Advanced Synthesis of Dabigatran Etexilate Intermediates for Commercial Scale-Up
Advanced Synthesis of Dabigatran Etexilate Intermediates for Commercial Scale-Up
The pharmaceutical landscape for anticoagulant therapies has been significantly shaped by the introduction of direct thrombin inhibitors, with dabigatran etexilate standing out as a cornerstone treatment for preventing stroke in patients with atrial fibrillation. As detailed in patent CN105523999B, recent advancements in synthetic methodology have unlocked a more efficient pathway for producing the critical intermediates required for this high-value active pharmaceutical ingredient. This technical insight report analyzes the breakthrough synthesis of the key intermediate, specifically the compound of formula (5), which serves as the structural backbone for the final drug substance. The patent discloses a robust process that reacts a halomethyl-benzimidazole derivative (formula 2) with a protected amidino-phenyl compound (formula 4) to construct the complex molecular architecture with superior efficiency. 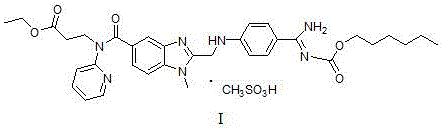 Understanding the precise chemical connectivity shown in Formula I is essential for R&D directors aiming to replicate or license this technology for commercial production, as it highlights the specific junction points where process optimization yields the greatest return on investment.
Understanding the precise chemical connectivity shown in Formula I is essential for R&D directors aiming to replicate or license this technology for commercial production, as it highlights the specific junction points where process optimization yields the greatest return on investment.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of dabigatran etexilate has been plagued by inefficient synthetic routes that rely on cumbersome coupling strategies and harsh reaction conditions. As illustrated in the prior art route from patent WO9837075, the traditional method involves the condensation of an amino-benzoyl propionate with N-(4-cyanophenyl) glycine using expensive coupling agents like EDCI and HOBT. 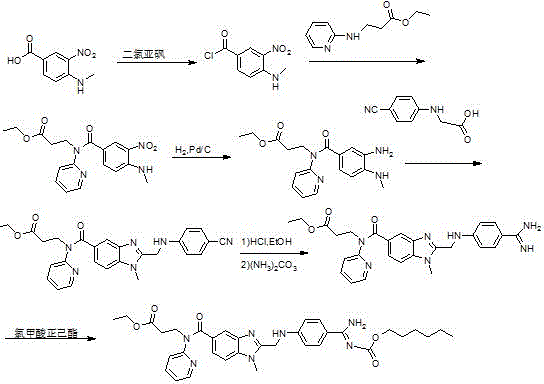 This conventional approach suffers from critically low yields, often hovering around 50 percent during the benzimidazole derivative formation, necessitating purification by column chromatography which is impractical for multi-kilogram manufacturing. Furthermore, the subsequent conversion of the cyano group to the required amidino functionality typically involves hydrolysis in ethanol saturated with hydrogen chloride, a process that is operationally complex and generates substantial quantities of hazardous waste acid. These bottlenecks not only inflate the cost of goods sold but also introduce significant supply chain risks due to the reliance on specialized reagents and the generation of difficult-to-treat effluent streams.
This conventional approach suffers from critically low yields, often hovering around 50 percent during the benzimidazole derivative formation, necessitating purification by column chromatography which is impractical for multi-kilogram manufacturing. Furthermore, the subsequent conversion of the cyano group to the required amidino functionality typically involves hydrolysis in ethanol saturated with hydrogen chloride, a process that is operationally complex and generates substantial quantities of hazardous waste acid. These bottlenecks not only inflate the cost of goods sold but also introduce significant supply chain risks due to the reliance on specialized reagents and the generation of difficult-to-treat effluent streams.
