Advanced Pure Water Suzuki Coupling for Scalable Fluorobiaryl Intermediate Manufacturing
Advanced Pure Water Suzuki Coupling for Scalable Fluorobiaryl Intermediate Manufacturing
The chemical manufacturing landscape is undergoing a profound transformation driven by the urgent need for greener, more sustainable synthetic methodologies that do not compromise on yield or purity. A pivotal advancement in this domain is documented in patent CN101857517B, which discloses a novel method for preparing fluorobiaryl compounds via Suzuki cross-coupling reactions conducted entirely in pure water solutions. This technology represents a significant departure from conventional practices that rely heavily on volatile organic solvents and complex workup procedures. By utilizing a specialized temperature-controlled phosphine ligand in conjunction with palladium catalysts, this process effectively solves the longstanding challenge of reacting water-insoluble substrates in aqueous media. For R&D directors and process chemists, this patent offers a compelling route to high-purity intermediates essential for pharmaceuticals and liquid crystal materials, while simultaneously addressing critical environmental compliance standards. The ability to operate without organic co-solvents during the reaction phase not only aligns with green chemistry principles but also drastically simplifies the downstream processing infrastructure required for commercial production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of fluorobiaryl structures, which are ubiquitous motifs in bioactive molecules and advanced electronic materials, has relied on Suzuki-Miyaura coupling reactions performed in organic solvents or mixtures of organic solvents and water. These conventional methods suffer from several inherent drawbacks that impact both economic efficiency and environmental sustainability. Firstly, the extensive use of organic solvents such as toluene, dioxane, or DMF necessitates rigorous safety protocols for handling flammable and toxic materials, increasing operational costs and regulatory burdens. Secondly, the separation and recovery of expensive palladium catalysts from organic reaction mixtures are notoriously difficult and often result in significant metal loss, driving up the cost of goods sold. Furthermore, traditional aqueous Suzuki reactions are typically limited to water-soluble substrates, restricting their utility for the vast array of hydrophobic intermediates common in drug discovery. The need for phase transfer catalysts or substrate modification to overcome solubility issues adds further complexity and potential impurities to the final product, complicating the purification process and reducing overall atom economy.
The Novel Approach
The methodology outlined in patent CN101857517B introduces a paradigm shift by enabling efficient cross-coupling in pure water without the need for organic co-solvents or phase transfer agents. This breakthrough is achieved through the strategic application of a temperature-controlled phosphine ligand, which possesses non-ionic surfactant cloud point characteristics. This unique ligand design allows the reaction to proceed homogeneously at elevated temperatures (80-100°C) even with water-insoluble substrates, ensuring high reaction rates and excellent conversion. Upon completion of the reaction, the system facilitates a straightforward phase separation where the product can be extracted into a minimal amount of organic solvent like diethyl ether, while the catalyst remains dissolved in the aqueous phase. This innovative approach not only eliminates the environmental hazards associated with bulk organic solvents but also enables the direct recycling of the catalyst-containing water phase for multiple cycles. Such a process design significantly enhances the robustness of the manufacturing workflow, making it highly attractive for the commercial scale-up of complex pharmaceutical intermediates and specialty chemicals.
Mechanistic Insights into Temperature-Controlled Ligand Catalysis
The core innovation driving this technology lies in the molecular architecture of the temperature-controlled phosphine ligand, often referred to as Ligand I in the patent literature. This ligand features a polyethylene glycol (PEG) chain attached to a diphenylphosphine group, creating an amphiphilic structure that responds dynamically to temperature changes. At room temperature, the PEG chain renders the ligand soluble in water, allowing for the formation of a stable palladium-ligand complex. As the reaction temperature is raised to the optimal range of 80-100°C, the ligand undergoes a phase transition characterized by its cloud point, effectively creating a micro-environment that solubilizes the hydrophobic organic substrates within the aqueous medium. This mechanism ensures that the reactants and the catalyst are in close proximity, facilitating the oxidative addition, transmetallation, and reductive elimination steps of the catalytic cycle with high efficiency. The precise tuning of the ethylene oxide units (where n = 10, 16, 22) allows chemists to optimize the cloud point for specific substrate profiles, providing a versatile toolkit for process optimization.
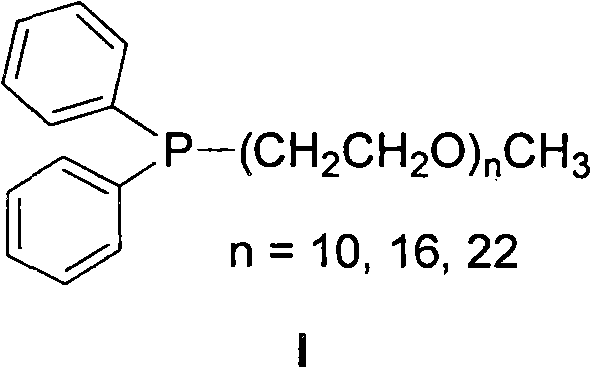
Beyond merely enabling the reaction, this mechanistic design plays a crucial role in impurity control and catalyst management. The ability to separate the catalyst into the aqueous phase post-reaction means that palladium contamination in the final organic product is minimized, a critical parameter for pharmaceutical intermediates where heavy metal limits are strictly regulated. The aqueous phase, retaining the active catalytic species, can be directly reused for subsequent batches without extensive regeneration, maintaining consistent activity over multiple cycles as demonstrated in the patent examples. This recyclability not only reduces the consumption of precious metals but also prevents the accumulation of palladium black or other inactive species that often plague homogeneous catalysis in organic solvents. For quality assurance teams, this translates to a more consistent impurity profile and reduced risk of batch-to-batch variability, ensuring that the high-purity fluorobiaryl compounds meet stringent analytical specifications required for downstream applications in medicine and agrochemicals.
