Advanced Green Manufacturing of Benproperine Phosphate Intermediates for Global Pharma Supply Chains
Advanced Green Manufacturing of Benproperine Phosphate Intermediates for Global Pharma Supply Chains
The pharmaceutical industry is currently undergoing a significant paradigm shift towards sustainable manufacturing, driven by both regulatory pressure and the economic necessity of efficient resource utilization. A pivotal development in this arena is detailed in patent CN108752290B, which outlines a revolutionary green synthesis method for benproperine phosphate, a critical intermediate for non-narcotic antitussive agents. This technology addresses long-standing inefficiencies in the production of cough suppressants by introducing a novel catalytic system that replaces hazardous reagents with environmentally benign alternatives. For R&D directors and supply chain leaders, this patent represents more than just a chemical curiosity; it offers a tangible pathway to drastically reduce production costs while enhancing the safety profile of the manufacturing process. By leveraging a 1,1,3,3-tetra-trifluoromethanesulfonyl propylene (TTP) and Hexafluoroisopropanol (HFIP) catalytic system, the process achieves unprecedented yields in the benzylation step, effectively solving the bottleneck of low selectivity that has plagued traditional methods for decades.
Furthermore, the substitution of toxic chlorinating agents with a methyl dichlorosilane and anhydrous ferric trichloride system marks a substantial advancement in process safety and waste management. This dual-innovation approach not only streamlines the synthetic route but also aligns perfectly with the stringent environmental standards required by modern regulatory bodies in Europe and North America. As a reliable pharmaceutical intermediate supplier, understanding and adopting such technologies is crucial for maintaining competitiveness in a market that increasingly values green chemistry credentials. The implications of this patent extend beyond mere compliance; they offer a strategic advantage in cost reduction in API manufacturing by minimizing raw material waste and simplifying downstream purification protocols. This report delves deep into the technical nuances of this breakthrough, providing a comprehensive analysis for stakeholders looking to optimize their supply chains for high-purity antitussive intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of benproperine phosphate has been fraught with significant technical and environmental challenges that hindered scalable production. Traditional routes typically relied on aluminum oxide (Al2O3) activation for the initial benzylation of phenol, a process notorious for its poor selectivity and cumbersome operational requirements. This legacy method necessitated high temperatures and extended reaction times, leading to substantial energy consumption and the generation of difficult-to-remove aluminum salt byproducts. Moreover, the subsequent chlorination step conventionally utilized thionyl chloride in the presence of pyridine and benzene, creating a hazardous working environment due to the evolution of corrosive sulfur dioxide and hydrogen chloride gases. These toxic reagents not only posed severe health risks to operators but also imposed heavy burdens on waste treatment facilities, driving up the overall cost of production. The cumulative effect of these inefficiencies was a low overall yield, often hovering around 35%, which meant that nearly two-thirds of the valuable starting materials were lost as waste, severely impacting the economic viability of the process.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN108752290B introduces a streamlined, high-efficiency workflow that fundamentally reimagines the synthetic strategy. The core innovation lies in the deployment of a TTP/HFIP catalytic system for the benzylation reaction, which operates under milder conditions while delivering exceptional conversion rates. This new approach completely bypasses the need for aluminum oxide activation, thereby eliminating the associated purification headaches and metal waste streams. Additionally, the replacement of the thionyl chloride/pyridine/benzene triad with a methyl dichlorosilane and ferric trichloride system in ethylene glycol dimethyl ether represents a masterstroke in green chemistry design. This modification not only boosts the yield of the chlorination step to over 90% but also eradicates the release of noxious gases, ensuring a safer and more compliant production environment. The result is a robust, scalable process that doubles the total yield to approximately 70.7%, offering a compelling value proposition for commercial scale-up of complex pharmaceutical intermediates.
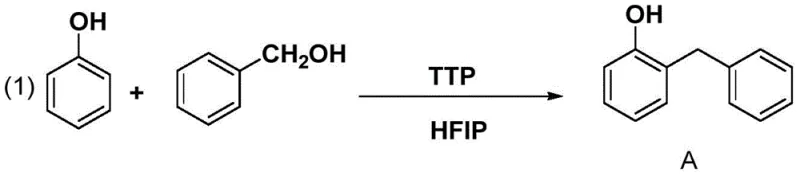
The visual representation of the first step highlights the elegance of the new catalytic system, where phenol and benzyl alcohol are coupled efficiently without the need for harsh Lewis acids. This reaction serves as the foundation for the entire synthesis, setting the stage for high-purity downstream processing. By securing a near-quantitative yield at this early stage, the process minimizes the accumulation of impurities that would otherwise complicate later crystallization steps. This level of control is essential for meeting the rigorous quality standards demanded by global health authorities, ensuring that the final active pharmaceutical ingredient is free from genotoxic impurities often associated with traditional alkylation methods.
