Scalable Solution-Phase Synthesis of GHK Tripeptide and Blue Copper Peptide for Commercial Production
Scalable Solution-Phase Synthesis of GHK Tripeptide and Blue Copper Peptide for Commercial Production
The landscape of peptide manufacturing is undergoing a significant transformation driven by the urgent need for cost-effective and environmentally sustainable production methods. As detailed in the recent patent CN116143867A, a novel methodology has emerged that challenges the dominance of traditional solid-phase synthesis for producing the highly sought-after GHK tripeptide and its copper complex, blue copper peptide. This innovative approach摒弃s the reliance on expensive condensing agents and solid supports, opting instead for a streamlined solution-phase route that leverages specific aminolysis and ammonolysis reactions. For R&D directors and procurement managers in the cosmetic and pharmaceutical sectors, this represents a pivotal shift towards more efficient supply chains. The technology not only promises to lower the barrier to entry for high-purity peptide actives but also addresses the critical issue of waste reduction in fine chemical manufacturing, aligning perfectly with modern green chemistry initiatives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of GHK tripeptide has been dominated by solid-phase peptide synthesis (SPPS), a technique that, while robust for small batches, suffers from severe economic and logistical drawbacks when scaled. The conventional SPPS process requires the anchoring of the C-terminal amino acid to a polymeric resin, followed by iterative cycles of deprotection and coupling using potent condensing agents. These reagents, such as carbodiimides or phosphonium salts, are not only costly but also generate substantial amounts of hazardous byproducts that complicate downstream purification. Furthermore, the physical limitations of resin swelling restrict the concentration of reactants, leading to large solvent volumes and inefficient reactor utilization. For a procurement manager, this translates to volatile raw material costs and extended lead times due to the complexity of resin cleavage and freeze-drying steps, making it difficult to secure a reliable supply of cost-competitive active ingredients for mass-market cosmetic formulations.
The Novel Approach
In stark contrast, the method disclosed in patent CN116143867A introduces a sophisticated solution-phase strategy that completely circumvents the need for condensing agents and solid supports. This route utilizes a unique "latent glycine" precursor, where a chloroacetyl group is installed on the histidine intermediate to serve as the future N-terminal glycine. By employing thermal aminolysis to form the peptide bond between the histidine and lysine fragments, the process achieves high conversion rates without the auxiliary activation typically required. This fundamental change in synthetic logic allows for higher reactant concentrations and simpler work-up procedures, such as extraction and crystallization, rather than complex chromatographic separations. The result is a manufacturing protocol that is inherently more scalable and economically viable, offering a compelling alternative for companies seeking a reliable cosmetic peptide supplier capable of delivering bulk quantities without the premium price tag associated with legacy technologies.
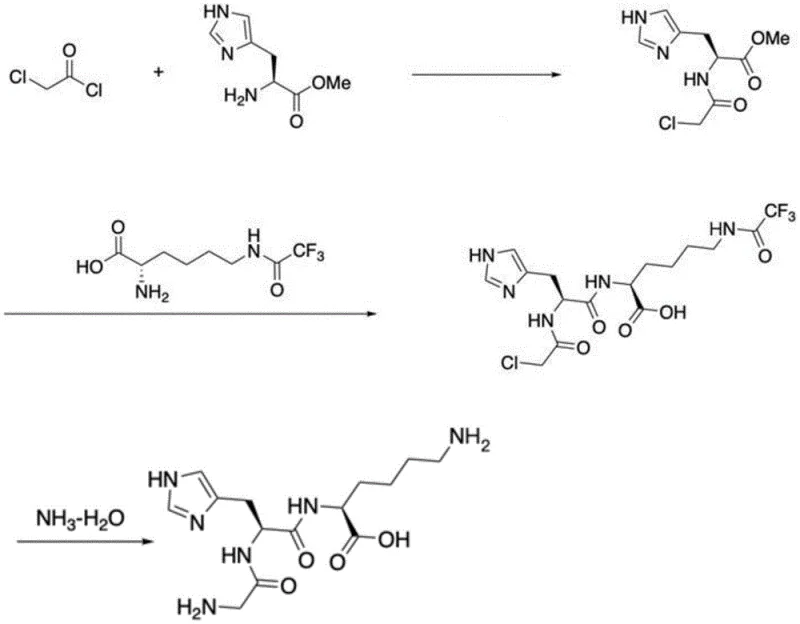
Mechanistic Insights into Solution-Phase Aminolysis and Ammonolysis
