Advanced Rhodium-Catalyzed C-H Activation for Scalable Novel Amino Acid Manufacturing
Advanced Rhodium-Catalyzed C-H Activation for Scalable Novel Amino Acid Manufacturing
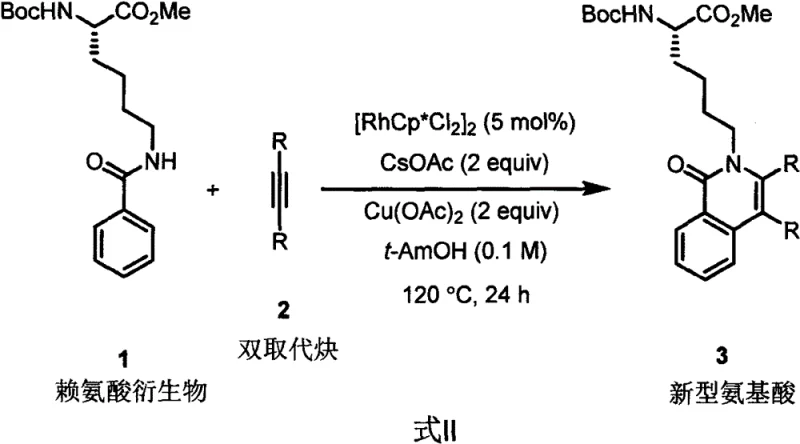
The landscape of polypeptide drug development is undergoing a significant transformation, driven by the urgent need for unnatural amino acids that offer superior biological activity and stability compared to their natural counterparts. Patent CN115894366A introduces a groundbreaking methodology utilizing rhodium-catalyzed carbon-hydrogen (C-H) activation to construct novel amino acid scaffolds directly from lysine derivatives. This technology represents a paradigm shift in organic synthesis, moving away from laborious multi-step functionalization towards direct, atom-economical annulation strategies. For R&D directors and procurement specialists in the pharmaceutical sector, this innovation offers a robust pathway to access complex isoquinolinone-fused amino acids, which serve as critical building blocks for next-generation peptidomimetics and therapeutic agents. The process leverages a pentamethylcyclopentadienyl rhodium catalyst system to achieve selective C-H bond cleavage and subsequent cyclization with disubstituted alkynes, delivering high-value intermediates with remarkable efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for modifying natural amino acids to create unnatural variants often suffer from significant inefficiencies that hinder large-scale production. Conventional strategies typically rely heavily on pre-functionalized substrates, necessitating the installation of leaving groups such as halides or triflates prior to the key coupling step. This requirement not only adds multiple synthetic steps, thereby increasing the overall lead time and operational expenditure, but also generates substantial amounts of hazardous waste associated with halogenation reagents. Furthermore, classical cross-coupling reactions often demand stringent anhydrous conditions and expensive palladium catalysts, which can be sensitive to air and moisture, complicating the manufacturing process. The cumulative effect of these limitations is a higher cost of goods sold (COGS) and a more complex supply chain, making it difficult for pharmaceutical manufacturers to scale these processes to the multi-kilogram or tonnage levels required for clinical and commercial supply without compromising purity profiles.
The Novel Approach
In stark contrast, the rhodium-catalyzed C-H activation strategy disclosed in the patent data offers a streamlined, direct approach that bypasses the need for pre-functionalization entirely. By utilizing readily available lysine derivatives and simple disubstituted alkynes as starting materials, this method achieves the construction of the isoquinolinone core in a single pot. The reaction proceeds under relatively mild oxidative conditions using copper acetate as a terminal oxidant, which regenerates the active rhodium species and drives the catalytic cycle forward. This direct annulation strategy significantly enhances step economy, effectively collapsing what would traditionally be a three-to-four-step sequence into a single transformative operation. The versatility of this approach is evidenced by its tolerance to a wide range of substituents on the alkyne component, including electron-rich and electron-deficient aryl groups, allowing for the rapid generation of diverse chemical libraries for drug discovery efforts without the burden of synthesizing unique precursors for each variant.
Mechanistic Insights into Rhodium-Catalyzed C-H Activation and Annulation
The core of this technological advancement lies in the sophisticated mechanism of Rh(III)-catalyzed C-H activation, which enables the selective functionalization of inert C-H bonds adjacent to the directing group. The catalytic cycle initiates with the coordination of the amide oxygen of the lysine derivative to the cationic rhodium species, generated in situ from the dimeric precursor [RhCp*Cl2]2. This coordination facilitates the concerted metalation-deprotonation (CMD) process, assisted by the acetate base, leading to the formation of a stable five-membered rhodacycle intermediate. This step is crucial as it determines the regioselectivity of the reaction, ensuring that activation occurs specifically at the ortho-position of the benzamide moiety. Subsequently, the disubstituted alkyne coordinates to the metal center and undergoes migratory insertion into the Rh-C bond, expanding the metallacycle and setting the stage for ring closure. The final step involves reductive elimination or oxidation-assisted protodemetalation, facilitated by the Cu(II) oxidant, which releases the novel isoquinolinone product and regenerates the active Rh(III) catalyst for the next turnover.
From a quality control perspective, understanding this mechanism is vital for managing impurity profiles and ensuring batch-to-batch consistency. The use of cesium acetate as a base plays a dual role: it acts as a proton shuttle during the C-H cleavage step and helps maintain the solubility of the metal species in the alcoholic solvent system. The choice of 2-methyl-2-butanol (t-AmOH) as the solvent is particularly strategic, as it provides the necessary thermal stability at the reaction temperature of 120 °C while minimizing side reactions such as solvent oxidation or ester hydrolysis that might occur in other protic media. Furthermore, the oxidative nature of the reaction, driven by copper acetate, ensures that the rhodium catalyst remains in the active +3 oxidation state throughout the process, preventing the formation of inactive rhodium black precipitates that could contaminate the final product. This mechanistic robustness translates directly into easier downstream processing, as the reaction mixture typically contains fewer metal-containing byproducts compared to traditional palladium-catalyzed couplings, simplifying the purification workflow.
