Revolutionizing Asymmetric Cyanine Dye Production: A Breakthrough in Regioselective Synthesis for Biomedical Imaging
Revolutionizing Asymmetric Cyanine Dye Production: A Breakthrough in Regioselective Synthesis for Biomedical Imaging
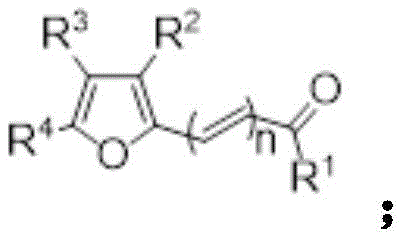
The landscape of biomedical imaging and photoelectric material science is undergoing a significant transformation driven by advancements in fluorophore chemistry. Specifically, the demand for high-performance asymmetric cyanine dyes, such as Cy5 and Cy7 derivatives, has surged due to their exceptional molar absorptivity and biocompatibility. A pivotal development in this sector is detailed in patent CN115724783A, which discloses a novel synthetic methodology utilizing furfural as a strategic starting material. This approach fundamentally addresses the longstanding challenge of regioselectivity in constructing heterocyclic different asymmetric cyanine dyes. By leveraging a stepwise condensation strategy, this technology enables the precise assembly of complex conjugated systems without the formation of difficult-to-remove symmetrical by-products. For R&D directors and procurement specialists in the fine chemical industry, this represents a critical opportunity to optimize supply chains for high-purity imaging agents while simultaneously reducing the environmental footprint associated with traditional dye synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of cyanine dyes has relied heavily on the reaction between pentadienal diphenylamine condensing agents and indole quaternary ammonium salts. The synthesis of these essential condensing agents is notoriously cumbersome, typically involving either the reaction of polysubstituted pyridine with 2,4-dinitrohalogenated benzene to form Zincke salts, or the Vilsmeier-Haack formylation of cyclohexanone followed by aminolysis. These multi-step pathways not only increase the overall cost of goods sold but also introduce significant variability in batch quality. Furthermore, a critical technical bottleneck exists in the final coupling stage: when attempting to synthesize asymmetric dyes with different heterocyclic rings, conventional methods frequently generate symmetrical by-products with polarities nearly identical to the target molecule. This similarity in physicochemical properties renders separation via standard chromatographic techniques extremely inefficient, leading to low overall yields and compromised purity profiles that are unacceptable for sensitive biomedical applications.
The Novel Approach
In stark contrast to these legacy processes, the methodology outlined in patent CN115724783A introduces a streamlined, stepwise condensation protocol that capitalizes on the unique reactivity of the furan ring. The process initiates with the condensation of a furfural derivative with a single indole quaternary ammonium salt to form a stable hemicyanine intermediate. Subsequently, under the catalytic influence of a Lewis acid and an arylamine, the furan ring undergoes a controlled ring-opening reaction. This activated intermediate then reacts selectively with a second, distinct molecule of indole quaternary ammonium salt. This sequential addition mechanism inherently prevents the formation of symmetrical dimers, ensuring that the final product is exclusively the desired asymmetric cyanine dye. This strategic shift from a one-pot chaotic coupling to a controlled, linear assembly line offers a robust solution for producing high-purity dyes suitable for rigorous analytical chemistry and medical diagnostics.
Mechanistic Insights into Lewis Acid-Catalyzed Furan Ring Opening
The core innovation of this synthetic route lies in the mechanistic elegance of the furan ring-opening step, which serves as the linchpin for achieving regioselective control. In the second stage of the synthesis, the hemicyanine intermediate, possessing a furan moiety, is treated with a Lewis acid such as zinc chloride (ZnCl2) or aluminum chloride in the presence of an arylamine like p-anisidine. The Lewis acid coordinates with the oxygen atom of the furan ring, increasing its electrophilicity and facilitating nucleophilic attack by the arylamine. This interaction triggers the cleavage of the carbon-oxygen bond within the furan ring, effectively linearizing the conjugated system and generating a highly reactive electrophilic center. This transient species is then perfectly poised to undergo a condensation reaction with the active methylene group of the second heterocyclic quaternary salt. This mechanism bypasses the statistical distribution of products seen in traditional aldehyde condensations, providing a deterministic pathway to the asymmetric target.
From an impurity control perspective, this mechanism offers substantial advantages for process chemists aiming for GMP-grade materials. Because the ring-opening event is chemically distinct and requires specific catalytic conditions, side reactions are minimized compared to the harsh acidic or basic conditions often required for Zincke salt preparations. The use of mild solvents such as methanol and ethanol further suppresses the degradation of sensitive fluorophore structures. Additionally, the ability to isolate the hemicyanine intermediate allows for quality control checkpoints before the final coupling step. If the intermediate does not meet purity specifications, it can be purified prior to the ring-opening reaction, thereby preventing the propagation of impurities into the final high-value dye product. This modularity is essential for maintaining the stringent purity specifications required for in vivo imaging applications where trace metal contaminants or organic impurities could induce toxicity.
