Advanced Rhodium-Catalyzed Synthesis of Polysubstituted Isoquinoline Derivatives for Commercial Scale-Up
Advanced Rhodium-Catalyzed Synthesis of Polysubstituted Isoquinoline Derivatives for Commercial Scale-Up
The landscape of heterocyclic chemistry is undergoing a significant transformation driven by the urgent need for greener, more efficient synthetic methodologies, particularly for high-value scaffolds like isoquinolines. Patent CN108484499B introduces a groundbreaking method for preparing polysubstituted isoquinoline derivatives directly from hydroxylamine and diaryl alkyne compounds. This technology represents a paradigm shift away from classical condensation reactions that rely on harsh acidic conditions and stoichiometric oxidants. By utilizing a trivalent rhodium catalyst system in an ethanol solvent, this process achieves high atom utilization and exceptional chemical selectivity. For R&D directors and procurement specialists in the fine chemical sector, this patent offers a robust pathway to access complex nitrogen-containing heterocycles which are critical building blocks for pharmaceuticals and advanced electronic materials. The ability to synthesize these structures in a single pot with minimal byproduct formation addresses long-standing challenges in process chemistry regarding waste management and operational safety.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the isoquinoline skeleton has relied heavily on named reactions such as the Bischler-Napieralski or Pictet-Spengler reactions, which, while effective, suffer from severe drawbacks in a modern industrial context. These traditional pathways typically necessitate the use of strong dehydrating agents like phosphorus pentoxide or harsh Lewis acids, creating significant environmental hazards and complicating waste disposal protocols. Furthermore, many contemporary transition-metal catalyzed approaches reported in literature often require the pre-functionalization of substrates with halogens or the addition of stoichiometric amounts of expensive oxidants like copper acetate to drive the catalytic cycle. This reliance on external oxidants not only increases the raw material costs drastically but also generates substantial quantities of metal salt waste that must be removed during purification. Additionally, the narrow substrate scope of many older methods limits the ability to generate diverse molecular libraries required for modern drug discovery programs, often failing when electron-rich or sterically hindered groups are present on the aromatic rings.
The Novel Approach
In stark contrast to these legacy technologies, the method disclosed in CN108484499B utilizes a direct annulation strategy that bypasses the need for pre-activated nitrogen sources or external oxidants. The core innovation lies in the use of readily available hydroxylamine as the nitrogen source, which reacts with diaryl alkynes under the influence of a pentamethylcyclopentadienyl rhodium(III) catalyst. This system operates efficiently in green solvents like ethanol at elevated temperatures, completing the cyclization process solely through dehydration. The elimination of stoichiometric oxidants is a major breakthrough, as it simplifies the reaction profile and reduces the burden on downstream purification processes. This novel approach ensures that the only byproducts generated are water and catalytic amounts of potassium chloride, aligning perfectly with the principles of green chemistry and sustainable manufacturing. Such a streamlined process significantly enhances the feasibility of scaling this reaction from gram-scale laboratory synthesis to multi-ton commercial production.