The core of this technological breakthrough lies in the precise manipulation of nucleophilic substitution and acyl transfer reactions under controlled thermal conditions. The first stage involves the acylation of methyl histidine with chloroacetyl chloride, creating a reactive chloroacetyl-histidine methyl ester intermediate. Unlike traditional coupling which activates the carboxyl group, this method activates the amine terminus with a functionality that can later be transformed. The subsequent step is particularly ingenious: the methyl ester of this intermediate acts as the electrophile, undergoing direct aminolysis by the alpha-amino group of trifluoroacetyl-protected lysine. Driven by elevated temperatures of 60-80°C and the presence of organic bases like triethylamine, this reaction forms the critical peptide bond while releasing methanol, a benign byproduct. This mechanism avoids the formation of racemization-prone activated esters, ensuring the stereochemical integrity of the histidine residue is maintained throughout the synthesis, which is paramount for the biological activity of the final blue copper peptide complex.
Following the assembly of the tripeptide backbone, the final transformation involves the conversion of the N-terminal chloroacetyl group into a glycine residue via ammonolysis. In this step, ammonia water serves as a potent nucleophile, attacking the methylene carbon adjacent to the carbonyl to displace the chloride ion. This reaction effectively extends the peptide chain by one glycine unit in situ, completing the GHK sequence without the need to couple a separate glycine amino acid. The use of ammonia water is not only cost-effective but also simplifies the removal of excess reagents, as unreacted ammonia can be easily evaporated or neutralized. This elegant cascade of reactions demonstrates a deep understanding of physical organic chemistry, allowing for the construction of complex peptide architectures using simple, commodity-grade starting materials and minimizing the generation of difficult-to-remove impurities that often plague peptide synthesis.
How to Synthesize GHK Tripeptide Efficiently
The implementation of this synthesis route requires careful control of reaction parameters to maximize yield and purity, particularly regarding pH and temperature management during the acylation and aminolysis steps. The process begins with the preparation of the chloroacetyl-histidine intermediate, followed by its coupling with the lysine derivative, and concludes with the ammonolysis step to reveal the final tripeptide. Each stage has been optimized to balance reaction kinetics with product stability, ensuring that the sensitive peptide bonds are not hydrolyzed while driving the equilibrium towards the desired product. For technical teams looking to adopt this methodology, understanding the specific stoichiometry and solvent choices is critical for successful technology transfer from the laboratory to the pilot plant. The detailed standardized synthesis steps see the guide below.
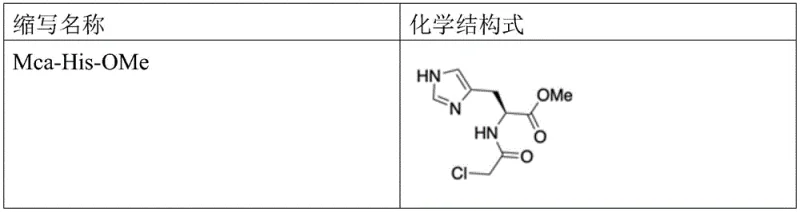
- React methyl histidine with chloroacetyl chloride under basic conditions to form Mca-His-OMe.
- Perform thermal aminolysis of Mca-His-OMe with trifluoroacetyl lysine to establish the peptide bond.
- Execute ammonolysis using ammonia water to convert the chloroacetyl group into the terminal glycine residue.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement specialists, the shift from solid-phase to this novel solution-phase chemistry offers profound strategic advantages that extend beyond simple unit cost savings. The elimination of expensive coupling reagents and polymeric resins removes two of the most significant cost drivers in peptide manufacturing, directly impacting the bottom line. Moreover, the simplified purification workflow, which relies on crystallization and extraction rather than preparative HPLC, significantly reduces solvent consumption and processing time. This efficiency gain translates into a more robust supply chain capable of responding rapidly to market fluctuations in demand for anti-aging actives. By adopting this technology, manufacturers can secure a more stable source of high-purity GHK tripeptide, mitigating the risks associated with the supply volatility of specialized solid-phase reagents and ensuring consistent quality for downstream formulation.
