Scalable Manufacturing of High-Purity Fruquintinib Intermediates via Novel Nickel Catalysis
Scalable Manufacturing of High-Purity Fruquintinib Intermediates via Novel Nickel Catalysis
The pharmaceutical industry's relentless pursuit of effective oncology treatments has placed Vascular Endothelial Growth Factor Receptor (VEGFR) inhibitors at the forefront of therapeutic development. Among these, Fruquintinib stands out as a highly selective oral inhibitor with demonstrated efficacy against metastatic colorectal cancer and non-small cell lung cancer. However, the commercial viability of such potent drugs often hinges on the robustness and cost-efficiency of their synthetic pathways. Patent CN112724110B, published in late 2021, introduces a transformative synthesis method for Fruquintinib and its critical benzofuran intermediates. This technology addresses long-standing bottlenecks in process chemistry by eliminating hazardous reagents and expensive transition metals, thereby offering a streamlined route that is exceptionally well-suited for industrial scale-up. For procurement and supply chain leaders, this patent represents a pivotal shift towards more sustainable and economically predictable manufacturing of high-value API intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of key benzofuran intermediates like 6-hydroxy-N,2-dimethylbenzofuran-3-carboxamide has been plagued by significant process safety and efficiency challenges. Traditional routes often involve the generation of large quantities of sodium sulfide, a byproduct notorious for its extremely unpleasant odor and toxicity, necessitating rigorous and costly waste treatment protocols. Furthermore, to manage these sulfur-containing byproducts, manufacturers frequently resort to using strong oxidizing agents like sodium hypochlorite. This oxidative post-treatment step is not only environmentally burdensome but also poses a severe risk of oxidative decomposition of the sensitive product, leading to suboptimal yields often hovering around 53% with purity levels that require extensive downstream purification. Additionally, many established pathways rely heavily on palladium-catalyzed cross-coupling reactions. While effective, palladium is a precious metal with volatile pricing and supply chain constraints, making it a less desirable choice for cost-sensitive, large-volume pharmaceutical production.
The Novel Approach
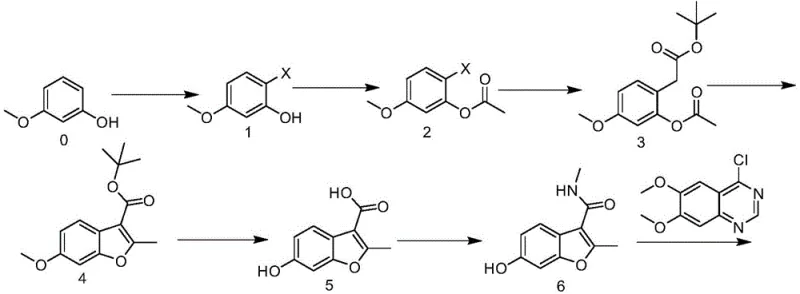
In stark contrast to these legacy methods, the methodology disclosed in CN112724110B offers a radically simplified and safer alternative. The new route comprises seven distinct steps: halogenation, acylation, coupling, ring closure, hydrolysis, amination, and final substitution. By strategically redesigning the synthetic tree, this approach completely bypasses the formation of malodorous sulfur byproducts and eliminates the need for hazardous oxidizing agents. The process utilizes readily available starting materials such as 3-methoxyphenol and employs a nickel-catalyzed coupling strategy that drastically reduces catalyst costs compared to palladium systems. The operational simplicity is further enhanced by the reliance on standard separation techniques like liquid-liquid extraction and crystallization, ensuring high reproducibility. This holistic optimization results in a process that is not only chemically elegant but also commercially superior, delivering high-purity intermediates with improved overall throughput.