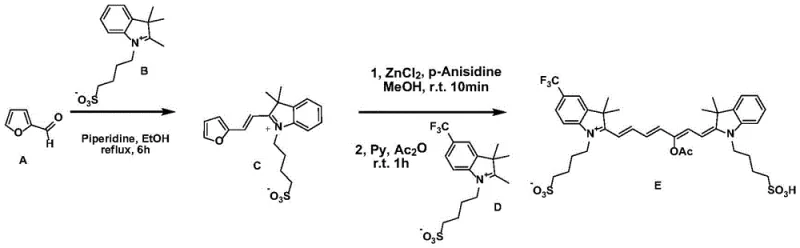
How to Synthesize Asymmetric Cyanine Dyes Efficiently
The practical implementation of this technology involves a straightforward two-stage protocol that is amenable to both laboratory scale optimization and pilot plant operations. The process begins with the formation of the hemicyanine precursor, followed by the critical ring-opening and coupling sequence. Detailed operational parameters, including specific stoichiometric ratios, temperature gradients, and workup procedures, are essential for maximizing yield and minimizing waste. For technical teams looking to replicate or license this technology, understanding the precise order of addition and the role of the secondary amine catalyst in the first step is vital. The following guide outlines the standardized synthesis steps derived from the patent examples, serving as a foundational reference for process development.
- Condense furfural derivative A with heterocycle B in an alcoholic solvent using a secondary amine catalyst at 60-140°C to form hemicyanine C.
- React hemicyanine C with a Lewis acid and arylamine in methanol, then couple with heterocycle D in the presence of acid anhydride and base to yield the final asymmetric dye E.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this furfural-based synthesis route presents a compelling value proposition centered on cost efficiency and supply reliability. The primary driver for cost reduction is the substitution of complex, multi-step condensing agents with readily available commodity chemicals like furfural and indole derivatives. By eliminating the need for proprietary or difficult-to-source intermediates such as Zincke salts, manufacturers can significantly lower their raw material expenditure. Furthermore, the simplified purification process, which avoids the challenging separation of symmetrical by-products, reduces the consumption of chromatographic media and solvents. This streamlining of the downstream processing phase translates directly into lower operational expenditures and a reduced environmental burden, aligning with modern green chemistry initiatives and sustainability goals.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived from the drastic simplification of the synthetic route. Traditional methods require the pre-synthesis of unstable condensing agents, which adds time, labor, and material costs. In this novel approach, the use of stable furfural derivatives allows for a direct convergent synthesis. The elimination of expensive transition metal catalysts or hazardous reagents further contributes to cost savings. Additionally, the higher selectivity of the reaction means that less starting material is wasted on unwanted side products, improving the overall atom economy and reducing the cost per gram of the final high-purity dye.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of ubiquitous raw materials. Furfural is a biomass-derived platform chemical with a robust global supply network, reducing the risk of shortages associated with specialized petrochemical intermediates. The reaction conditions are mild, typically ranging from room temperature to moderate reflux, which reduces the energy intensity of the manufacturing process and lowers the barrier for contract manufacturing organizations (CMOs) to adopt the technology. This accessibility ensures that multiple qualified suppliers can be onboarded quickly, mitigating the risk of single-source dependency for critical imaging reagents.
- Scalability and Environmental Compliance: The scalability of this process is supported by its compatibility with standard pharmaceutical equipment. The use of common alcoholic solvents like ethanol and methanol simplifies solvent recovery and recycling systems, which is a key factor in meeting increasingly strict environmental regulations. The absence of heavy metal catalysts in the final coupling step minimizes the need for complex metal scavenging procedures, which are often a bottleneck in scaling up catalytic processes. This clean profile facilitates faster regulatory approval for new dye formulations and ensures a consistent supply of compliant materials for clinical and diagnostic applications.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric cyanine dye synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear understanding of the method's capabilities and limitations. Understanding these details is crucial for R&D teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What is the primary advantage of this furfural-based method over traditional condensing agents?
A: The primary advantage is the elimination of symmetrical by-products. Traditional methods using glutaraldehyde diphenylamine often generate polarity-similar symmetrical impurities that are difficult to separate. This stepwise furfural route ensures regioselective monoselectivity, drastically simplifying purification.
Q: Can this synthesis method be scaled for industrial production of biomedical dyes?
A: Yes, the method utilizes easily available raw materials like furfural and operates under mild conditions (room temperature to reflux) using common solvents like ethanol and methanol. The absence of complex multi-step condensing agent preparation makes it highly suitable for large-scale commercial manufacturing.
Q: What specific applications do these asymmetric cyanine dyes support?
A: These dyes exhibit excellent water solubility and fluorescence properties, making them ideal for near-infrared fluorescence imaging, photoacoustic imaging, photodynamic therapy, and sonodynamic therapy in the biomedical field.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Asymmetric Cyanine Dyes Supplier
The technological potential of the furfural-mediated synthesis route described in patent CN115724783A offers a clear pathway to next-generation biomedical imaging agents. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate such innovative academic and patent discoveries into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to market-ready product is seamless. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of asymmetric cyanine dye meets the exacting standards required for in vivo diagnostics and therapeutic applications.
We invite you to collaborate with us to optimize your supply chain for high-value fluorophores. Our engineering team is ready to conduct a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. By leveraging our process development capabilities, we can help you reduce lead time for high-purity asymmetric cyanine dyes and secure a stable supply of critical materials. Please contact our technical procurement team today to request specific COA data and route feasibility assessments for your upcoming projects.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