How to Synthesize Fluorobiaryl Compounds Efficiently
Implementing this aqueous Suzuki coupling protocol requires careful attention to the preparation of the catalytic system and the control of reaction parameters to maximize yield and catalyst longevity. The process begins with the complexation of the palladium source, such as palladium acetate or palladium chloride, with the temperature-controlled ligand in deoxygenated water under an inert nitrogen atmosphere. This pre-activation step is vital to ensure the formation of the active catalytic species before the introduction of substrates. Following this, the base, halogenated aromatic compound, and fluoroarylboronic acid are added sequentially, and the mixture is heated to maintain the temperature between 80°C and 100°C for a duration of 1 to 4 hours depending on the specific reactivity of the substrates. The detailed standardized synthesis steps, including specific molar ratios and workup procedures for various derivatives, are provided in the guide below to assist technical teams in replicating these high-yielding results.
- Complex the palladium catalyst and temperature-controlled phosphine ligand in deoxygenated water under nitrogen protection for 30 minutes to form the active catalytic species.
- Add the base, halogenated aromatic compound, and fluoroarylboronic acid sequentially, then heat the mixture to 80-100°C for 1-4 hours to facilitate the coupling reaction.
- Extract the product with diethyl ether to separate the organic phase, allowing the aqueous phase containing the catalyst to be recycled for subsequent batches.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this pure water Suzuki coupling technology offers transformative advantages that directly impact the bottom line and operational resilience. The elimination of bulk organic solvents during the reaction phase leads to a substantial reduction in raw material costs and waste disposal fees, as water is significantly cheaper and easier to manage than specialized organic solvents. Furthermore, the simplified workup procedure, which relies on simple phase separation rather than complex distillation or chromatography for solvent removal, drastically reduces energy consumption and processing time. This streamlining of the manufacturing process enhances throughput capacity, allowing facilities to produce larger volumes of high-purity intermediates without proportional increases in infrastructure investment. For supply chain managers, the ability to recycle the catalyst aqueous phase multiple times ensures a more stable supply of critical reagents and reduces dependency on fluctuating markets for precious metal catalysts, thereby mitigating supply risk.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the drastic reduction in solvent usage and the efficient recycling of the palladium catalyst. By replacing expensive organic solvents with water, the direct material costs are significantly lowered, while the ability to reuse the catalyst solution over multiple cycles minimizes the consumption of high-value palladium salts. Additionally, the simplified purification workflow reduces the labor and equipment time required for product isolation, leading to lower overall operating expenses. These cumulative efficiencies result in a more cost-effective production model that enhances competitiveness in the global market for fine chemical intermediates without compromising on product quality or yield.
- Enhanced Supply Chain Reliability: Implementing a water-based synthesis route improves supply chain robustness by reducing reliance on volatile organic solvents that are subject to strict transportation regulations and supply fluctuations. The stability of the aqueous catalyst system allows for safer storage and handling, minimizing the risk of production delays due to hazardous material incidents. Moreover, the high yields and reproducibility demonstrated across a wide range of substrates ensure consistent output, enabling reliable fulfillment of customer orders. This reliability is crucial for maintaining long-term partnerships with pharmaceutical and electronic material clients who require uninterrupted supply of critical intermediates for their own manufacturing pipelines.
- Scalability and Environmental Compliance: The green chemistry attributes of this method facilitate easier regulatory approval and scalability from pilot plant to commercial production. The absence of toxic organic solvents simplifies waste treatment processes and reduces the environmental footprint of the manufacturing facility, aligning with increasingly stringent global environmental standards. This compliance advantage accelerates the time-to-market for new products and reduces the administrative burden associated with environmental reporting. The process is inherently scalable, as the heat transfer and mixing characteristics of aqueous systems are well-understood in large-scale reactors, ensuring a smooth transition from laboratory optimization to multi-ton annual production capacities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this pure water Suzuki coupling technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the practical aspects of adopting this method. Understanding these details is essential for technical teams evaluating the feasibility of integrating this process into existing manufacturing lines or developing new synthetic routes for fluorinated biaryl targets.
Q: How does the temperature-controlled ligand facilitate catalyst recovery?
A: The ligand exhibits non-ionic surfactant cloud point characteristics, becoming insoluble in water at higher temperatures to drive the reaction, but allowing phase separation for catalyst retention in the aqueous layer upon cooling or extraction.
Q: What are the environmental benefits of this pure water method?
A: By eliminating organic co-solvents during the reaction phase, this method significantly reduces volatile organic compound (VOC) emissions and simplifies waste treatment protocols compared to traditional organic solvent-based Suzuki couplings.
Q: Can this method be applied to water-insoluble substrates?
A: Yes, the specific design of the temperature-controlled ligand overcomes the solubility limitations of traditional aqueous Suzuki reactions, enabling high yields even with hydrophobic halogenated aromatic substrates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fluorobiaryl Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the immense potential of green catalytic technologies like the one described in patent CN101857517B to revolutionize the production of high-value chemical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of fluorobiaryl compounds delivered meets the highest standards required by the pharmaceutical and electronic materials industries. We are committed to leveraging our technical expertise to help clients optimize their supply chains through sustainable and cost-effective manufacturing solutions.
We invite you to collaborate with us to explore how this advanced aqueous Suzuki coupling technology can benefit your specific project requirements. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating the tangible economic advantages of switching to this green synthesis route. Please contact us today to request specific COA data for our available intermediates or to discuss route feasibility assessments for your custom synthesis projects. Together, we can drive innovation and efficiency in the global supply of critical fine chemical intermediates.
