Advanced Manufacturing of Abemaciclib Intermediates via Selective Fluorination Technology
Advanced Manufacturing of Abemaciclib Intermediates via Selective Fluorination Technology
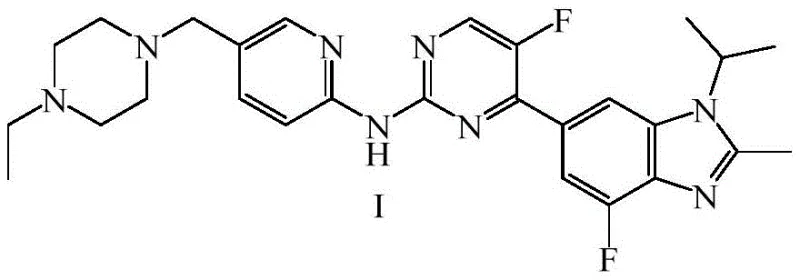
The pharmaceutical landscape for oncology treatments has been significantly transformed by the introduction of CDK4/6 inhibitors, with Abemaciclib standing out as a pivotal therapy for HR+ and HER2- breast cancer. As detailed in the recent patent CN113912548A, a groundbreaking preparation method for Abemaciclib and its critical intermediates has been disclosed, offering a transformative approach to manufacturing this high-value active pharmaceutical ingredient. This technical insight report analyzes the novel synthetic route which centers on the efficient production of the key intermediate 3-(dimethylamino)-2-fluoro-1-(1-isopropyl-2-methyl-1H-benzo[d]imidazol-6-yl)prop-2-en-1-one (Intermediate V). By shifting away from traditional palladium-catalyzed cross-couplings and hazardous fluorinating agents, this methodology presents a compelling value proposition for global supply chains seeking robust, compliant, and cost-effective solutions for complex API intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Abemaciclib has relied heavily on transition metal catalysis, specifically palladium-mediated Suzuki couplings, as illustrated in earlier patents such as US20100160340. While chemically effective, these legacy routes impose severe burdens on commercial manufacturing, primarily due to the stringent regulatory requirement for palladium residues to remain below 10ppm in the final drug substance. Achieving this level of purity necessitates expensive scavenging resins and complex purification protocols, which drastically inflate production costs and extend lead times. Furthermore, alternative routes reported in literature, such as those utilizing fluoromethyl magnesium bromide, face critical supply chain bottlenecks because this specific Grignard reagent is not commercially available and is notoriously difficult to synthesize with consistent quality. The reliance on hazardous reagents like sodium hydride and phosphorus oxychloride in other known methods further exacerbates environmental compliance issues and operational safety risks, making these conventional pathways increasingly untenable for modern, green chemistry-focused production facilities.
The Novel Approach
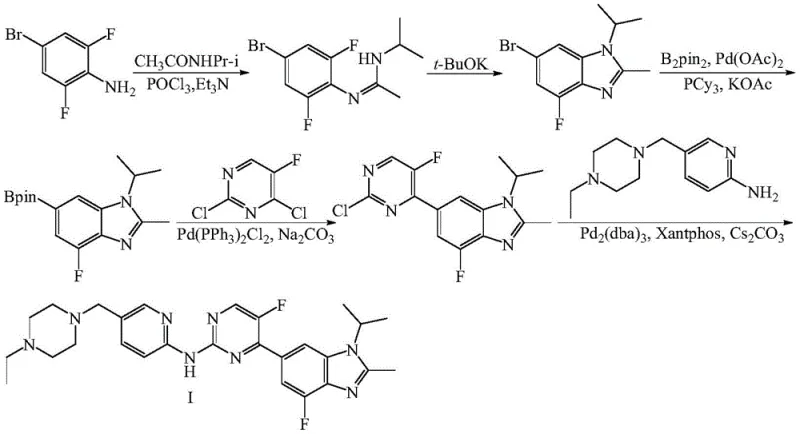
The innovative strategy outlined in patent CN113912548A effectively circumvents these historical bottlenecks by introducing a streamlined, non-palladium synthetic sequence. Instead of struggling with hard-to-source fluorinated Grignard reagents, the new method employs readily available methyl magnesium halides (chloride, bromide, or iodide) to construct the carbon skeleton, followed by a highly selective electrophilic fluorination step. This paradigm shift not only simplifies the raw material procurement process but also inherently improves the impurity profile of the intermediate by avoiding the competing side reactions often associated with boronic ester couplings. By replacing corrosive and environmentally taxing reagents like phosphorus trichloride with milder alternatives, the process aligns with modern sustainability goals while maintaining high chemical efficiency. This approach represents a significant technological leap, transforming a complex, multi-step hazard-laden process into a more manageable and economically viable manufacturing operation suitable for large-scale deployment.
