Revolutionizing o-Diamine Production: A Metal-Free Strategy for Scalable Pharmaceutical Intermediates
The landscape of organic synthesis for pharmaceutical intermediates is constantly evolving, driven by the need for greener, more cost-effective, and scalable methodologies. A significant breakthrough in this domain is documented in patent CN114957016B, which discloses a novel method for synthesizing o-diamine compounds without the reliance on transition metal catalysts. This innovation addresses a critical bottleneck in the production of bioactive molecules, where traditional routes often suffer from high costs associated with precious metal catalysts and complex purification steps required to remove metal residues. By utilizing alkenylsulfonium salts as key electrophilic intermediates reacting with primary amines, this technology offers a streamlined pathway to access structurally diverse ortho-diamines. For R&D directors and procurement managers in the fine chemical industry, this represents a pivotal shift towards more sustainable manufacturing processes that maintain high purity standards while significantly reducing the environmental footprint and operational complexity of synthetic routes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the vicinal diamine motif, a core structural element found in numerous drugs such as dexrazoxane and bepridil, has relied heavily on transition metal catalysis or the use of hazardous reagents. Conventional strategies often necessitate the use of expensive palladium, copper, or rhodium catalysts, which not only inflate the raw material costs but also introduce significant supply chain vulnerabilities due to the fluctuating market prices of these precious metals. Furthermore, the presence of metal residues in the final active pharmaceutical ingredient (API) is strictly regulated, requiring rigorous and costly purification protocols such as scavenging or recrystallization to meet ppm-level specifications. Additionally, many traditional methods require specialized amine sources or harsh reaction conditions that limit the functional group tolerance, thereby restricting the chemical space accessible for drug discovery and process optimization. These factors collectively contribute to longer lead times and higher overall manufacturing costs for pharmaceutical intermediates.
The Novel Approach
In stark contrast, the methodology outlined in the patent introduces a metal-free catalytic system that leverages the unique reactivity of alkenylsulfonium salts. This approach fundamentally changes the economic and operational dynamics of o-diamine synthesis by eliminating the need for any transition metal catalyst. The reaction proceeds efficiently under mild conditions, typically at room temperature and under an air atmosphere, which drastically reduces energy consumption and the need for inert gas protection systems. The use of commercially available primary amines and easily preparable sulfonium salts ensures that the starting materials are both cheap and readily accessible, mitigating supply chain risks. Moreover, the reaction exhibits excellent functional group compatibility, allowing for the introduction of various substituents such as halogens and alkyl groups without compromising yield. This robustness makes the process highly attractive for the commercial scale-up of complex pharmaceutical intermediates, offering a cleaner and more direct route to high-value chemical building blocks.
Mechanistic Insights into Metal-Free Diamination via Sulfonium Salts
The core of this innovative synthesis lies in the generation and utilization of alkenylsulfonium salts as potent electrophiles. As illustrated in the general reaction scheme below, the process initiates with the formation of the sulfonium salt from a styrene derivative, tetramethylene sulfoxide, and trifluoromethanesulfonic anhydride. This activation step converts the relatively inert alkene into a highly reactive species capable of undergoing nucleophilic attack. The subsequent reaction with a primary amine involves a nucleophilic addition-elimination sequence or a concerted mechanism depending on the specific substrate electronics, ultimately leading to the formation of the C-N bonds characteristic of the o-diamine structure. The absence of a metal center means that the reaction mechanism is driven purely by organic electronic effects and the stability of the sulfonium leaving group, which simplifies the mechanistic landscape and reduces the likelihood of side reactions often associated with metal coordination complexes.
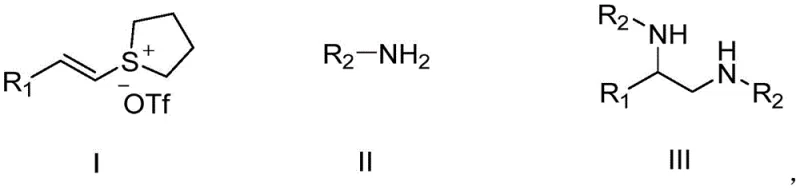
From a quality control perspective, the metal-free nature of this mechanism provides a distinct advantage in impurity profiling. Without transition metals, there is no risk of metal-catalyzed homocoupling or other metallation side products that can be difficult to separate from the desired product. The primary impurities are likely to be unreacted starting materials or simple organic byproducts that can be easily removed via standard silica gel column chromatography or crystallization. The patent data indicates that additives such as potassium bromide play a crucial role in enhancing the reaction efficiency, possibly by stabilizing intermediates or facilitating the leaving group ability of the sulfonium moiety. Understanding these mechanistic nuances allows process chemists to fine-tune reaction parameters, such as base selection and solvent choice, to maximize yield and purity, ensuring that the final product meets the stringent specifications required for pharmaceutical applications.