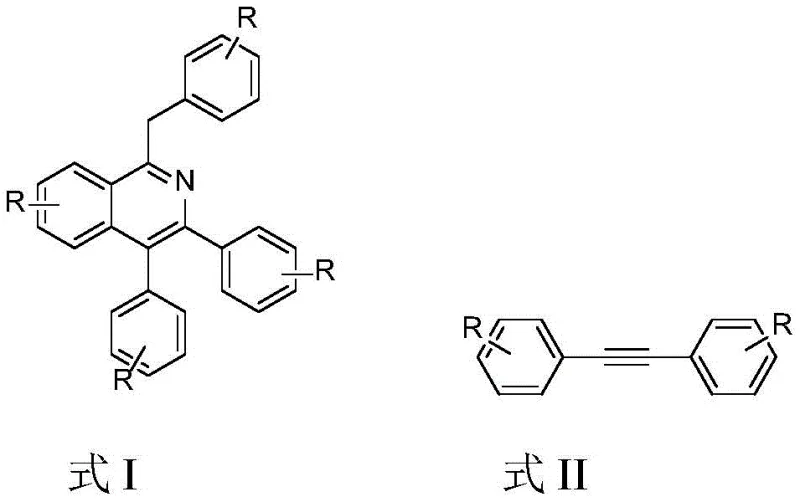
Mechanistic Insights into Rh(III)-Catalyzed C-H Activation and Cyclization
The mechanistic elegance of this transformation centers on the capability of the cationic Rh(III) species to activate inert carbon-hydrogen bonds directly, facilitating the formation of new carbon-nitrogen and carbon-carbon bonds in a concerted manner. The catalytic cycle likely initiates with the coordination of the alkyne substrate to the rhodium center, followed by the insertion of the nitrogen species derived from hydroxylamine. Unlike oxidative coupling mechanisms that consume external oxidants to regenerate the active catalyst, this system appears to utilize an internal redox neutral pathway where the dehydration step drives the aromatization of the isoquinoline ring. The choice of potassium acetate as the base is critical, as it assists in the C-H activation step by acting as a proton shuttle, lowering the energy barrier for the metallacycle formation. This precise control over the reaction coordinate ensures high regioselectivity, preventing the formation of unwanted isomers that often plague non-directed C-H functionalization reactions. Understanding this mechanism allows process chemists to fine-tune reaction parameters such as temperature and solvent polarity to maximize turnover numbers.
From an impurity control perspective, the high chemoselectivity of this rhodium-catalyzed system is paramount for producing pharmaceutical-grade intermediates. The reaction conditions are mild enough to tolerate sensitive functional groups such as halogens (fluorine, chlorine, bromine) and electron-donating groups (methoxy, alkyl) without causing dehalogenation or over-oxidation side reactions. This tolerance is crucial because it preserves the structural integrity of complex molecules that may undergo further derivatization in later synthetic steps. The absence of heavy metal oxidants like copper eliminates the risk of difficult-to-remove metal residues in the final product, thereby reducing the need for extensive scavenging treatments. Furthermore, the use of ethanol as a solvent minimizes the formation of solvent-derived impurities that are common when using chlorinated or high-boiling polar aprotic solvents. This clean reaction profile translates directly into higher purity profiles for the final isoquinoline derivatives, reducing the analytical burden on quality control laboratories.
How to Synthesize Polysubstituted Isoquinoline Derivatives Efficiently
Implementing this synthesis route in a practical setting requires careful attention to the stoichiometry of the hydroxylamine source and the maintenance of an inert atmosphere to prevent catalyst deactivation. The patent outlines a straightforward one-pot procedure where the diaryl alkyne, hydroxylamine solution, base, and catalyst are combined in ethanol before heating. The simplicity of the operation belies the sophistication of the underlying chemistry, allowing for robust execution even in large-scale reactors. Operators should note that the reaction time can vary between 12 to 24 hours depending on the electronic nature of the substituents on the alkyne substrate, necessitating monitoring via TLC or GC to determine the optimal endpoint. The detailed standardized synthesis steps below provide a clear roadmap for replicating the high yields reported in the patent examples, ensuring consistency across different batches.
- Mix diaryl alkyne compound, aqueous hydroxylamine solution, potassium acetate base, and [Cp*RhCl2]2 catalyst in ethanol solvent.
- Seal the reaction vessel under nitrogen atmosphere and heat the mixture to 140°C for approximately 18 hours with stirring.
- Upon completion, purify the crude mixture via silica gel column chromatography using petroleum ether and ethyl acetate eluents to isolate the target isoquinoline.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers tangible strategic advantages that extend beyond mere technical feasibility. The primary benefit lies in the drastic simplification of the raw material supply chain, as both diaryl alkynes and hydroxylamine are commodity chemicals available from multiple global suppliers, mitigating the risk of single-source dependency. The elimination of expensive and hazardous stoichiometric oxidants translates directly into substantial cost savings in terms of raw material procurement and waste disposal fees. Moreover, the use of ethanol as the primary solvent enhances workplace safety and reduces the regulatory burden associated with the handling and disposal of volatile organic compounds or chlorinated solvents. These factors collectively contribute to a more resilient and cost-effective manufacturing process that can withstand market fluctuations and regulatory changes.
