Advanced Copper-Catalyzed Synthesis of Aryl Trifluoromethyl Thioethers for Commercial Scale Manufacturing
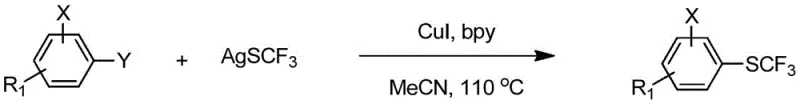
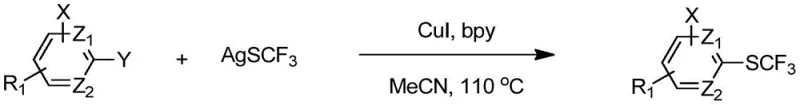
The pharmaceutical and agrochemical industries continuously seek robust methodologies to introduce fluorine-containing functional groups, particularly the trifluoromethylthio (-SCF3) moiety, due to its profound impact on lipophilicity and metabolic stability. Patent CN112939829B discloses a groundbreaking synthesis method for aryl trifluoromethyl sulfide compounds that addresses critical limitations in existing technologies. This innovation leverages a copper salt catalyst system combined with silver trifluoromethanesulfonate to achieve direct trifluoromethylthiolation of aryl halides under remarkably mild conditions. The technical breakthrough lies in the ability to efficiently utilize cheap and readily available aryl bromides and iodides, overcoming the historical reliance on expensive palladium catalysts or harsh fluorination reagents. By operating within a temperature range of 20-120 °C and utilizing common organic solvents, this process offers a sustainable pathway for generating high-value intermediates used in anticoccidial drugs like Toltrazuril and antihypertensive agents. The strategic implementation of nitrogen-containing organic ligands further stabilizes the active catalytic species, ensuring high yields and exceptional substrate universality across complex molecular architectures.

The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of the trifluoromethylthio group into aromatic systems has been plagued by significant technical and economic hurdles that hinder efficient commercial manufacturing. Traditional indirect methods often involve harsh reaction conditions that generate substantial amounts of waste acid water, creating severe environmental burdens and complicating waste treatment protocols for large-scale facilities. Alternative approaches relying on palladium catalysis, while effective for certain substrates, necessitate the use of prohibitively expensive noble metal catalysts and specialized ligands, driving up the overall cost of goods sold for final API products. Furthermore, earlier copper-catalyzed attempts were frequently limited to highly reactive aryl iodides or required the pre-formation of unstable metal complexes, failing to activate the more abundant and cost-effective aryl bromides. These legacy processes also suffered from narrow substrate scope, often failing when electron-withdrawing groups or heterocyclic rings were present, thereby restricting their utility in synthesizing modern, complex pharmaceutical intermediates required by global R&D teams.
The Novel Approach
The methodology described in patent CN112939829B represents a paradigm shift by enabling the direct coupling of aryl halides with silver trifluoromethanesulfonate using a simple yet highly effective copper catalytic system. This novel approach eliminates the need for expensive palladium sources and avoids the generation of hazardous waste streams associated with older fluorination techniques. A key differentiator is the system's remarkable ability to activate aryl bromides, which are typically less reactive than iodides, thus allowing manufacturers to utilize cheaper starting materials without sacrificing yield or purity. The reaction proceeds smoothly in common solvents such as acetonitrile or DMF at moderate temperatures, typically around 110 °C, making it inherently safer and easier to control in a production environment. By simply mixing the aryl halide, silver reagent, copper salt, and ligand, the process achieves high conversion rates across a diverse array of substrates, including those with sensitive functional groups, thereby streamlining the supply chain for critical drug intermediates.
Mechanistic Insights into Copper-Catalyzed Trifluoromethylthiolation
The core of this synthetic advancement relies on the synergistic interaction between the monovalent copper salt catalyst and the nitrogen-containing organic ligand, which facilitates the formation of an active copper-trifluoromethylthio species in situ. Mechanistically, the copper catalyst, such as cuprous iodide (CuI), coordinates with the ligand (e.g., 2,2'-bipyridine) to stabilize the reactive intermediate, preventing premature decomposition and enhancing the nucleophilicity of the trifluoromethylthio group. This stabilized complex then undergoes oxidative addition with the aryl halide substrate, a step that is traditionally rate-limiting for less reactive aryl bromides but is significantly accelerated in this optimized system. The subsequent reductive elimination releases the desired aryl trifluoromethyl thioether product and regenerates the copper catalyst, completing the catalytic cycle with high turnover efficiency. This mechanism explains the observed high yields even for sterically hindered or electron-deficient substrates, providing R&D directors with a reliable tool for late-stage functionalization of complex drug candidates without compromising structural integrity.
Impurity control is another critical aspect where this mechanism offers distinct advantages over conventional routes. The mild reaction conditions minimize side reactions such as homocoupling of the aryl halide or decomposition of the sensitive -SCF3 group, which are common issues in high-temperature or strongly basic environments. The use of silver trifluoromethanesulfonate as the sulfur source ensures a clean transfer of the trifluoromethylthio moiety, reducing the formation of sulfur-containing byproducts that are difficult to remove during purification. Furthermore, the compatibility of the system with various functional groups means that protecting group strategies can often be simplified or eliminated, reducing the total number of synthetic steps. For quality assurance teams, this translates to a cleaner crude reaction profile, facilitating easier downstream processing and ensuring that the final high-purity pharmaceutical intermediates meet stringent regulatory specifications for residual metals and organic impurities.