Mechanistic Insights into Nickel-Catalyzed Coupling and Cyclization
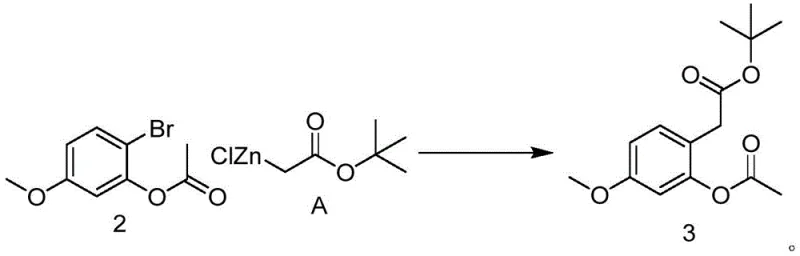
The cornerstone of this innovative synthesis lies in the third step, where a nickel-catalyzed cross-coupling reaction is employed to construct the carbon framework essential for the benzofuran core. Unlike traditional palladium cycles, this method utilizes bis(triphenylphosphine)nickel dibromide in conjunction with an organozinc reagent, specifically 2-tert-butoxy-2-carbonylethyl zinc chloride. The mechanistic advantage here is twofold: firstly, nickel is significantly more abundant and less expensive than palladium, directly impacting the bill of materials; secondly, the specific ligand environment provided by the triphenylphosphine groups facilitates efficient oxidative addition and reductive elimination cycles under mild conditions. The reaction is typically conducted in tetrahydrofuran at low temperatures (around 0°C), which helps in controlling exotherms and minimizing side reactions, thereby preserving the integrity of the sensitive ester and ether functionalities present in the substrate.
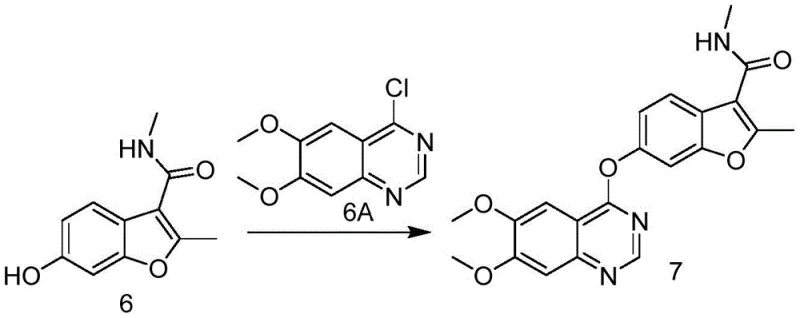
Following the coupling, the pathway proceeds through a base-mediated cyclization to form the benzofuran ring, a critical structural motif. This step utilizes a strong non-nucleophilic base, preferably potassium tert-butoxide, to induce intramolecular condensation. The choice of base is crucial as it must be strong enough to deprotonate the active methylene group without causing hydrolysis of the adjacent ester groups prematurely. Subsequent steps involve a carefully controlled demethylation and hydrolysis using boron tribromide, which selectively cleaves the methyl ether to reveal the phenolic hydroxyl group while simultaneously hydrolyzing the tert-butyl ester to the free carboxylic acid. This tandem transformation is highly efficient, setting the stage for the final amidation. The final coupling with 4-chloro-6,7-dimethoxyquinazoline is achieved under basic conditions in acetone, forging the ether linkage that connects the benzofuran pharmacophore to the quinazoline kinase inhibitor domain, ultimately yielding Fruquintinib with purity exceeding 99%.
How to Synthesize 6-Hydroxy-N,2-dimethylbenzofuran-3-carboxamide Efficiently
The synthesis of this critical intermediate is a multi-stage process that demands precise control over reaction parameters to ensure high yield and purity. The protocol begins with the halogenation of 3-methoxyphenol, followed by protection of the phenol group via acylation. The heart of the synthesis is the nickel-catalyzed coupling, which requires strict moisture control and inert atmosphere conditions to prevent the degradation of the organozinc species. Following the coupling, the cyclization step must be monitored closely to ensure complete ring closure before proceeding to the acidic workup. The subsequent hydrolysis and demethylation steps utilize boron halides, which are moisture-sensitive and require careful quenching procedures. Finally, the activation of the carboxylic acid using CDI followed by amidation with methylamine completes the intermediate synthesis. For detailed operational parameters, stoichiometry, and workup procedures, please refer to the standardized guide below.
- Halogenation of 3-methoxyphenol using NBS or NIS to form compound 1.
- Acylation of compound 1 with acetic anhydride to protect the phenol group, yielding compound 2.
- Nickel-catalyzed coupling of compound 2 with organozinc reagent to introduce the side chain, forming compound 3.