Mechanistic Insights into Selective Fluorination and Grignard Addition
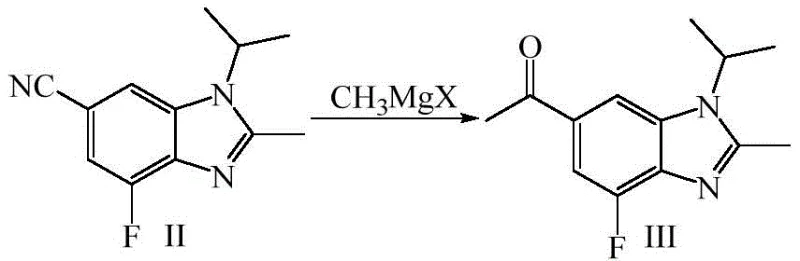
The core of this technological advancement lies in the precise execution of the Grignard addition and the subsequent selective fluorination, which together construct the crucial fluorinated enamine motif found in the final API. The process initiates with the reaction of 1-isopropyl-2-methyl-4-fluoro-1H-benzo[d]imidazole-6-carbonitrile with a methyl magnesium halide in a solvent system such as tetrahydrofuran. This nucleophilic addition converts the nitrile group into a ketone intermediate (Compound III) with exceptional efficiency, as evidenced by yields reaching up to 92% in optimized examples. The reaction is carefully controlled at low temperatures (0-5°C) to manage exothermicity and prevent over-addition or degradation of the sensitive benzimidazole core. Following isolation, the ketone undergoes condensation with N,N-dimethylformamide dimethyl acetal (DMF-DMA) to form an enamine intermediate, setting the stage for the critical fluorination event.
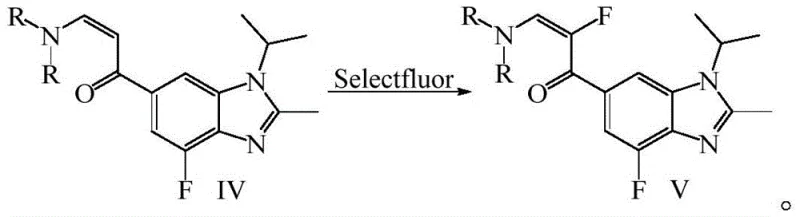
The defining step of this novel route is the electrophilic fluorination of the enamine intermediate using Selectfluor (1-chloromethyl-4-fluoro-1,4-diazoniabicyclo[2.2.2]octane bis(tetrafluoroborate)). This reagent allows for the direct introduction of the fluorine atom at the alpha-position of the carbonyl group under mild basic conditions. A key mechanistic feature described in the patent is the inclusion of a radical trapping agent, such as TEMPO or BHT (2,6-di-tert-butyl-p-cresol), which suppresses unwanted free-radical side reactions that could otherwise lead to polymerization or decomposition of the reactive enamine. This ensures high regioselectivity and purity of the resulting fluorinated intermediate (Compound V). The final assembly of Abemaciclib is achieved through a cyclization reaction between this fluorinated intermediate and a guanidine derivative, completing the pyrimidine ring formation without the need for further transition metal catalysis.
How to Synthesize Abemaciclib Intermediate V Efficiently
The synthesis of the critical Intermediate V is achieved through a robust three-step sequence that prioritizes safety and yield. The process begins with the Grignard addition to the nitrile precursor, followed by condensation with DMF-DMA, and concludes with the Selectfluor-mediated fluorination. Detailed operational parameters, including specific solvent choices like THF and toluene, temperature controls, and quenching procedures, are essential for reproducing the high yields reported in the patent examples. For a comprehensive understanding of the standardized operating procedures and quality control checkpoints required for GMP manufacturing, please refer to the technical guide below.
- React 1-isopropyl-2-methyl-4-fluoro-1H-benzo[d]imidazole-6-carbonitrile with methyl magnesium chloride in THF at 0-5°C to form the ketone intermediate.
- Perform condensation with N,N-dimethylformamide dimethyl acetal (DMF-DMA) in toluene under reflux to generate the enamine derivative.
