Revolutionizing Selenium-Functionalized Heterocycles: Scalable Synthesis for Next-Generation Antimicrobial Agents
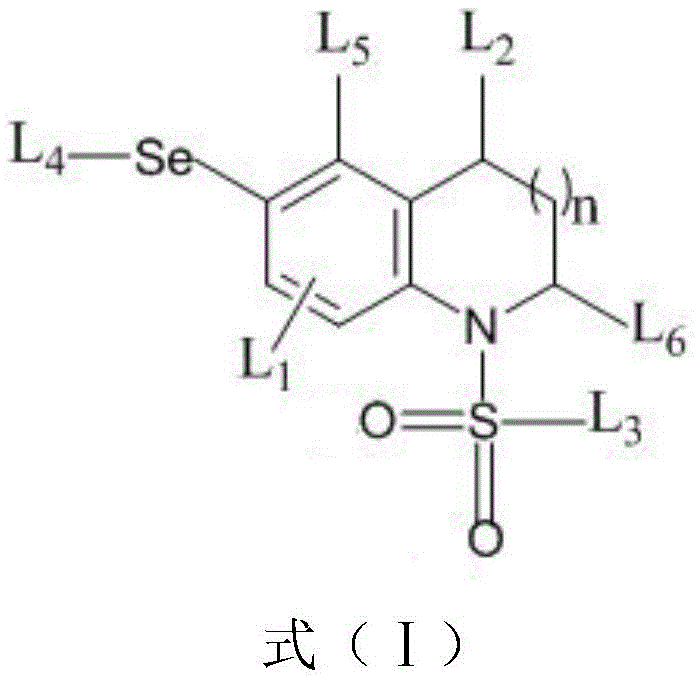
The landscape of medicinal chemistry is constantly evolving, with a particular emphasis on modifying heterocyclic scaffolds to enhance biological activity and metabolic stability. A significant breakthrough in this domain is documented in Chinese Patent CN110003080B, which discloses a novel class of selenium-containing compounds and their preparation methods. These compounds, characterized by a core structure of formula (I), represent a strategic advancement in the functionalization of indoline and tetrahydroquinoline skeletons. Historically, the benzene rings of these heterocycles have been challenging to functionalize selectively, as reactivity is typically dominated by the nitrogen-containing positions. However, this patent introduces a robust copper-catalyzed strategy that achieves site-selective C-H selenization at the C5 position of indolines and the C6 position of tetrahydroquinolines. This innovation not only expands the chemical space available for drug discovery but also provides a streamlined pathway for generating diverse libraries of organoselenium derivatives with potent antibacterial properties.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to functionalizing indoline and quinoline scaffolds have largely focused on the N1, C2, and C3 positions due to their inherent high reactivity. Modifying the benzene ring portion (C4-C7 positions) has historically required harsh reaction conditions or the installation of specific directing groups to guide transition metal catalysts. For instance, previous literature describes Rh(III)-catalyzed C7-selective selenylation, but this necessitates the use of a pyrimidine directing group, which adds extra synthetic steps for installation and subsequent removal. Furthermore, existing methods often suffer from limited substrate scope, poor functional group tolerance, and the use of expensive noble metal catalysts. These factors collectively increase the cost of goods sold (COGS) and complicate the supply chain for pharmaceutical intermediates, making it difficult to access diverse selenium-substituted analogs efficiently for structure-activity relationship (SAR) studies.
The Novel Approach
The methodology presented in the patent overcomes these hurdles by utilizing a copper-catalyzed oxidative coupling system that operates without the need for exogenous directing groups. By leveraging the electronic properties of the N-sulfonyl group, the reaction achieves remarkable regioselectivity for the C5 position in indolines and the C6 position in tetrahydroquinolines. This approach utilizes readily available diaryl diselenides as the selenium source and PhI(OAc)2 as a mild oxidant. The reaction conditions are notably mild, typically proceeding at 80°C in common solvents like THF or acetonitrile. This shift from noble metals like rhodium to base metals like copper represents a paradigm shift in process chemistry, offering a more sustainable and cost-effective route. The ability to directly install selenium moieties onto the benzene ring opens new avenues for developing antimicrobial agents, as organoselenium compounds are known for their antioxidant, anti-inflammatory, and antibacterial activities.
