Advanced Synthesis of Four-Coordinate N,N-Chelated Diarylboronate Compounds for Commercial Scale-up
Introduction to Advanced Boronate Ester Technology
The landscape of fine chemical manufacturing is continuously evolving, driven by the demand for more stable and versatile intermediates capable of supporting complex downstream applications in optoelectronics and pharmaceuticals. Patent CN111620896A introduces a groundbreaking methodology for the preparation of four-coordinate N,N-chelated diarylboronate compounds, specifically utilizing 8-aminoquinoline derivatives as bidentate ligands. This technology addresses critical bottlenecks in the synthesis of AQDAB (8-aminoquinoline diarylboronic acid ester) structures, which are pivotal precursors for high-performance dyes, fluorescent probes, and photocatalysts. By shifting away from traditional, highly reactive organometallic reagents towards stable potassium aryl trifluoroborates, this invention establishes a new standard for safety and operational simplicity in the production of specialized boron-containing materials. The strategic implementation of this synthetic route allows manufacturers to access a diverse library of functionalized boronates that were previously difficult or dangerous to produce at scale.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
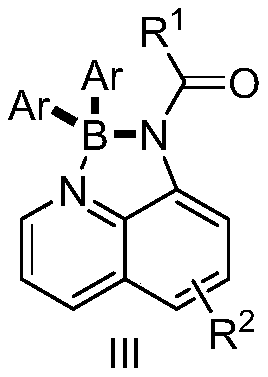
Historically, the construction of four-coordinate N,N-chelated diarylboronate skeletons has been plagued by significant synthetic challenges that hinder efficient commercial production. Traditional protocols predominantly rely on the use of dipyrrole structures or require the introduction of aryl groups via highly reactive organometallic species such as Grignard reagents, organolithiums, or organozinc compounds. These conventional reagents possess extreme sensitivity to moisture and oxygen, necessitating rigorous anhydrous conditions and often cryogenic temperatures to prevent decomposition or side reactions. Furthermore, the strong basicity and nucleophilicity inherent to these metal reagents severely limit functional group compatibility, rendering common moieties like esters, aldehydes, ketones, and amides incompatible with the reaction environment. This lack of chemoselectivity forces chemists to employ lengthy protection and deprotection sequences, drastically reducing overall yield and increasing the environmental footprint of the manufacturing process through excessive solvent and reagent consumption.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent leverages the unique stability and reactivity profile of potassium aryl trifluoroborates to achieve high-efficiency coupling under much milder conditions. This innovative strategy employs 8-aminoquinoline derivatives as robust bidentate ligands that coordinate with the boron center to form stable four-coordinate complexes, effectively locking the geometry and enhancing the photophysical properties of the final product. The reaction proceeds smoothly in the presence of inexpensive transition metal promoters like manganese or iron, along with organic additives, at elevated temperatures ranging from 110 to 140 degrees Celsius without the need for inert atmosphere gloveboxes. This paradigm shift not only simplifies the operational workflow but also dramatically expands the substrate scope, allowing for the direct incorporation of sensitive functional groups such as halogens, cyano groups, and ethers directly onto the aryl rings.
