Advanced Ruthenium-Catalyzed C-H Silylation for Scalable Pharmaceutical Intermediate Manufacturing
Advanced Ruthenium-Catalyzed C-H Silylation for Scalable Pharmaceutical Intermediate Manufacturing
The rapid evolution of medicinal chemistry has placed silicon-containing heterocycles at the forefront of drug discovery, particularly for their enhanced metabolic stability and lipophilicity profiles. Among these, 2-aryl ortho-substituted triethylsilylpyridine compounds represent a critical class of building blocks for developing novel antiviral and antitumor agents. However, traditional synthetic routes have often been plagued by inefficiencies and high costs. The recent disclosure in patent CN109651421B introduces a transformative approach utilizing a ruthenium-catalyzed C-H activation strategy. This method enables the direct ortho-silylation of 2-arylpyridine derivatives using triethylsilane in a single pot. By shifting away from expensive noble metals like rhodium and iridium, this technology offers a robust pathway for the commercial scale-up of complex pharmaceutical intermediates, addressing both economic and operational challenges faced by modern R&D departments.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of silylated pyridine derivatives relied heavily on electrophilic substitution reactions involving reagents such as trimethylsilyl trifluoromethanesulfonate (TMSOTf) or trimethylchlorosilane (TMSCl). These classical approaches suffer from significant drawbacks, including multi-step sequences that generate substantial amounts of inorganic salt waste, thereby complicating downstream processing and environmental compliance. Furthermore, these methods often exhibit poor chemical selectivity and limited functional group compatibility, leading to difficult-to-separate impurity profiles that compromise the purity required for high-purity pharmaceutical intermediates. While transition metal-catalyzed C-H activation using rhodium or iridium complexes improved efficiency, the prohibitive cost and scarcity of these metals created a bottleneck for industrial adoption, making the cost reduction in pharmaceutical intermediate manufacturing a critical priority for procurement teams.
The Novel Approach
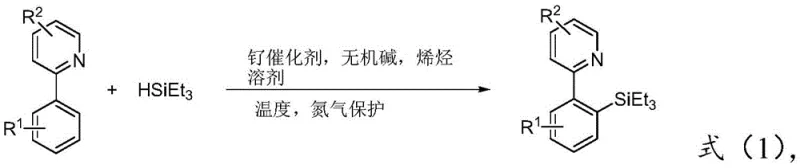
The innovative methodology outlined in the patent data overcomes these barriers by employing a relatively inexpensive ruthenium complex catalyst in conjunction with an inorganic base and an unsaturated olefin. This system facilitates the direct coupling of 2-arylpyridines with triethylsilane under heating conditions, achieving high yields through a streamlined one-pot process. As illustrated in the general reaction scheme below, the process eliminates the need for intermediate isolation, significantly reducing labor and solvent inputs. This advancement not only enhances the economic viability of producing these scaffolds but also aligns with green chemistry principles by minimizing waste generation. For supply chain managers, this translates to a more resilient sourcing strategy for key organic building blocks.
Mechanistic Insights into Ru-Catalyzed C-H Silylation
The core of this technological breakthrough lies in the unique catalytic cycle driven by the ruthenium center, which effectively activates the inert ortho-C-H bond of the 2-arylpyridine substrate. The nitrogen atom of the pyridine ring acts as a native directing group, coordinating with the ruthenium catalyst to position the metal center in proximity to the target carbon-hydrogen bond. In the presence of an inorganic base, such as potassium acetate, and a hydrogen acceptor like norbornene or cyclohexene, the catalyst facilitates the cleavage of the C-H bond and the subsequent formation of the C-Si bond with triethylsilane. This mechanism ensures exceptional regioselectivity, predominantly yielding the ortho-substituted product while suppressing the formation of meta- or para-isomers. Such precision is vital for R&D directors who require consistent impurity profiles to accelerate regulatory filings and ensure batch-to-batch reproducibility in clinical supply chains.
Furthermore, the choice of ligands and additives plays a pivotal role in stabilizing the active catalytic species and promoting turnover. The use of tris(triphenylphosphine)carbonylruthenium hydrochloride, as preferred in the patent examples, provides a balance of stability and reactivity that allows the reaction to proceed at moderate temperatures around 120°C. The inclusion of unsaturated olefins serves to regenerate the active catalyst by removing hydrogen equivalents, driving the equilibrium towards product formation. This sophisticated interplay of components results in a robust process capable of tolerating various substituents on the aryl ring, including electron-donating groups like methyl and methoxy, as well as electron-withdrawing groups like trifluoromethyl. The specific transformation of 2-phenylpyridine shown below exemplifies the high efficiency and selectivity achievable with this system.

