Revolutionizing API Intermediate Synthesis via Cost-Effective Iron-Catalyzed Reductive Coupling Technology
The pharmaceutical and fine chemical industries are constantly seeking robust, cost-effective methodologies for constructing carbon-carbon bonds, particularly for complex API intermediates. Patent CN111302880B introduces a groundbreaking application of iron catalysts in the reductive coupling reaction of phenol derivatives and alkyl halides, marking a significant departure from traditional noble metal catalysis. This technology leverages the unique redox properties of ferrous iodide or ferrous bromide to facilitate the formation of sp2-sp3 carbon-carbon bonds under mild conditions. By utilizing readily available phenol derivatives as electrophiles instead of expensive aryl halides, this method addresses critical pain points regarding raw material costs and environmental toxicity. For R&D directors and procurement managers alike, this innovation represents a paradigm shift towards sustainable and economically viable synthetic routes for high-value aromatic and heterocyclic derivatives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional transition metal-catalyzed cross-coupling reactions, such as Suzuki or Kumada couplings, have long been the standard for forming carbon-carbon bonds, yet they suffer from inherent logistical and economic drawbacks. These methods typically require the preparation of unstable organometallic nucleophiles, such as Grignard reagents or organoboron compounds, which demand strict anhydrous and oxygen-free conditions, thereby increasing operational complexity and safety risks. Furthermore, the reliance on precious metal catalysts like palladium or nickel introduces significant cost burdens and potential toxicity issues, necessitating rigorous and expensive downstream purification steps to meet stringent pharmaceutical purity standards. The use of aryl halides as substrates also contributes to higher material costs and generates halogenated waste, posing environmental challenges that modern green chemistry initiatives strive to eliminate from the supply chain.
The Novel Approach
The novel approach detailed in the patent data circumvents these limitations by employing an earth-abundant iron catalyst system that operates efficiently with phenol derivatives and alkyl halides. This method utilizes a diethylcarbamoyl group as a protecting group on the phenol, which, despite its strong bond dissociation energy, is effectively activated by the iron catalyst in the presence of a reducing agent like bis(pinacolato)diboron. The reaction proceeds smoothly in methyl tert-butyl ether (MTBE) at moderate temperatures ranging from 50°C to 90°C, eliminating the need for cryogenic conditions or highly sensitive reagents. This strategic shift not only drastically reduces the cost of goods sold (COGS) by replacing expensive catalysts and substrates but also simplifies the workflow, making it an attractive option for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Fe-Catalyzed Reductive Coupling
The mechanistic pathway of this iron-catalyzed reductive coupling involves a sophisticated interplay between the iron center, the ligand, and the reducing agents to achieve oxidative addition into the robust C-O bond. The catalytic cycle likely initiates with the reduction of the Fe(II) species to a low-valent active iron species by the diboron reagent in the presence of the alkoxide base. This active iron complex then undergoes oxidative addition with the phenol derivative, specifically targeting the carbon-oxygen bond activated by the electron-withdrawing carbamate group. Subsequent transmetallation or radical capture steps involving the alkyl halide allow for the formation of the new carbon-carbon bond, followed by reductive elimination to release the product and regenerate the catalyst. The use of TMEDA as a ligand is crucial for stabilizing the iron species and modulating its electronic properties to facilitate this challenging transformation.
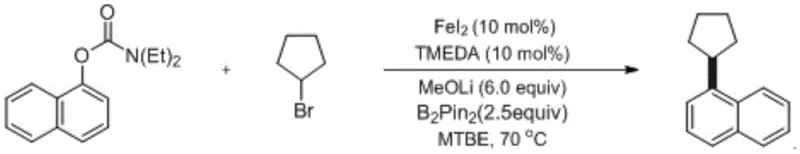
Impurity control in this system is inherently superior due to the high chemoselectivity of the iron catalyst towards the specific C-O bond activation without affecting other sensitive functional groups often present in drug molecules. The mild reaction conditions prevent thermal degradation of the substrates, while the use of non-toxic iron salts minimizes the risk of heavy metal contamination in the final product. This is particularly vital for pharmaceutical applications where residual metal limits are strictly regulated. The protocol ensures a clean reaction profile, as evidenced by the high yields reported in the examples, such as the 84% yield obtained for 1-cyclopentylnaphthalene, demonstrating the robustness of the method against side reactions like homocoupling or beta-hydride elimination which often plague nickel or palladium systems.
How to Synthesize 1-Cyclopentylnaphthalene Efficiently
The synthesis of 1-cyclopentylnaphthalene serves as a prime example of the efficiency and practicality of this iron-catalyzed protocol. The process begins with the precise weighing of ferrous iodide, the phenolic substrate, lithium methoxide, and the diboron reducing agent under strictly anaerobic conditions to prevent catalyst deactivation. Following the initial mixing and nitrogen purging, the alkyl halide and solvent are introduced, and the mixture is heated to 70°C for 16 hours to ensure complete conversion. This standardized procedure highlights the reproducibility of the method, making it an ideal candidate for technology transfer from laboratory to pilot plant scales. For detailed operational parameters and safety guidelines, please refer to the specific synthesis instructions provided below.