- Base-mediated cyclization using potassium tert-butoxide to close the benzofuran ring, affording compound 4.
- Hydrolysis and demethylation using boron tribromide to reveal the phenol and carboxylic acid groups in compound 5.
- Amidation with methylamine using CDI activation to produce the key intermediate compound 6.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers compelling strategic advantages that extend beyond simple chemical yield. The primary value driver is the substantial reduction in raw material costs achieved by replacing expensive palladium catalysts with a nickel-based system. This switch not only lowers the direct cost of goods sold but also mitigates the supply risk associated with precious metal availability. Furthermore, the elimination of hazardous reagents like sodium sulfide and strong oxidizers simplifies the environmental, health, and safety (EHS) compliance burden. This translates to lower waste disposal costs and reduced regulatory friction, allowing for smoother continuous production runs without the interruptions often caused by complex effluent treatment requirements.
- Cost Reduction in Manufacturing: The economic impact of this process is profound due to the strategic substitution of catalysts and reagents. By utilizing a nickel catalyst instead of palladium, the process significantly lowers the input cost for one of the most expensive components in the synthesis. Additionally, the use of commercially available starting materials like 3-methoxyphenol ensures a stable and competitive supply base. The avoidance of complex post-treatment steps, such as the oxidation of sulfur byproducts, further reduces the consumption of auxiliary chemicals and energy, leading to a leaner and more cost-effective manufacturing profile that enhances overall margin potential.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the simplicity and robustness of the reaction sequence. The reliance on standard unit operations such as filtration, extraction, and crystallization means that the process can be executed in a wide range of multipurpose chemical facilities without the need for specialized equipment. The high reproducibility of the synthetic route ensures consistent batch-to-batch quality, reducing the risk of production delays caused by failed batches or out-of-specification results. This reliability is critical for maintaining uninterrupted supply lines to downstream API manufacturers and ensuring timely delivery of finished drug products to the market.
- Scalability and Environmental Compliance: From a sustainability perspective, this method aligns perfectly with modern green chemistry principles. The absence of toxic sulfur byproducts and the minimization of heavy metal usage significantly reduce the environmental footprint of the manufacturing process. This makes the technology highly scalable, as it avoids the bottlenecks often encountered when trying to scale up processes with difficult waste streams. The ability to handle larger batch sizes safely and efficiently allows manufacturers to respond agilely to market demand fluctuations, ensuring that supply can be ramped up rapidly to meet clinical or commercial needs without compromising on safety or environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear picture of the process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for their own manufacturing portfolios.
Q: How does this new synthesis route improve upon conventional methods for Fruquintinib intermediates?
A: Conventional routes often rely on expensive palladium catalysts or generate hazardous byproducts like sodium sulfide, which requires complex post-treatment with oxidizing agents that can degrade the product. This novel method utilizes a cost-effective nickel catalyst system and avoids dangerous reagents, resulting in a safer process with higher reproducibility and easier purification.
Q: What represents the key technological breakthrough in this patent regarding catalysis?
A: The core innovation lies in the substitution of traditional palladium catalysts with a bis(triphenylphosphine)nickel dibromide system for the cross-coupling step. This switch significantly reduces raw material costs while maintaining high coupling efficiency, making the process economically viable for large-scale industrial production without compromising yield.
Q: Is this synthetic route suitable for commercial scale-up?
A: Yes, the patent explicitly states that the method uses commercially available starting materials and involves simple unit operations such as liquid-liquid and solid-liquid separations. The absence of explosive or highly toxic reagents, combined with good reproducibility across multiple steps, ensures that the process is robust enough for metric-ton scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fruquintinib Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic pathways in the development of life-saving oncology therapies. Our team of expert process chemists has thoroughly analyzed the technology disclosed in CN112724110B and is fully equipped to translate this laboratory-scale innovation into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot plant to full-scale manufacturing is seamless. Our state-of-the-art facilities are designed to handle complex organic syntheses with stringent purity specifications, supported by rigorous QC labs that guarantee every batch meets the highest international standards for pharmaceutical intermediates.
We invite you to collaborate with us to leverage this advanced synthesis technology for your supply chain. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our manufacturing expertise can drive efficiency and reliability in your Fruquintinib supply chain.