How to Synthesize 2-Aryl Ortho-Substituted Triethyl Silicon Pyridine Efficiently
Implementing this synthesis route requires careful attention to reaction parameters to maximize yield and minimize side reactions. The process begins with the precise weighing of 2-arylpyridine substrates and triethylsilane, followed by the addition of the ruthenium catalyst, inorganic base, and olefin additive in a suitable solvent such as toluene. The reaction vessel must be purged with nitrogen to maintain an oxygen-free environment, which is crucial for preventing catalyst deactivation and ensuring safety when handling silanes. Heating the mixture to 120°C with vigorous stirring allows the catalytic cycle to proceed to completion over a period of approximately 16 hours. Detailed standardized operating procedures regarding stoichiometry, workup, and purification are essential for technology transfer.
- Combine 2-arylpyridine, triethylsilane, inorganic base (e.g., potassium acetate), unsaturated olefin (e.g., norbornene), and ruthenium catalyst in a solvent like toluene.
- Heat the reaction mixture to 120°C under nitrogen protection with electromagnetic stirring for approximately 16 hours.
- Remove solvent by rotary evaporation and purify the crude product via column chromatography to obtain the target silylated compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this ruthenium-catalyzed protocol offers tangible strategic benefits beyond mere technical feasibility. The shift from scarce precious metals to abundant ruthenium significantly mitigates the risk of price volatility associated with rhodium and iridium markets. Moreover, the one-pot nature of the reaction drastically simplifies the manufacturing workflow, eliminating multiple isolation and purification steps that typically consume time and resources. This streamlining leads to substantial cost savings in terms of solvent usage, energy consumption, and labor hours, directly impacting the bottom line. Additionally, the high selectivity of the reaction reduces the burden on quality control laboratories, as fewer impurities need to be monitored and removed, thereby accelerating the release of materials for downstream synthesis.
- Cost Reduction in Manufacturing: The utilization of a low-cost ruthenium catalyst instead of expensive rhodium or iridium complexes fundamentally alters the cost structure of the synthesis. By reducing the catalyst loading and eliminating the need for costly protecting group strategies or harsh electrophilic reagents, the overall production cost is significantly lowered. Furthermore, the ability to run the reaction in a single pot without intermediate isolation minimizes solvent waste and reduces the physical footprint required for manufacturing, contributing to a leaner and more economical production model that enhances competitiveness in the global market.
- Enhanced Supply Chain Reliability: The reagents required for this synthesis, including triethylsilane, 2-arylpyridines, and common inorganic bases, are widely available commodity chemicals with stable supply chains. Unlike specialized catalysts that may have long lead times or single-source dependencies, the components of this reaction system can be sourced from multiple vendors, ensuring continuity of supply even during market disruptions. This diversification of the supply base reduces the risk of production delays and allows for more accurate forecasting and inventory management, which is critical for meeting the demanding timelines of pharmaceutical development projects.
- Scalability and Environmental Compliance: The simplicity of the workup procedure, which typically involves solvent removal followed by column chromatography, makes this process highly amenable to scale-up from laboratory to pilot and commercial scales. The reduction in inorganic salt waste and the avoidance of hazardous electrophilic silylating agents align with increasingly stringent environmental regulations. This eco-friendly profile not only reduces disposal costs but also enhances the corporate sustainability image, making the resulting intermediates more attractive to environmentally conscious partners and regulators who prioritize green manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear understanding of the process capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing platforms.
Q: What are the advantages of using Ruthenium over Rhodium or Iridium for this synthesis?
A: Ruthenium complexes are significantly more cost-effective than Rhodium or Iridium catalysts, reducing raw material costs without compromising catalytic activity. Additionally, the Ru-catalyzed process described in CN109651421B operates efficiently under standard heating conditions with high functional group tolerance.
Q: Does this method require complex purification steps for intermediates?
A: No, the process is designed as a one-pot synthesis. Except for the final product, intermediates generated during the conversion process do not require separation or purification, which drastically simplifies the workflow and reduces solvent consumption.
Q: What is the typical reaction temperature and time for this silylation?
A: The optimal reaction conditions involve heating the mixture to approximately 120°C for about 16 hours under a nitrogen atmosphere, ensuring complete conversion while maintaining safety and stability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Aryl Ortho-Substituted Triethylsilylpyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes in the fast-paced pharmaceutical industry. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to full-scale manufacturing is seamless and reliable. We are committed to delivering stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 2-aryl ortho-substituted triethylsilylpyridine meets the highest standards required for drug substance synthesis. Our state-of-the-art facilities are equipped to handle the specific requirements of organometallic catalysis, including inert atmosphere operations and precise temperature control.
We invite potential partners to engage with our technical procurement team to discuss how this advanced ruthenium-catalyzed technology can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic benefits specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive innovation and efficiency in your drug development programs.