- Under sealed anaerobic conditions, weigh ferrous iodide catalyst, phenolic derivative (e.g., 1-diethylcarbamate naphthalene), lithium methoxide, and bis(pinacolato)diboron. Purge the system with nitrogen multiple times to ensure an oxygen-free environment.
- Add the ligand (TMEDA), alkyl halide (e.g., bromocyclopentane), and organic solvent (MTBE) to the mixture. Maintain the reaction at a constant temperature between 50°C and 90°C for 12 to 16 hours to facilitate the reductive coupling.
- Quench the reaction with saturated ammonium chloride solution. Perform extraction with ethyl acetate, dry the organic phase over anhydrous sodium sulfate, concentrate under vacuum, and purify via column chromatography to obtain the final aromatic derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this iron-catalyzed technology translates directly into enhanced operational efficiency and risk mitigation. The substitution of precious metal catalysts with inexpensive iron salts results in substantial cost savings on raw materials, while the use of phenol derivatives instead of aryl halides further drives down the input costs. Moreover, the elimination of toxic heavy metals simplifies the waste treatment process and reduces the regulatory burden associated with hazardous material handling. This streamlined approach ensures a more reliable supply of high-purity intermediates by minimizing the variables that typically cause batch failures or delays in traditional cross-coupling processes.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the replacement of expensive palladium or nickel catalysts with ferrous iodide, which is orders of magnitude cheaper and more abundant. Additionally, the avoidance of pre-formed organometallic reagents eliminates the need for specialized storage and handling infrastructure, further reducing overhead costs. The simplified purification process, which does not require extensive heavy metal scavenging steps, leads to higher overall throughput and lower processing time per batch, contributing to a leaner manufacturing model.
- Enhanced Supply Chain Reliability: By utilizing widely available and stable starting materials such as phenol derivatives and alkyl halides, the supply chain becomes more resilient to market fluctuations that often affect specialty reagents. The robustness of the iron catalyst system means that production schedules are less likely to be disrupted by reagent instability or sensitivity to environmental conditions. This reliability ensures consistent delivery timelines for critical API intermediates, allowing downstream partners to plan their production cycles with greater confidence and reduced inventory buffers.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing common solvents like MTBE and operating at moderate temperatures that are easily managed in large-scale reactors. The green chemistry credentials of using non-toxic iron and generating less hazardous waste align perfectly with increasingly strict environmental regulations, reducing the risk of compliance-related shutdowns. This environmental compatibility not only safeguards the company's reputation but also future-proofs the manufacturing process against tightening global standards on chemical emissions and waste disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this iron-catalyzed reductive coupling technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, ensuring accuracy and relevance for industrial application. Understanding these nuances is essential for evaluating the feasibility of integrating this method into existing production workflows.
Q: What are the primary advantages of using iron catalysts over nickel or palladium in reductive coupling?
A: Iron catalysts offer significant advantages in terms of cost and toxicity. Unlike nickel or palladium, which are expensive and often possess biological toxicity affecting drug safety, iron is abundant, low-cost, and environmentally benign. Furthermore, this specific protocol utilizes phenol derivatives instead of expensive aryl halides, further reducing raw material costs and environmental impact.
Q: Why are phenol derivatives preferred over traditional aryl halides in this synthesis?
A: Phenol derivatives are naturally abundant, inexpensive, and generate less hazardous waste compared to aryl halides. While C-O bonds are traditionally harder to activate than C-Halogen bonds, this iron-catalyzed method effectively cleaves the carbon-oxygen bond using a diethylcarbamoyl protecting group, providing a greener and more economical pathway for constructing sp2-sp3 carbon-carbon bonds.
Q: Is this iron-catalyzed process suitable for large-scale commercial production?
A: Yes, the process is highly scalable. The reaction conditions are relatively mild (50-90°C) and utilize common solvents like MTBE. The elimination of expensive transition metals and unstable organometallic reagents simplifies the purification process and reduces the burden of heavy metal removal, making it ideal for commercial scale-up in the pharmaceutical industry.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Cyclopentylnaphthalene Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this iron-catalyzed reductive coupling technology for the synthesis of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a commercial setting. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 1-cyclopentylnaphthalene or related derivatives meets the highest quality standards required by global regulatory bodies.
We invite you to collaborate with us to leverage this cost-effective and environmentally friendly synthesis route for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. Please contact us today to request specific COA data and comprehensive route feasibility assessments, and let us help you optimize your supply chain with our advanced catalytic solutions.