Mechanistic Insights into Copper-Catalyzed C-H Selenization
The mechanistic pathway for this transformation likely involves a copper-mediated radical or electrophilic selenylation process. Initially, the copper(II) trifluoroacetate catalyst interacts with the diaryl diselenide and the hypervalent iodine oxidant to generate a reactive selenium species, possibly a selenyl radical or a high-valent copper-selenium complex. The N-sulfonyl group on the indoline or tetrahydroquinoline substrate plays a crucial role in modulating the electron density of the aromatic ring, activating the C5 or C6 position towards electrophilic attack while deactivating other positions. This electronic bias ensures high regioselectivity without the need for steric directing groups. The oxidative environment provided by PhI(OAc)2 facilitates the regeneration of the active copper catalyst and drives the formation of the C-Se bond. Understanding this mechanism is vital for process optimization, as it highlights the importance of maintaining anhydrous conditions and precise stoichiometry to prevent side reactions such as over-oxidation or diselenide homocoupling.
From an impurity control perspective, the simplicity of the reaction mixture is advantageous. The primary byproducts are typically reduced iodine species and copper salts, which are easily removed during the aqueous workup and silica gel chromatography steps described in the patent. The high yields reported, ranging from 53% to 95% across various substrates, indicate a clean reaction profile with minimal formation of complex organic impurities. This is particularly important for pharmaceutical intermediates, where strict limits on heavy metal residues and organic impurities must be met. The robustness of the catalytic system allows for a wide range of functional groups, including halogens, alkoxy groups, and alkyl chains, to remain intact during the selenization process, thereby preserving the structural diversity required for modern drug discovery campaigns.
How to Synthesize 5-Selenylindolines and 6-Selenyltetrahydroquinolines Efficiently
The synthesis protocol outlined in the patent is designed for operational simplicity and reproducibility, making it highly suitable for both laboratory-scale SAR exploration and pilot-scale production. The general procedure involves mixing the N-sulfonyl substrate with two equivalents of diaryl diselenide in a reaction vessel under an inert atmosphere. The addition of 20 mol% Cu(TFA)2 and two equivalents of PhI(OAc)2 initiates the catalytic cycle. The reaction is heated to 80°C for approximately 12 hours, ensuring complete conversion of the starting material. Post-reaction processing is straightforward, involving extraction with ethyl acetate and water, drying over anhydrous sodium sulfate, and purification via flash column chromatography. This standardized workflow minimizes the need for specialized equipment or hazardous reagents, facilitating rapid adoption by process chemistry teams.
- Mix N-sulfonyl indoline or tetrahydroquinoline substrate (0.25 mmol) with diaryl diselenide (2.0 equivalents) in a reaction vessel.
- Add Cu(TFA)2 catalyst (20 mol%) and PhI(OAc)2 oxidant (2.0 equivalents) to the mixture under an inert argon atmosphere.
- Heat the reaction in THF or acetonitrile at 80°C for 12 hours, followed by aqueous workup and silica gel chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this copper-catalyzed selenization technology offers tangible benefits in terms of cost structure and supply reliability. The shift away from expensive noble metal catalysts like rhodium to abundant copper salts significantly reduces the raw material costs associated with catalyst procurement. Additionally, the elimination of directing group installation and removal steps shortens the overall synthetic sequence, leading to substantial reductions in solvent usage, labor hours, and waste generation. This streamlined process enhances the overall atom economy and aligns with green chemistry principles, which are increasingly important for regulatory compliance and corporate sustainability goals. The use of stable, commercially available starting materials further mitigates supply chain risks, ensuring consistent availability of key intermediates.