The Novel Approach
In stark contrast, the methodology disclosed in CN105523999B introduces a paradigm shift by streamlining the construction of the benzimidazole core and the subsequent assembly of the final intermediate. The novel approach utilizes a direct cyclization strategy where 3-amino-4-methylaminobenzoic acid reacts with chloroacetic anhydride or chloroacetic acid to form the benzimidazole carboxylic acid derivative with yields exceeding 90 percent. 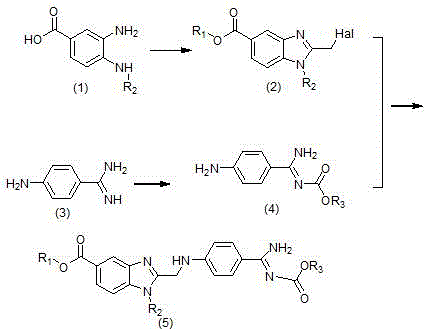 This eliminates the need for expensive condensing agents such as CDI during the critical ring-closing step, which is a common pain point in alternative syntheses described in documents like WO2007071743. By pre-forming the stable benzimidazole halide intermediate and reacting it with a pre-protected amidino compound in a mixed solvent system of organic solvent and water, the process achieves high conversion rates under mild thermal conditions. This strategic simplification reduces the total number of unit operations and avoids the formation of difficult-to-remove impurities, thereby enhancing the overall throughput and economic viability of the manufacturing process for global supply chains.
This eliminates the need for expensive condensing agents such as CDI during the critical ring-closing step, which is a common pain point in alternative syntheses described in documents like WO2007071743. By pre-forming the stable benzimidazole halide intermediate and reacting it with a pre-protected amidino compound in a mixed solvent system of organic solvent and water, the process achieves high conversion rates under mild thermal conditions. This strategic simplification reduces the total number of unit operations and avoids the formation of difficult-to-remove impurities, thereby enhancing the overall throughput and economic viability of the manufacturing process for global supply chains.
Mechanistic Insights into Benzimidazole Cyclization and Coupling
The core innovation of this patent lies in the mechanistic efficiency of the benzimidazole ring formation and the subsequent nucleophilic substitution. The reaction begins with the condensation of the diamine precursor (formula 1) with a haloacetic anhydride, such as chloroacetic anhydride, in an aprotic organic solvent like ethyl acetate. The mechanism proceeds through an initial amide formation followed by an intramolecular cyclodehydration to close the imidazole ring, driven by the thermal energy provided at temperatures ranging from 40 ℃ to the boiling point of the solvent. This specific pathway is highly favorable because it avoids the formation of oligomeric byproducts often seen when using carbodiimide-based coupling reagents. The resulting 2-halomethyl-benzimidazole (formula 2) possesses a highly reactive electrophilic center at the methyl position, which is perfectly poised for the subsequent alkylation step. The use of chloroacetic anhydride ensures that the leaving group is a chloride ion, which provides an optimal balance between reactivity and stability, preventing premature decomposition of the intermediate during isolation and storage.
Furthermore, the protection strategy for the amidino group plays a pivotal role in the success of the final coupling reaction. By reacting 4-amidinophenylamine with n-hexyl chloroformate in the presence of a base like sodium hydroxide, the highly basic amidino nitrogen is selectively protected as a carbamate (formula 4). This protection is crucial because it prevents the amidino group from interfering with the benzimidazole formation or undergoing unwanted side reactions during the coupling phase. When the protected amidino compound (formula 4) reacts with the benzimidazole halide (formula 2) in a mixed solvent system containing tetrahydrofuran and water, the secondary amine of the aniline moiety acts as a nucleophile, displacing the halide on the benzimidazole methyl group. The presence of water in the solvent mixture is a subtle but critical parameter that likely aids in solubilizing the inorganic bases used to scavenge the generated acid, while the organic component ensures the solubility of the lipophilic intermediates. This biphasic or mixed-solvent environment facilitates a clean SN2 substitution, resulting in the high-purity intermediate (formula 5) required for the final API synthesis.
How to Synthesize Dabigatran Intermediate Efficiently
The synthesis of the key dabigatran intermediate described in this patent offers a clear roadmap for process chemists aiming to establish a robust manufacturing line. The procedure leverages readily available starting materials and standard reactor configurations, making it highly accessible for contract development and manufacturing organizations (CDMOs). The initial step involves the cyclization of the diamine with chloroacetic anhydride in ethyl acetate, followed by a simple filtration and drying process to isolate the benzimidazole acid in high purity. Subsequent steps involve the protection of the amidino partner and the final coupling in a THF-water mixture. For a comprehensive understanding of the specific stoichiometric ratios, temperature profiles, and workup procedures required to replicate these results, please refer to the standardized synthesis guide below.