- Execute selective fluorination using Selectfluor reagent with a radical trapping agent like BHT to yield the final fluorinated intermediate V.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers substantial strategic benefits that extend beyond simple chemical yield improvements. By fundamentally altering the reagent profile, the process mitigates several high-risk factors associated with the traditional manufacturing of Abemaciclib intermediates. The elimination of palladium catalysts removes a major cost driver related to both the expensive metal itself and the downstream purification technologies required to meet regulatory standards. Furthermore, the substitution of obscure, custom-synthesized reagents with commodity chemicals enhances supply security, reducing the risk of production stoppages due to raw material shortages. This route also significantly lowers the environmental burden by avoiding chlorinated phosphorus reagents, thereby simplifying waste treatment protocols and reducing the overall carbon footprint of the manufacturing process.
- Cost Reduction in Manufacturing: The economic impact of switching to this palladium-free route is profound, primarily driven by the removal of precious metal costs and the associated scavenging processes. Traditional methods require significant capital expenditure on palladium salts and specialized filtration media to ensure residual metal levels comply with ICH Q3D guidelines. By utilizing inexpensive methyl magnesium halides and Selectfluor, the direct material costs are drastically lowered. Additionally, the simplified workup procedures, which avoid complex chromatographic separations often needed to remove metal impurities, result in reduced labor and utility consumption. The overall process efficiency is enhanced by higher yields in the early stages, meaning less raw material is wasted, contributing to a leaner and more cost-effective production model.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly improved by the reliance on globally available commodity reagents rather than specialized, low-volume intermediates. The previous dependence on fluoromethyl magnesium bromide created a single point of failure, as this reagent is not stocked by major chemical suppliers and requires custom synthesis with variable quality. In contrast, methyl magnesium chloride and Selectfluor are produced at industrial scales by multiple vendors worldwide, ensuring a stable and competitive supply market. This diversification of the supplier base protects against geopolitical disruptions and price volatility, allowing for more accurate long-term forecasting and inventory planning. The robustness of the reaction conditions also means that the process is less susceptible to batch failures, ensuring consistent on-time delivery to downstream API manufacturers.
- Scalability and Environmental Compliance: From an operational perspective, the new route is inherently safer and easier to scale, addressing key concerns for EHS (Environment, Health, and Safety) teams. The avoidance of sodium hydride, a pyrophoric solid that poses significant fire hazards in large-scale reactors, reduces the need for specialized handling equipment and rigorous safety protocols. Similarly, eliminating phosphorus oxychloride removes the generation of acidic phosphorus waste streams, which are costly and difficult to treat. The use of standard organic solvents like THF, toluene, and methanol facilitates solvent recovery and recycling, aligning with green chemistry principles. These factors collectively lower the barrier for technology transfer from pilot plant to commercial production, enabling faster time-to-market for generic or biosimilar versions of the drug while maintaining strict adherence to environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel Abemaciclib synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in patent CN113912548A, offering clarity on how this method resolves specific pain points in API manufacturing. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this greener, more efficient pathway for their own production lines.
Q: How does this new route address Palladium residue concerns in Abemaciclib synthesis?
A: Unlike conventional methods relying on Suzuki couplings with homogeneous palladium catalysts, this novel route utilizes a Grignard addition and Selectfluor fluorination strategy. This completely eliminates the need for palladium, thereby removing the complex and costly purification steps required to meet the strict <10ppm residual metal limits mandated by drug regulatory authorities.
Q: What are the safety advantages of avoiding fluoromethyl magnesium bromide?
A: Traditional routes often require fluoromethyl magnesium bromide, a reagent that is difficult to source commercially and challenging to synthesize safely due to stability issues. The patented method substitutes this with readily available methyl magnesium chloride or bromide, followed by a controlled electrophilic fluorination, significantly enhancing operational safety and supply chain reliability.
Q: Is this process scalable for commercial API production?
A: Yes, the process is designed for scalability. It avoids hazardous reagents like sodium hydride and phosphorus oxychloride, using milder conditions and common solvents like THF and toluene. The high yields reported in the examples (up to 92% for the Grignard step) indicate a robust process suitable for multi-kilogram to ton-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Abemaciclib Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global pharmaceutical market. Our team of expert process chemists has thoroughly analyzed the route described in CN113912548A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are fully equipped to implement this palladium-free, Selectfluor-based technology, ensuring that our clients receive high-purity Abemaciclib intermediates that meet stringent purity specifications. Our state-of-the-art facilities include rigorous QC labs capable of detecting trace impurities and residual metals, guaranteeing that every batch delivered adheres to the highest international quality standards required for oncology drug manufacturing.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific volume and cost requirements. By leveraging our expertise in process optimization, we can provide a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this greener methodology. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing you to make informed decisions that will enhance your supply chain efficiency and reduce your overall manufacturing costs.