How to Synthesize o-Diamine Compounds Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to the preparation of the key alkenylsulfonium salt intermediate and the optimization of the diamination conditions. The process is designed to be operationally simple, avoiding the need for gloveboxes or specialized high-pressure equipment. Detailed standardized synthetic procedures, including specific molar ratios, solvent volumes, and workup protocols, are essential for reproducibility and successful technology transfer. The following guide outlines the critical stages involved in executing this transformation effectively, ensuring that R&D teams can rapidly evaluate the feasibility of this route for their specific target molecules.
- Preparation of the alkenylsulfonium salt precursor by reacting aryl olefins with tetramethylene sulfoxide and trifluoromethanesulfonic anhydride at low temperature.
- Combining the alkenylsulfonium salt with a primary amine substrate in an organic solvent such as toluene under air atmosphere.
- Adding a base like potassium tert-butoxide and an additive such as potassium bromide, then stirring at room temperature for 12 to 24 hours to complete the diamination.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this metal-free synthesis technology translates into tangible strategic benefits that extend beyond mere chemical efficiency. The elimination of precious metal catalysts directly impacts the bill of materials, removing a significant cost driver that is subject to volatile global market trends. Furthermore, the reliance on commodity chemicals like styrenes, sulfoxides, and simple amines ensures a robust and diversified supply base, reducing the risk of production stoppages due to raw material shortages. The mild reaction conditions also imply lower energy costs and reduced wear and tear on reactor equipment, contributing to overall operational expenditure savings. These factors combined create a compelling business case for integrating this methodology into the manufacturing supply chain for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The most immediate financial benefit arises from the complete removal of transition metal catalysts, which are often among the most expensive reagents in a synthetic sequence. By substituting these with inexpensive organic salts and bases, the direct material cost per kilogram of product is significantly lowered. Additionally, the simplified purification process, which does not require specialized metal scavengers or extensive washing steps to meet residual metal limits, reduces solvent consumption and waste disposal costs. This leaner process flow enhances the overall profit margin for the manufactured intermediates, providing a competitive edge in pricing negotiations with downstream API manufacturers.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, including various substituted styrenes and anilines, are widely produced commodity chemicals with established global supply chains. This abundance ensures consistent availability and stable pricing, shielding the production schedule from the supply disruptions often seen with specialized organometallic reagents. The ability to source materials from multiple vendors further strengthens supply security, allowing for flexible procurement strategies. Moreover, the stability of the reagents under ambient conditions simplifies logistics and storage requirements, reducing the need for cold chain transportation or inert atmosphere handling, which streamlines the entire inbound logistics process.
- Scalability and Environmental Compliance: The operational simplicity of running reactions at room temperature and under air atmosphere makes this process inherently scalable from gram to ton quantities without significant re-engineering. This scalability is crucial for meeting the growing demand for pharmaceutical intermediates while maintaining consistent quality. From an environmental standpoint, the metal-free nature of the process aligns perfectly with green chemistry principles, minimizing the generation of heavy metal waste that requires hazardous waste treatment. This compliance with increasingly strict environmental regulations reduces the regulatory burden and potential liability associated with waste management, fostering a more sustainable and socially responsible manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel o-diamine synthesis method. These insights are derived directly from the patent data and are intended to clarify the practical aspects of adopting this technology for industrial applications. Understanding these details helps stakeholders make informed decisions about process integration and resource allocation.
Q: What are the primary advantages of this metal-free synthesis method over traditional transition metal catalysis?
A: The primary advantage is the complete elimination of expensive and toxic transition metal catalysts, which simplifies downstream purification and removes the risk of heavy metal contamination in the final pharmaceutical product.
Q: Is this synthesis method scalable for industrial production of API intermediates?
A: Yes, the method utilizes commercially available reagents, operates at room temperature under air atmosphere, and avoids sensitive conditions, making it highly suitable for large-scale commercial manufacturing.
Q: What types of substrates are compatible with this alkenylsulfonium salt reaction?
A: The reaction demonstrates a broad substrate scope, tolerating various substituents on both the styrene and aniline components, including electron-withdrawing and electron-donating groups like halogens, alkyls, and tert-butyl groups.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable o-Diamine Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of metal-free synthesis technologies in modernizing the production of pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN114957016B can be seamlessly translated into robust industrial processes. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of o-diamine compound meets the highest quality standards required by global regulatory bodies. We are committed to leveraging our technical expertise to optimize these green chemistry routes for maximum efficiency and cost-effectiveness.
We invite pharmaceutical companies and research institutions to collaborate with us to explore the full potential of this metal-free diamination strategy. Whether you require custom synthesis of specific o-diamine derivatives or support for process development and scale-up, our technical procurement team is ready to assist. Please contact us to request a Customized Cost-Saving Analysis tailored to your specific project needs. We encourage you to reach out for specific COA data and route feasibility assessments to determine how this advanced synthesis method can enhance your supply chain resilience and product quality.