- Cyclize 3-amino-4-methylaminobenzoic acid with chloroacetic anhydride in ethyl acetate at elevated temperatures to form the benzimidazole carboxylic acid core with high yield.
- Protect the amidino group of 4-amidinophenylamine by reacting with n-hexyl chloroformate in acetone under alkaline conditions to generate the stable carbamate derivative.
- Condense the benzimidazole halide intermediate with the protected amidino compound in a mixed solvent of tetrahydrofuran and water to obtain the final key intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route translates into tangible strategic advantages that extend beyond mere technical feasibility. The primary value driver is the drastic simplification of the production workflow, which directly correlates to reduced operational expenditures and enhanced supply security. By eliminating the dependency on scarce or expensive coupling reagents like CDI and EDCI, manufacturers can insulate their production costs from the volatility of the specialty chemicals market. Moreover, the significant improvement in reaction yields, particularly in the benzimidazole formation step where yields jump from roughly 50 percent in older methods to over 90 percent in this new process, means that less raw material is required to produce the same amount of finished intermediate. This material efficiency is a powerful lever for cost reduction in high-volume pharmaceutical manufacturing, allowing for more competitive pricing structures in tender negotiations.
- Cost Reduction in Manufacturing: The elimination of expensive condensing agents and the avoidance of column chromatography purification steps result in a substantially lower cost of goods sold. The process relies on commodity chemicals such as chloroacetic anhydride and n-hexyl chloroformate, which are widely available and cost-effective compared to the specialized reagents required by prior art methods. Additionally, the high yield of the cyclization step minimizes the loss of valuable starting materials, ensuring that the maximum amount of input mass is converted into saleable product. This efficiency gain allows manufacturers to operate with thinner margins while maintaining profitability, a critical factor in the highly competitive generic pharmaceutical intermediates market.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route enhances supply chain continuity by reducing the risk of batch failures and production delays. Conventional methods that involve multiple low-yield steps and complex purifications are prone to variability, which can lead to stockouts and missed delivery windows. In contrast, the high-yielding and operationally simple nature of the patented process ensures consistent output quality and quantity. The use of stable intermediates that do not require cryogenic storage or inert atmosphere handling further simplifies logistics and warehousing requirements. This reliability is paramount for securing long-term contracts with major pharmaceutical companies that demand uninterrupted supply of critical anticoagulant ingredients.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this method offers a cleaner production profile that aligns with increasingly stringent global sustainability standards. The avoidance of harsh acidic hydrolysis conditions significantly reduces the generation of hazardous waste streams, lowering the costs associated with effluent treatment and disposal. The simplified workup procedures, which often involve straightforward filtration and crystallization rather than complex extractions or chromatography, reduce solvent consumption and energy usage. These factors make the process highly scalable from pilot plant to commercial tonnage production without encountering the environmental bottlenecks that often plague older synthetic technologies, ensuring long-term regulatory compliance and social license to operate.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer and for commercial teams assessing the competitive positioning of the resulting product in the global marketplace.
Q: What are the yield advantages of this new synthesis method compared to prior art?
A: The patented method achieves yields exceeding 90% for the benzimidazole derivative synthesis, significantly outperforming conventional methods that often struggle with yields around 50% to 75% due to complex coupling requirements.
Q: Does this process eliminate the need for expensive coupling agents?
A: Yes, the novel route avoids the use of costly condensing agents like CDI, EDCI, or HOBT during the critical benzimidazole ring formation and subsequent coupling steps, thereby reducing raw material costs.
Q: How does this method improve environmental compliance in manufacturing?
A: By avoiding the harsh hydrolysis of cyano groups with ethanol hydrochloride which generates large amounts of waste acid, this process utilizes milder conditions and simpler workup procedures, resulting in a cleaner production profile.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dabigatran Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes for high-value pharmaceutical intermediates like those used in dabigatran etexilate production. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent can be fully realized in a practical manufacturing setting. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of intermediate meets the exacting standards required for API synthesis. Our commitment to quality and consistency makes us an ideal partner for pharmaceutical companies seeking to secure their supply chain for anticoagulant medications.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis method can be integrated into your supply chain strategy. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the specific economic benefits applicable to your production volume. We encourage potential partners to contact us to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions based on hard data and proven technical capability. Let us collaborate to drive down costs and enhance the reliability of your pharmaceutical supply chain.