- Cost Reduction in Manufacturing: The economic viability of this process is significantly enhanced by the low loading of the rhodium catalyst, which is required in only catalytic amounts (0.5% to 2.5%), thereby minimizing the impact of precious metal costs on the overall bill of materials. By removing the need for stoichiometric oxidants like copper acetate, the process avoids the generation of large volumes of heavy metal waste, which incurs high treatment and disposal costs in compliant facilities. The high atom economy of the reaction ensures that the majority of the starting mass is incorporated into the final product, maximizing yield per kilogram of input. Additionally, the simplified workup procedure involving standard silica gel chromatography reduces the consumption of specialized purification resins and solvents, further driving down the operational expenditure per unit of production.
- Enhanced Supply Chain Reliability: The reliance on widely available feedstock chemicals such as substituted alkynes and aqueous hydroxylamine ensures a stable and continuous supply chain, free from the bottlenecks often associated with custom-synthesized specialty reagents. The robustness of the reaction conditions, which tolerate a wide range of substituents including halogens and alkyl groups, allows for flexibility in sourcing raw materials with varying purity grades without compromising the final product quality. This flexibility is critical for maintaining production schedules during periods of raw material scarcity or price volatility. Furthermore, the scalability of the one-pot protocol means that production can be ramped up quickly to meet surging demand without the need for complex process re-engineering or additional equipment investment.
- Scalability and Environmental Compliance: From an environmental standpoint, this process aligns perfectly with increasingly stringent global regulations regarding industrial emissions and waste generation. The generation of water and catalytic potassium chloride as the sole byproducts means that the effluent load is minimal and easily treatable, reducing the facility's environmental footprint. The use of bio-derived ethanol as a solvent supports sustainability goals and may qualify the manufacturing process for green chemistry certifications, which are becoming a prerequisite for supplying major multinational pharmaceutical companies. The thermal stability of the reaction mixture at 140°C allows for safe operation in standard stainless steel reactors, facilitating seamless scale-up from pilot plant to commercial tonnage without encountering exothermic runaway risks.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this rhodium-catalyzed isoquinoline synthesis technology. These answers are derived directly from the experimental data and specifications outlined in the patent documentation to ensure accuracy and reliability for potential partners. Understanding these details is essential for evaluating the fit of this technology within your existing manufacturing infrastructure and product portfolio. We encourage technical teams to review these points closely to appreciate the full scope of the process advantages.
Q: Does this synthesis method require external oxidants?
A: No, unlike traditional methods requiring stoichiometric copper acetate, this patented process achieves cyclization through dehydration without adding external oxidants, significantly reducing waste.
Q: What is the optimal catalyst loading for this reaction?
A: The patent specifies a trivalent rhodium catalyst loading of 0.5% to 2.5% molar ratio relative to the substrate, with 1.0% being the preferred optimal concentration for high yields.
Q: Can this method tolerate halogen substituents on the substrate?
A: Yes, the reaction system demonstrates excellent compatibility with various substrates including those bearing fluorine, chlorine, and bromine substituents, maintaining high isolated yields.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted Isoquinoline Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic route described in CN108484499B for the production of high-value pharmaceutical intermediates and functional materials. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate this laboratory-scale innovation into a robust, GMP-compliant commercial manufacturing process. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench to plant is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of polysubstituted isoquinoline derivatives meets the exacting standards required by the global pharmaceutical industry.
We invite you to collaborate with us to leverage this advanced technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact us to request specific COA data for similar isoquinoline scaffolds and comprehensive route feasibility assessments. By partnering with NINGBO INNO PHARMCHEM, you secure a reliable supply chain partner committed to delivering cost-effective, high-quality chemical solutions that accelerate your drug development timelines.