- Cost Reduction in Manufacturing: The economic impact of this methodology is driven by the replacement of high-cost noble metals with base metal catalysts and the reduction of synthetic steps. By avoiding the multi-step sequences required for directing group manipulation, manufacturers can achieve significant cost savings in both material and operational expenditures. The high yields obtained, often exceeding 90% for optimized substrates, minimize the loss of valuable starting materials and reduce the burden on downstream purification processes. This efficiency translates directly into a lower cost per kilogram for the final selenium-containing intermediates, providing a competitive edge in the pricing of complex pharmaceutical building blocks.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as Cu(TFA)2, PhI(OAc)2, and common solvents like THF and acetonitrile ensures a robust and resilient supply chain. Unlike specialized ligands or exotic catalysts that may have long lead times or single-source dependencies, the reagents for this process are widely available from multiple global suppliers. This diversification of the supply base reduces the risk of production delays caused by raw material shortages. Furthermore, the mild reaction conditions (80°C, atmospheric pressure) allow the process to be executed in standard glass-lined or stainless steel reactors, maximizing facility utilization and flexibility without requiring capital-intensive infrastructure upgrades.
- Scalability and Environmental Compliance: The process is inherently scalable due to its homogeneous nature and lack of hazardous gas evolution or extreme thermal hazards. The workup procedure involves standard liquid-liquid extraction, which is easily adaptable to large-scale continuous or batch processing. From an environmental standpoint, the reduction in step count and the use of less toxic copper catalysts contribute to a lower environmental footprint. The efficient conversion rates mean less chemical waste is generated per unit of product, simplifying waste treatment and disposal protocols. This alignment with environmental, social, and governance (ESG) criteria makes the technology attractive for long-term partnerships with major pharmaceutical companies seeking sustainable manufacturing solutions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this selenium functionalization technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear understanding of the method's capabilities and limitations for potential licensees or manufacturing partners.
Q: What is the primary advantage of this copper-catalyzed selenization method over traditional rhodium catalysis?
A: Unlike traditional Rh(III)-catalyzed methods that often require complex directing groups to achieve site selectivity, this copper-catalyzed protocol enables direct C5-selective selenization of indolines and C6-selective selenization of tetrahydroquinolines without additional directing moieties, significantly simplifying the synthetic route and reducing raw material costs.
Q: What are the typical yields and purity profiles for these selenium-containing intermediates?
A: According to the patent data, the method demonstrates excellent efficiency with isolated yields reaching up to 94% for indoline derivatives and 95% for tetrahydroquinoline derivatives. The use of standard silica gel chromatography ensures high purity suitable for downstream pharmaceutical applications.
Q: Can this synthesis method be scaled for commercial production of antimicrobial agents?
A: Yes, the reaction utilizes commercially available reagents such as Cu(TFA)2 and PhI(OAc)2 under relatively mild conditions (80°C), avoiding extreme pressures or temperatures. This operational simplicity facilitates commercial scale-up for the production of potential microbial inhibitors targeting pathogens like Pseudomonas aeruginosa and Staphylococcus aureus.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Selenium-Containing Compound Supplier
The development of selenium-functionalized heterocycles represents a critical frontier in the search for new antimicrobial and anti-inflammatory agents. At NINGBO INNO PHARMCHEM, we recognize the strategic value of this technology and possess the technical expertise to bring these complex molecules from benchtop discovery to commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We operate stringent purity specifications and maintain rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of selenium-containing intermediates meets the highest industry standards for potency and impurity profiles.
We invite you to collaborate with us to leverage this innovative copper-catalyzed selenization platform for your drug discovery programs. Whether you require custom synthesis of specific analogs for biological testing or large-scale manufacturing of key intermediates, our technical procurement team is ready to assist. Please contact us to request a Customized Cost-Saving Analysis tailored to your specific project requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can accelerate your timeline and optimize your budget for next-generation therapeutic candidates.