Mechanistic Insights into Transition Metal Promoted C-B Bond Formation
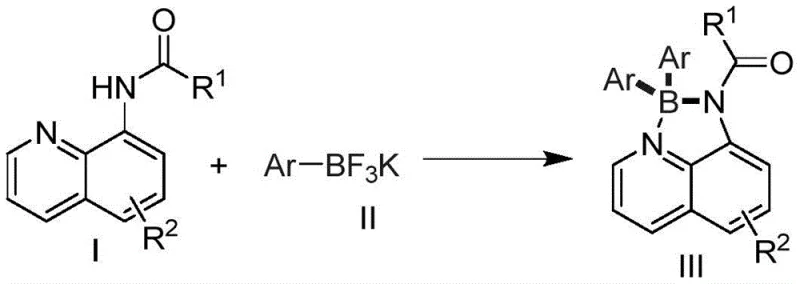
The core mechanistic advantage of this technology lies in the synergistic interaction between the stable boron source and the transition metal promoter, which facilitates a transmetallation pathway that bypasses the harsh conditions of classical organometallic chemistry. The 8-aminoquinoline ligand plays a dual role: initially acting as a directing group to assist in the activation of the boron species, and subsequently serving as a chelating arm that stabilizes the resulting tetra-coordinate boron center against hydrolysis. The metal promoter, whether it be elemental manganese, iron, copper, or zinc, likely undergoes a single-electron transfer or oxidative addition process to activate the carbon-boron bond of the trifluoroborate salt, generating a reactive organometallic intermediate in situ. This intermediate then attacks the electrophilic center or coordinates with the nitrogen atoms of the quinoline scaffold to close the chelate ring, forming the thermodynamically stable AQDAB structure. This mechanism ensures high selectivity for the desired four-coordinate product while minimizing the formation of tri-coordinate boronic acid byproducts that often complicate purification in other synthetic routes.
From an impurity control perspective, the use of potassium trifluoroborates significantly reduces the generation of homocoupling byproducts that are typical of radical-based organometallic reactions. The reaction conditions are tuned to favor the cross-coupling event between the specific aryl group of the trifluoroborate and the quinoline nitrogen-boron complex, ensuring a clean impurity profile that is essential for electronic grade materials. The stability of the starting materials means that degradation products arising from reagent hydrolysis are virtually eliminated, leading to a crude reaction mixture that is easier to purify via standard silica gel chromatography. This level of control over the reaction trajectory is critical for maintaining the high purity specifications required in the manufacture of OLED materials and fluorescent sensors, where trace metal contaminants or structural isomers can quench luminescence or alter emission wavelengths.
How to Synthesize Four-Coordinate N,N-Chelated Diarylboronate Efficiently
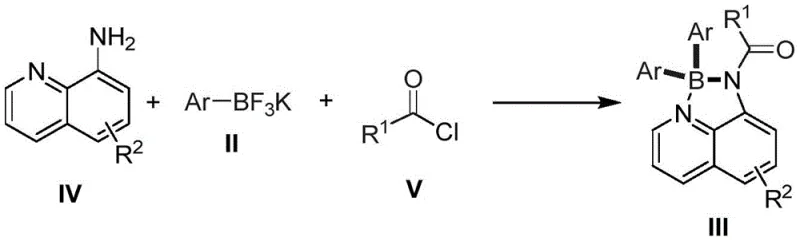
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to the stoichiometric ratios and thermal parameters defined in the patent to maximize yield and reproducibility. The process is designed to be flexible, accommodating both a stepwise approach using pre-acylated quinoline derivatives and a more streamlined one-pot procedure that combines the amine, acyl chloride, and boron source simultaneously. Operators should ensure that the reaction solvent, typically acetonitrile, toluene, or dioxane, is adequately degassed if using sensitive metal powders, although the protocol demonstrates remarkable tolerance to air compared to Grignard chemistry. The following guide outlines the standardized operational steps derived from the exemplary embodiments, providing a clear roadmap for technical teams to replicate the high yields reported in the intellectual property documentation.
- Combine 8-aminoquinoline derivative, potassium aryl trifluoroborate, base, additive, and metal promoter in a reaction solvent such as acetonitrile or toluene.
- Heat the reaction mixture to a temperature range of 110 to 140 degrees Celsius and maintain stirring for a duration of 12 to 48 hours to ensure complete conversion.
- Upon completion, filter the mixture, wash with dichloromethane, remove solvents via rotary evaporation, and purify the crude product using silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic methodology represents a strategic opportunity to optimize raw material costs and mitigate supply risks associated with volatile organometallic reagents. The substitution of expensive and hazardous Grignard or lithium reagents with shelf-stable potassium aryl trifluoroborates eliminates the need for specialized storage infrastructure and reduces the frequency of reagent replacement due to degradation. This shift translates into substantial cost savings in warehousing and inventory management, as the solid boron salts can be stored for extended periods without loss of potency, ensuring a consistent supply of key starting materials even during market fluctuations. Furthermore, the ability to run reactions at atmospheric pressure and moderate temperatures reduces the energy load on manufacturing facilities, contributing to lower utility costs and a smaller carbon footprint per kilogram of produced intermediate.