How to Synthesize Novel Amino Acids Efficiently
The practical implementation of this synthesis route is designed for operational simplicity, making it highly attractive for process chemistry teams aiming to scale up production. The protocol involves charging a pressure-resistant vessel with the lysine derivative substrate, the chosen disubstituted alkyne, the rhodium catalyst precursor, cesium acetate, and copper acetate in the specified molar ratios. The mixture is then heated to 120 °C for a period of 24 hours to ensure complete conversion. Following the reaction, the workup procedure is straightforward, involving the removal of the solvent under reduced pressure followed by standard silica gel column chromatography. This ease of execution, combined with the high atom economy of the transformation, positions this method as a superior alternative for the commercial scale-up of complex pharmaceutical intermediates. For detailed operational parameters and safety considerations, please refer to the standardized synthesis guide below.
- Charge a pressure-resistant reactor with lysine derivative, disubstituted alkyne, [RhCp*Cl2]2 catalyst (5 mol%), CsOAc (2 equiv), and Cu(OAc)2 (2 equiv) in t-AmOH solvent.
- Stir the reaction mixture at 120 °C for 24 hours to facilitate the oxidative annulation and C-H activation process.
- Remove the solvent via rotary evaporation and purify the crude residue using silica gel column chromatography to isolate the target novel amino acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this rhodium-catalyzed technology offers tangible strategic benefits that extend beyond mere technical novelty. The primary advantage lies in the drastic simplification of the raw material supply chain. By eliminating the need for pre-halogenated intermediates, which are often subject to volatile pricing and supply constraints due to their hazardous nature, manufacturers can source cheaper, more abundant commodity chemicals like simple alkynes and protected amino acids. This shift not only stabilizes the supply chain against market fluctuations but also reduces the regulatory burden associated with handling and transporting hazardous halogenated compounds. Additionally, the reduction in synthetic steps directly correlates to a significant decrease in manufacturing costs, as fewer unit operations mean lower consumption of solvents, energy, and labor hours per kilogram of finished product.
- Cost Reduction in Manufacturing: The elimination of pre-functionalization steps removes the cost associated with purchasing or synthesizing expensive halogenated building blocks, which often carry a premium price tag due to their specialized nature. Furthermore, the high atom economy of the C-H activation process means that a larger proportion of the starting material mass is incorporated into the final product, reducing waste disposal costs and improving the overall material efficiency of the plant. The use of a recyclable heterogeneous workup or simple chromatographic purification further lowers the operational expenditure compared to complex extraction sequences required for traditional methods.
- Enhanced Supply Chain Reliability: Relying on broadly available feedstocks such as diphenylacetylene derivatives and protected lysine ensures a resilient supply chain that is less susceptible to disruptions from single-source suppliers. The robustness of the reaction conditions, which tolerate a variety of functional groups, allows for flexibility in sourcing; if one specific alkyne supplier faces issues, alternative commercially available analogs can often be substituted without requiring a complete re-optimization of the synthetic route. This flexibility is critical for maintaining continuous production schedules and meeting tight delivery deadlines for clinical trial materials.
- Scalability and Environmental Compliance: The process operates in a single alcoholic solvent system, which simplifies solvent recovery and recycling protocols, aligning with modern green chemistry principles and reducing the environmental footprint of the manufacturing site. The absence of toxic tin or boron reagents, commonly found in traditional cross-coupling methodologies, simplifies waste stream treatment and ensures compliance with increasingly stringent environmental regulations regarding heavy metal discharge. This environmental compatibility facilitates smoother regulatory approvals and reduces the long-term liability associated with hazardous waste management.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this rhodium-catalyzed C-H activation technology. These insights are derived directly from the experimental data and beneficial effects described in the patent literature, providing a clear picture of the method's capabilities and limitations for potential adopters. Understanding these nuances is essential for integrating this chemistry into existing manufacturing pipelines effectively.
Q: What are the key advantages of this Rh-catalyzed method over traditional amino acid modification?
A: This method eliminates the need for pre-functionalized substrates such as halogenated intermediates, significantly improving atom economy and reducing synthetic steps compared to traditional cross-coupling approaches.
Q: What is the typical yield and purity profile for these novel amino acid derivatives?
A: The patent reports isolated yields ranging from 39% to 86% depending on the alkyne substituent, with products being easily purified via standard silica gel chromatography to meet stringent pharmaceutical specifications.
Q: Can this process be adapted for different alkyne substrates?
A: Yes, the methodology demonstrates broad substrate scope, successfully accommodating symmetric diaryl alkynes with electron-donating (methoxy, methyl) and electron-withdrawing (trifluoromethyl) groups, as well as dialkyl alkynes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Novel Amino Acids Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced C-H activation technologies in accelerating the development of novel therapeutics. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from bench-scale discovery to industrial manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped to handle sensitive organometallic chemistries safely, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch of novel amino acid intermediates meets the exacting standards required for pharmaceutical applications. We are committed to leveraging cutting-edge synthetic methodologies to deliver high-quality solutions that drive your drug development programs forward.
We invite you to collaborate with us to explore how this rhodium-catalyzed route can optimize your specific project needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your target molecules, demonstrating exactly how this technology can reduce your overall production costs. Please contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss custom route feasibility assessments for your proprietary candidates. Let us be your partner in turning complex chemical challenges into commercial successes.