How to Synthesize Aryl Trifluoromethyl Thioether Efficiently
To implement this cutting-edge technology in a laboratory or pilot plant setting, operators must adhere to precise stoichiometric ratios and reaction parameters to maximize yield and safety. The process begins with the careful selection of the aryl halide substrate, where both bromides and iodides are viable, though iodides generally react faster. The molar ratio of silver trifluoromethanesulfonate to aryl halide is typically maintained between 1:1 and 5:1, with a preferred range of 1.1:1 to 5:1 to ensure complete conversion while minimizing excess reagent costs. The copper catalyst loading is optimized between 0.01 to 2 molar equivalents, with 0.5 to 1 equivalent being ideal for balancing reaction speed and economic efficiency. Detailed standard operating procedures regarding solvent selection, specifically favoring acetonitrile or 1,4-dioxane, and temperature control are essential for reproducibility. The following section outlines the standardized synthesis steps derived directly from the patent examples to guide your technical team in achieving consistent results.
- Mix aryl halide (bromide or iodide), silver trifluoromethanesulfonate, copper salt catalyst (e.g., CuI), and nitrogen-containing ligand (e.g., bipyridine) in an organic solvent like acetonitrile.
- Stir the reaction mixture at temperatures between 20-120 °C, preferably 100-120 °C, for a duration of 1 to 60 hours depending on substrate reactivity.
- Upon completion, quench with saturated ammonium chloride, filter, extract, dry, and purify the crude product via column chromatography to obtain the high-purity aryl trifluoromethyl thioether.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this copper-catalyzed synthesis route offers transformative benefits that directly impact the bottom line and operational resilience. The primary advantage stems from the substitution of expensive palladium catalysts with abundant and low-cost copper salts, which drastically reduces the raw material expenditure per kilogram of produced intermediate. Additionally, the ability to use aryl bromides, which are often significantly cheaper and more readily available in bulk quantities than their iodide counterparts, further drives down the cost of goods. This cost reduction is not merely theoretical; it is structurally embedded in the chemistry by eliminating the need for complex ligand synthesis or specialized reagent preparation, simplifying the procurement portfolio. Moreover, the mild reaction conditions reduce energy consumption and equipment wear, contributing to lower overhead costs and a smaller carbon footprint, aligning with corporate sustainability goals.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts removes the necessity for expensive metal scavenging steps and rigorous residual metal testing, which are significant cost drivers in API manufacturing. By utilizing a base metal catalyst system, the process inherently lowers the barrier to entry for producing high-value fluorinated intermediates, allowing for more competitive pricing in the global market. The simplified workup procedure, involving standard filtration and chromatography, reduces solvent usage and labor hours compared to multi-step traditional methods. Consequently, the overall manufacturing economics are improved, enabling the production of complex molecules like Toltrazuril intermediates at a fraction of the historical cost.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals such as cuprous iodide, bipyridine, and common aryl halides mitigates the risk of supply disruptions often associated with specialized fluorinating agents or noble metals. The broad substrate scope ensures that supply chain managers can source alternative starting materials if specific aryl halides face temporary shortages, as the process is robust enough to handle various substituents. This flexibility enhances the continuity of supply for critical pharmaceutical ingredients, reducing the risk of production delays due to raw material unavailability. Furthermore, the stability of the reagents allows for longer shelf life and easier storage logistics, simplifying inventory management for large-scale operations.
- Scalability and Environmental Compliance: The process is designed with industrial scalability in mind, operating at atmospheric pressure and moderate temperatures that are easily achievable in standard stainless steel reactors. The absence of hazardous gases or extreme conditions simplifies safety protocols and reduces the capital investment required for specialized containment equipment. From an environmental perspective, the reduction in waste acid generation and the use of recyclable solvents support compliance with increasingly stringent global environmental regulations. This green chemistry profile not only avoids potential regulatory fines but also enhances the brand reputation of the manufacturer as a sustainable partner in the pharmaceutical value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These answers are derived from the experimental data and technical specifications outlined in patent CN112939829B, providing clarity for stakeholders evaluating this technology for their production pipelines. Understanding these nuances is crucial for making informed decisions about process adoption and integration into existing manufacturing workflows.
Q: What are the primary advantages of this copper-catalyzed method over palladium-based systems?
A: This method utilizes inexpensive copper salts instead of costly palladium catalysts, significantly reducing raw material costs. Furthermore, it demonstrates superior activity towards less reactive aryl bromides compared to prior art copper complexes, expanding substrate universality for diverse pharmaceutical intermediates.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process operates under mild conditions (20-120 °C) without requiring extreme pressures or hazardous reagents. The use of common solvents like acetonitrile and commercially available aryl halides facilitates straightforward scale-up from laboratory to multi-ton commercial manufacturing.
Q: What types of substrates are compatible with this trifluoromethylthiolation protocol?
A: The method exhibits broad substrate scope, successfully converting various aryl iodides and challenging aryl bromides into trifluoromethyl thioethers. It tolerates diverse functional groups including nitro, ester, ketone, and heterocyclic moieties found in complex drug molecules like Toltrazuril and Losartan analogues.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryl Trifluoromethyl Thioether Supplier
As the demand for fluorinated intermediates continues to surge in the development of next-generation therapeutics, partnering with an experienced CDMO is essential for navigating the complexities of commercial scale-up. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific requirements of copper-catalyzed reactions, maintaining stringent purity specifications through our rigorous QC labs and advanced analytical capabilities. We understand that consistency and quality are paramount in the pharmaceutical industry, and our dedicated technical team is committed to delivering high-purity aryl trifluoromethyl thioethers that meet the exacting standards of global regulatory bodies.
We invite you to collaborate with us to leverage this innovative synthesis technology for your specific project needs. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your target molecule, demonstrating how this copper-catalyzed route can optimize your budget without compromising quality. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you accelerate your drug development timeline with reliable, cost-effective, and scalable chemical solutions that drive your business forward.