Mechanistic Insights into TTP/HFIP Catalysis and FeCl3-Mediated Chlorination
To fully appreciate the technical superiority of this green synthesis route, one must examine the underlying mechanistic drivers that enable such high performance. The TTP/HFIP system functions through a unique activation mechanism where the hexafluoroisopropanol solvent acts as a strong hydrogen bond donor, significantly enhancing the electrophilicity of the reactive intermediates generated by TTP. This synergistic interaction facilitates the nucleophilic attack of phenol on the benzyl species with remarkable regioselectivity, favoring the ortho-substitution required for benproperine synthesis. Unlike traditional Friedel-Crafts conditions that rely on stoichiometric amounts of metal salts, this organocatalytic approach operates with high atom economy, reducing the chemical load on the reactor. The absence of metal residues at this stage is particularly beneficial for R&D teams focused on impurity profiling, as it simplifies the analytical workload and reduces the risk of metal-catalyzed degradation pathways in the final drug product.

Moving to the chlorination step, the mechanism involves the activation of the hydroxyl group by methyl dichlorosilane in the presence of anhydrous ferric trichloride. This combination generates a highly reactive silyl intermediate that undergoes nucleophilic substitution by chloride ions, effectively converting the alcohol to the corresponding alkyl chloride. The use of FeCl3 as a Lewis acid catalyst here is critical, as it promotes the reaction at moderate temperatures (around 84°C), preventing thermal decomposition of the sensitive ether linkage. This stands in sharp contrast to the aggressive conditions required by thionyl chloride, which often lead to side reactions and racemization. The precision of this chlorination method ensures that the stereochemical integrity of the molecule is preserved, a factor that is increasingly important for the biological activity of chiral drugs. Furthermore, the byproduct of this reaction is easily manageable, avoiding the corrosive nature of HCl gas and allowing for simpler workup procedures involving standard aqueous washes.
Impurity control is another area where this mechanism shines, as the mild reaction conditions suppress the formation of elimination byproducts and polymeric tars. The high selectivity of the FeCl3-catalyzed system means that the crude product requires minimal purification before proceeding to the amination step. This efficiency translates directly into higher throughput and reduced solvent consumption, key metrics for any process chemist aiming to optimize a manufacturing line. The ability to achieve a purity of 99.5% in the final phosphate salt is a direct consequence of these clean reaction profiles, demonstrating that green chemistry principles can indeed lead to superior product quality. For procurement managers, this reliability in quality assurance reduces the risk of batch failures and ensures a consistent supply of material for formulation teams.
How to Synthesize Benproperine Phosphate Efficiently
The implementation of this green synthesis route requires careful attention to reaction parameters and reagent quality to maximize the benefits outlined in the patent. The process begins with the precise mixing of phenol, TTP, and benzyl alcohol in HFIP, followed by controlled heating to ensure complete conversion without degradation. Subsequent steps involve alkaline ring-opening with propylene oxide and the critical chlorination using the silane/iron system, each requiring specific temperature controls and inert atmosphere conditions to prevent moisture sensitivity issues. The final stages involve amination with piperidine and salt formation with phosphoric acid, where pH control is paramount to obtaining the correct crystalline form of the phosphate salt. While the general workflow is straightforward, the devil is in the details, and adherence to the specific molar ratios and solvent volumes described in the intellectual property is essential for reproducibility. For those seeking to implement this technology, the following standardized guide provides the foundational framework for execution.
- Perform benzylation of phenol and benzyl alcohol using TTP and HFIP solvent at 100°C to obtain o-benzylphenol with high selectivity.
- React o-benzylphenol with propylene oxide under alkaline conditions to form the hydroxy-propyl ether intermediate.
