but also demonstrates excellent regioselectivity, ensuring that the desired spiro-center is formed exclusively. The ability to tolerate air atmosphere during the reaction further distinguishes this method from many transition-metal catalyzed processes that require rigorous exclusion of oxygen and moisture, thereby simplifying the engineering requirements for scale-up and reducing the operational burden on manufacturing teams.
The reaction proceeds via a ruthenium-catalyzed mechanism that activates the C=N bond, facilitating a smooth cyclization under relatively mild thermal conditions (60-100°C). This novel approach not only drastically reduces the number of unit operations but also demonstrates excellent regioselectivity, ensuring that the desired spiro-center is formed exclusively. The ability to tolerate air atmosphere during the reaction further distinguishes this method from many transition-metal catalyzed processes that require rigorous exclusion of oxygen and moisture, thereby simplifying the engineering requirements for scale-up and reducing the operational burden on manufacturing teams.
Mechanistic Insights into Ru-Catalyzed Cyclization
The core of this transformation lies in the synergistic action of the ruthenium catalyst and the silver/additive system. The catalyst, specifically dichlorobis(4-methylisopropylphenyl)ruthenium(II) dimer, acts as the primary activator for the C-H bond adjacent to the nitrogen, while the silver salt (preferably silver bistrifluoromethanesulfonimide) assists in halide abstraction to generate the active cationic ruthenium species. The addition of zinc trifluoromethanesulfonate as an additive plays a critical role in modulating the Lewis acidity of the system, enhancing the electrophilicity of the alkyne moiety in the 2-alkynoate. This precise tuning of the electronic environment ensures that the nucleophilic attack occurs selectively at the intended position on the imidazole ring. Understanding this mechanistic nuance is vital for process optimization, as it explains why specific combinations of acids and solvents yield superior results compared to others.
Regarding impurity control, the high specificity of this catalytic cycle minimizes the formation of side products such as linear addition adducts or polymerization of the alkyne. The use of benzoic acid as a proton source helps to regenerate the active catalyst and stabilize the intermediate species, preventing catalyst deactivation which is a common issue in prolonged heating cycles. Experimental data indicates that deviations from the optimal additive profile, such as using nickel or copper triflates instead of zinc, result in noticeably lower yields (dropping from roughly 60-70% down to 40-50%). This sensitivity underscores the importance of strict adherence to the optimized reagent grades and ratios to maintain the stringent purity specifications required for pharmaceutical applications, ensuring that the final API intermediate meets regulatory standards without extensive downstream purification.
How to Synthesize 2,3-Dihydrospiro[imidazole-4,1'-indene] Efficiently
To implement this synthesis effectively, one must carefully balance the stoichiometry of the reactants and the catalytic loading. The patent specifies a molar ratio of 1:1.5 for the imidazole derivative to the alkynoate, ensuring complete conversion of the more valuable heterocyclic starting material. The reaction is typically conducted in 1,2-dichloroethane (DCE) at 100°C for 12 hours, although temperatures ranging from 60°C to 100°C are viable depending on the specific substrate reactivity. Detailed standardized synthetic steps see the guide below.
- Mix 2H-imidazole derivative, 2-alkynoate ester, [RuCl2(p-cymene)]2 catalyst, silver salt additive, zinc triflate, benzoic acid, and 1,2-dichloroethane solvent.
- Heat the reaction mixture to 100°C under air atmosphere and stir for 12 hours to facilitate the intramolecular cyclization.
- Cool to room temperature, quench with saturated NaHCO3, extract with ethyl acetate, dry over sodium sulfate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology offers tangible benefits beyond mere chemical elegance. The primary advantage lies in the significant simplification of the manufacturing workflow. By consolidating what was previously a multi-step sequence into a single pot, the process inherently reduces labor costs, energy consumption, and solvent usage. The raw materials, including various substituted 2H-imidazoles and alkynoates, are commercially available or easily synthesized from commodity chemicals, ensuring a stable and resilient supply base. This accessibility mitigates the risk of raw material shortages that often plague complex custom synthesis projects, allowing for more accurate forecasting and inventory management.
- Cost Reduction in Manufacturing: The elimination of intermediate isolation steps translates directly into substantial cost savings. In traditional synthesis, each isolation involves filtration, drying, and quality control testing, all of which add time and expense. This one-pot method removes these overheads entirely. Furthermore, the reaction tolerates air, removing the capital expenditure associated with maintaining inert atmosphere reactors (nitrogen/argon lines and monitoring systems). The use of earth-abundant additives like zinc triflate instead of more exotic metals also contributes to a lower bill of materials, driving down the overall cost of goods sold (COGS) for the final intermediate.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions—specifically the tolerance to air and the use of standard solvents like DCE or trifluoroethanol—makes this process highly transferable between different manufacturing sites. This flexibility is crucial for supply chain continuity, as it allows for dual-sourcing or rapid technology transfer to CDMO partners without the need for specialized equipment retrofitting. The high regioselectivity ensures consistent product quality batch-to-batch, reducing the variability that often leads to supply disruptions caused by failed quality releases.
- Scalability and Environmental Compliance: Scaling this reaction from gram to kilogram scale is straightforward due to the absence of hazardous reagents or extreme pressure conditions. The workup procedure involves a simple aqueous wash and extraction, which generates less hazardous waste compared to processes requiring heavy metal scavengers or complex chromatographic separations at early stages. This aligns well with modern green chemistry initiatives and environmental regulations, reducing the burden on waste treatment facilities and lowering the environmental compliance costs associated with production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this spiro-imidazole synthesis platform. These answers are derived directly from the experimental data and optimization studies presented in the patent documentation, providing a clear picture of the process capabilities and limitations for potential partners.
Q: What is the key advantage of this Ru-catalyzed method over traditional spiro-compound synthesis?
A: Unlike traditional multi-step syntheses of spiro-imidazolidine-diones which suffer from low yields and poor selectivity, this novel method utilizes the C=N bond of 2H-imidazoles as an electrophile for a direct, one-step cyclization with high regioselectivity and yields up to 82%.
Q: Is the reaction sensitive to atmospheric conditions?
A: No, the process is robust and can be conducted efficiently under air atmosphere, eliminating the need for costly inert gas protection systems typically required for sensitive organometallic catalysis, which simplifies operational complexity.
Q: What represents the optimal catalytic system for this transformation?
A: The preferred system employs [RuCl2(p-cymene)]2 as the catalyst, silver bistrifluoromethanesulfonimide as the silver salt, zinc trifluoromethanesulfonate as the additive, and benzoic acid as the promoter in 1,2-dichloroethane at 100°C.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3-Dihydrospiro[imidazole-4,1'-indene] Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this Ru-catalyzed cyclization technology for developing next-generation therapeutics. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can seamlessly transition from laboratory discovery to full-scale manufacturing. Our facility is equipped with state-of-the-art rigorous QC labs capable of verifying the structural integrity and purity of these complex spiro-scaffolds, adhering to stringent purity specifications required by global regulatory bodies. We understand that time-to-market is critical, and our team is ready to deploy this efficient synthesis route to accelerate your development timelines.
We invite you to collaborate with us to leverage this advanced chemistry for your specific pipeline needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data from pilot runs and comprehensive route feasibility assessments to demonstrate how this technology can optimize your supply chain and reduce overall project costs. Let us be your partner in turning this innovative patent into a commercial reality.