- Cost Reduction in Manufacturing: The most immediate impact of this technology is the drastic reduction in raw material expenses associated with peptide coupling. By removing the requirement for stoichiometric amounts of condensing agents like HBTU or PyBOP, which are often priced at a premium, the variable cost of goods sold is significantly lowered. Additionally, the ability to recover and recycle solvents like tetrahydrofuran and dioxane further enhances the economic profile of the process. This cost structure allows suppliers to offer competitive pricing models, making high-end peptide actives accessible for a broader range of cosmetic and dermatological products without compromising on margin.
- Enhanced Supply Chain Reliability: Dependence on specialized solid-phase resins and coupling reagents often creates bottlenecks in the supply chain, as these materials may have long lead times or limited global availability. This new method utilizes commodity chemicals such as chloroacetyl chloride, amino acid esters, and ammonia, which are readily available from multiple global sources. This diversification of the raw material base ensures continuous production capability even during periods of market disruption. Furthermore, the solution-phase nature of the reaction allows for the use of standard glass-lined or stainless steel reactors, removing the need for specialized solid-phase synthesis equipment and facilitating easier scale-up across different manufacturing sites.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this process offers a cleaner alternative to traditional peptide synthesis. The absence of heavy metal catalysts or complex coupling byproducts simplifies waste treatment and reduces the burden on effluent processing systems. The high atom economy of the aminolysis and ammonolysis steps means that a greater proportion of the starting materials end up in the final product, minimizing waste generation. This alignment with green chemistry principles not only reduces disposal costs but also enhances the sustainability profile of the final ingredient, a factor that is increasingly important for brand owners seeking eco-friendly certifications for their cosmetic lines.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this condensing-agent-free synthesis route. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for evaluating the technology's potential impact on your supply chain. Understanding these nuances is essential for making informed decisions about sourcing strategies and process development investments.
Q: Why is eliminating condensing agents beneficial for GHK production?
A: Traditional solid-phase synthesis relies heavily on expensive coupling reagents like HBTU or HATU, which generate significant chemical waste and increase raw material costs. By utilizing a solution-phase approach that leverages thermal aminolysis and ammonolysis, this method removes the need for these costly activators, drastically simplifying the purification process and reducing the overall environmental footprint of the manufacturing cycle.
Q: How is the glycine residue formed without adding glycine directly?
A: The process employs a clever 'latent glycine' strategy where a chloroacetyl group is initially attached to the histidine N-terminus. In the final step, ammonia water acts as a nucleophile to displace the chlorine atom via ammonolysis, effectively converting the chloroacetyl moiety into a glycine residue in situ, thereby streamlining the synthetic sequence.
Q: Is this method suitable for large-scale industrial manufacturing?
A: Yes, the transition from solid-phase to solution-phase chemistry inherently improves scalability by eliminating the need for expensive resin supports and the associated swelling limitations. The use of common organic solvents and straightforward thermal conditions allows for easy adaptation to standard reactor vessels, facilitating the commercial mass production required to meet global demand for blue copper peptides.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable GHK Tripeptide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this advanced synthesis technology for the global cosmetics and pharmaceutical markets. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a GMP-compliant manufacturing environment. Our rigorous QC labs and stringent purity specifications guarantee that every batch of GHK tripeptide or blue copper peptide meets the highest international standards, providing our partners with the confidence needed to launch premium skincare formulations. We are committed to bridging the gap between innovative academic research and industrial reality, delivering complex peptide actives with unmatched consistency and reliability.
We invite forward-thinking procurement directors and R&D leaders to collaborate with us to leverage this cost-effective synthesis route for their next-generation products. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing you to evaluate how this cutting-edge technology can optimize your supply chain and enhance your product portfolio's competitiveness in the rapidly evolving beauty and wellness industry.