- Convert the hydroxyl group to chloride using methyl dichlorosilane and anhydrous ferric trichloride in DME, followed by amination and salt formation.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this green synthesis method offers profound advantages that extend well beyond the laboratory bench, directly impacting the bottom line and supply chain resilience. The most immediate benefit is the dramatic improvement in overall process yield, which effectively doubles the output from the same amount of raw materials compared to legacy methods. This efficiency gain translates into significant cost savings on starting materials, which are often subject to market volatility, thereby stabilizing the cost of goods sold (COGS). Moreover, the elimination of hazardous reagents like benzene and pyridine removes the need for expensive specialized containment systems and complex scrubbing equipment, lowering capital expenditure requirements for new production lines. The reduction in toxic waste generation also means lower disposal fees and a smaller environmental footprint, which is increasingly becoming a criterion for vendor selection by major pharmaceutical companies committed to sustainability goals.
- Cost Reduction in Manufacturing: The transition to a high-yield catalytic process fundamentally alters the economics of production by maximizing the utility of every kilogram of input material. By avoiding the use of stoichiometric aluminum oxide and toxic chlorinating agents, the process reduces the consumption of auxiliary chemicals and the associated costs of neutralization and waste treatment. The simplified workup procedures, which do not require extensive chromatography or complex extractions to remove metal salts, further decrease the operational expenses related to solvent usage and labor hours. Consequently, manufacturers can achieve a leaner production model that is less susceptible to fluctuations in raw material pricing, ensuring more stable margins in a competitive market.
- Enhanced Supply Chain Reliability: The robustness of the new synthetic route contributes to a more dependable supply chain by minimizing the risk of batch failures due to impurity buildup or reaction stalling. Traditional methods often suffered from inconsistent yields due to the sensitivity of the aluminum activation step, leading to unpredictable delivery schedules. In contrast, the TTP/HFIP and FeCl3 systems offer reproducible results with high selectivity, allowing for tighter production planning and shorter lead times. Additionally, the use of more common and less regulated solvents like ethylene glycol dimethyl ether reduces the logistical complexities associated with sourcing and transporting highly controlled substances, ensuring uninterrupted production flows even during periods of regulatory tightening.
- Scalability and Environmental Compliance: Scaling this process from pilot to commercial production is facilitated by the inherent safety of the reagents and the mild operating conditions. The absence of exothermic hazards associated with thionyl chloride and the elimination of corrosive gas evolution make the process easier to manage in large-scale reactors, reducing the engineering controls needed for safe operation. This ease of scale-up allows manufacturers to respond quickly to surges in demand for antitussive medications without compromising on safety or quality. Furthermore, the alignment with green chemistry principles ensures long-term regulatory compliance, future-proofing the manufacturing asset against increasingly stringent environmental laws and avoiding potential fines or shutdowns related to pollutant emissions.
Frequently Asked Questions (FAQ)
The following questions address common inquiries regarding the technical feasibility and commercial implications of this green synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing a clear picture of what stakeholders can expect when transitioning to this new method. Understanding these details is crucial for making informed decisions about process adoption and supplier qualification.
Q: How does the TTP/HFIP system improve the benzylation step compared to traditional methods?
A: The TTP/HFIP system eliminates the need for aluminum oxide activation, significantly increasing reaction yield to 95-100% and simplifying the purification process by avoiding aluminum salt byproducts.
Q: What are the environmental advantages of the new chlorination method?
A: By replacing thionyl chloride, pyridine, and benzene with methyl dichlorosilane and FeCl3, the process avoids generating corrosive sulfur dioxide and hydrogen chloride gases, drastically reducing hazardous waste treatment requirements.
Q: What is the overall purity and yield achievable with this green synthesis route?
A: The optimized process achieves a total yield of approximately 70.7%, nearly double that of conventional methods, with a final product purity reaching 99.5%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benproperine Phosphate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced manufacturing processes like the one described in CN108752290B requires a partner with deep technical expertise and a commitment to quality. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of green chemistry are fully realized in practical, large-scale operations. Our state-of-the-art facilities are equipped to handle the specific solvent systems and catalytic reagents required for this synthesis, maintaining stringent purity specifications throughout the entire value chain. With our rigorous QC labs and dedicated process development teams, we guarantee that every batch of benproperine phosphate meets the highest international standards, providing our clients with the confidence they need to bring life-saving medications to market faster.
We invite you to collaborate with us to leverage this cutting-edge technology for your supply chain needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this green route can optimize your budget. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing you to evaluate the superior quality and reliability of our manufacturing capabilities firsthand. Together, we can build a more sustainable and efficient future for pharmaceutical production.