- Cost Reduction in Manufacturing: The elimination of cryogenic cooling systems and the use of inexpensive earth-abundant metal promoters like manganese or iron significantly lower the capital expenditure and operational costs associated with reactor maintenance. By avoiding the multi-step protection strategies required for sensitive functional groups in traditional routes, the overall step count is reduced, which directly correlates to higher throughput and lower labor costs per batch. The high atom economy of the one-pot variant further minimizes waste disposal fees, as fewer byproducts and solvent volumes are generated during the synthesis of complex diarylboronate structures.
- Enhanced Supply Chain Reliability: Potassium aryl trifluoroborates are commercially available for a wide range of aryl substituents, providing procurement teams with a diverse and reliable sourcing network that is less susceptible to geopolitical supply chain disruptions than specialized organometallics. The robustness of the reaction conditions means that production schedules are less likely to be delayed by environmental factors such as humidity spikes, which frequently halt operations in facilities relying on moisture-sensitive chemistry. This reliability ensures consistent lead times for downstream customers in the pharmaceutical and agrochemical sectors who depend on just-in-time delivery of high-purity intermediates for their own synthesis campaigns.
- Scalability and Environmental Compliance: The simplified workup procedure, involving basic filtration and standard chromatography, is inherently scalable from gram-scale laboratory experiments to multi-ton commercial production without requiring complex engineering modifications. The use of low-toxicity metal salts aligns with increasingly stringent environmental regulations regarding heavy metal residues in active pharmaceutical ingredients and electronic materials, facilitating easier regulatory approval and waste stream management. This green chemistry approach enhances the corporate sustainability profile of manufacturers, making the final products more attractive to eco-conscious global brands seeking to reduce the environmental impact of their supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented synthesis technology, providing clarity for R&D teams evaluating its fit for their specific project requirements. These answers are derived directly from the experimental data and claims within the patent documentation, ensuring accuracy and relevance for process development discussions. Understanding these nuances is essential for assessing the feasibility of adapting this chemistry to novel substrates or integrating it into existing manufacturing workflows for fine chemical intermediates.
Q: What are the advantages of using potassium trifluoroborates over Grignard reagents in this synthesis?
A: Potassium trifluoroborates offer superior stability against moisture and air compared to Grignard reagents, allowing for broader functional group tolerance including esters and nitriles without requiring cryogenic conditions.
Q: Can this method be adapted for a one-pot synthesis strategy?
A: Yes, the patent describes a three-component one-pot method where the 8-aminoquinoline derivative, acyl chloride, and potassium trifluoroborate are reacted simultaneously, significantly improving atom economy.
Q: What types of metal promoters are effective for this transformation?
A: The process utilizes inexpensive and low-toxicity transition metals such as manganese, iron, copper, or zinc, either as elemental powders or salts like manganese acetate, to facilitate the cross-coupling reaction.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Four-Coordinate N,N-Chelated Diarylboronate Supplier
As a premier CDMO partner, NINGBO INNO PHARMCHEM possesses the technical expertise and infrastructure necessary to translate this advanced patent technology into commercial reality for global clients. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of four-coordinate N,N-chelated diarylboronate meets the exacting standards required for optoelectronic and pharmaceutical applications. Our commitment to quality assurance means that we can deliver materials with consistent impurity profiles, enabling our partners to achieve reproducible results in their downstream device fabrication or drug synthesis processes.
We invite potential collaborators to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific molecular targets and volume requirements. By requesting a Customized Cost-Saving Analysis, you can gain valuable insights into the economic benefits of switching to this stable and efficient manufacturing platform. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that will enhance the competitiveness and sustainability of your supply chain for high-value fine chemical intermediates.
