Advanced Solid-Acid Catalysis for High-Purity 7-Hydroxy-3,4-dihydroquinolone Production and Commercial Scale-Up
The pharmaceutical industry's relentless pursuit of cost-effective and environmentally sustainable manufacturing processes has brought significant attention to the synthesis of key antipsychotic intermediates. Patent CN101302195B discloses a novel and highly efficient synthetic method for 7-Hydroxy-3,4-dihydroquinolone, a critical building block in the production of Aripiprazole, a third-generation atypical antipsychotic medication. This technology represents a paradigm shift from traditional stoichiometric Lewis acid methodologies to a greener, catalytic approach utilizing solid acid catalysts such as HZSM-5 molecular sieves. By addressing long-standing issues related to low yields, severe environmental pollution, and complex purification protocols, this innovation offers a robust pathway for reliable pharmaceutical intermediates supplier networks aiming to enhance their production capabilities. The method not only streamlines the operational workflow but also ensures the delivery of high-purity 7-Hydroxy-3,4-dihydroquinolone with minimized ecological impact, aligning perfectly with modern green chemistry principles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of 7-hydroxyl-3,4-dihydro-quinolone has been plagued by significant technical and environmental hurdles that hinder cost reduction in API manufacturing. One conventional route involves a tedious five-step sequence starting from 2-nitro-4-anisidine, proceeding through condensation, esterification, hydrogenation cyclization, and demethylation, which results in a dismal total recovery rate of less than 20 percent. Another prevalent method utilizes m-aminophenyl methyl ether and 3-chloropropionyl chloride but relies heavily on excessive amounts of aluminum chloride (AlCl3) as a catalyst for the Friedel-Crafts alkylation. This traditional approach generates substantial quantities of hazardous acidic waste and requires complex post-reaction treatment to remove metal residues, leading to increased operational expenditures and stringent regulatory compliance burdens. Furthermore, the difficulty in separating isomeric byproducts often compromises the final purity profile, necessitating additional recrystallization steps that further erode overall process efficiency.
The Novel Approach
In stark contrast, the methodology outlined in the patent introduces a streamlined two-step process that fundamentally reimagines the cyclization strategy through the application of heterogeneous catalysis. The core innovation lies in the intramolecular Friedel-Crafts alkylation of N-(3-chloropropionyl)-3-hydroxyaniline under the catalysis of a solid acid, specifically preferring HZSM-5 type molecular sieves, at temperatures ranging between 132 and 200 degrees Celsius. This solvent-free or low-solvent melt reaction eliminates the need for stoichiometric quantities of corrosive Lewis acids, thereby drastically simplifying the workup procedure to a mere filtration and crystallization. The catalyst can be recovered from the filter cake and reused repeatedly, transforming a linear consumption model into a circular one. This novel approach not only boosts the yield significantly compared to the nitro-reduction route but also ensures excellent regioselectivity for the 7-hydroxy position, delivering a product with purity levels exceeding 99 percent as confirmed by HPLC analysis.
Mechanistic Insights into Solid Acid-Catalyzed Intramolecular Cyclization
The mechanistic elegance of this synthesis rests on the unique properties of the zeolite molecular sieve, which acts as a solid proton donor to activate the carbonyl group of the amide side chain. Under thermal conditions of 140 to 180 degrees Celsius, the solid acid catalyst facilitates the generation of an electrophilic acylium-like species or activates the alkyl halide moiety for nucleophilic attack by the electron-rich aromatic ring. The specific pore structure and acidity of the HZSM-5 catalyst play a crucial role in directing the cyclization to occur ortho to the hydroxyl group, ensuring the formation of the desired quinolone ring system with high fidelity. This heterogeneous catalytic cycle avoids the formation of stable complexes between the product and the catalyst, which is a common issue with homogeneous Lewis acids like AlCl3 that require hydrolysis for product release. Consequently, the reaction proceeds cleanly to form the cyclic ketone structure without generating heavy metal contaminated sludge.
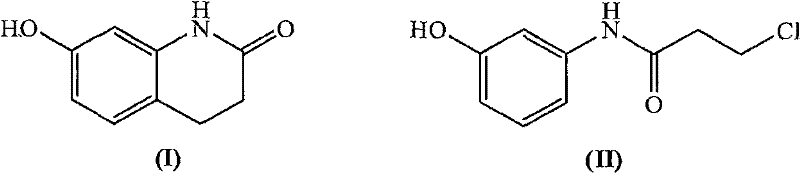
Furthermore, the control of impurities is inherently superior in this solid-state or melt-phase reaction environment. The absence of bulk liquid solvents during the critical cyclization step minimizes side reactions such as polymerization or intermolecular alkylation that often plague solution-phase Friedel-Crafts reactions. The precursor, N-(3-chloropropionyl)-3-hydroxyaniline, is itself synthesized under mild aqueous conditions using a phase-transfer catalyst, which ensures that the starting material entering the cyclization reactor is of high quality. Any unreacted precursor or minor byproducts are effectively separated during the subsequent hot filtration and cooling crystallization steps, where the distinct solubility differences between the product and impurities are exploited. This rigorous control over the reaction microenvironment ensures that the final commercial scale-up of complex heterocyclic intermediates meets the stringent quality standards required for GMP-grade API production.
How to Synthesize 7-Hydroxy-3,4-dihydroquinolone Efficiently
The practical implementation of this synthesis involves a carefully controlled sequence beginning with the preparation of the acyclic precursor in an aqueous medium, followed by the thermal cyclization step. Operators must maintain precise temperature control during the acylation phase, keeping the reaction between 5 and 10 degrees Celsius to prevent hydrolysis of the acid chloride, before heating the mixture for the cyclization. The detailed standardized synthesis steps, including specific molar ratios of sodium bicarbonate and phase-transfer catalysts, as well as the optimal loading of the molecular sieve, are critical for reproducibility. For a comprehensive guide on executing this protocol with maximum efficiency and safety, please refer to the technical instructions below.
- Prepare N-(3-chloropropionyl)-3-hydroxyaniline by reacting 3-hydroxyaniline with 3-chloropropionyl chloride in water using sodium bicarbonate and a phase-transfer catalyst at 5-10°C.
- Mix the intermediate with HZSM-5 molecular sieve catalyst and heat to 140-180°C to induce melting and intramolecular Friedel-Crafts alkylation.
- Elute the reaction mixture with a solvent like water or methanol, filter to recover the catalyst, and crystallize the filtrate to obtain high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this solid-acid catalyzed route presents a compelling value proposition centered on operational stability and long-term cost optimization. By transitioning away from processes that rely on hazardous, single-use reagents, manufacturers can significantly reduce the volatility associated with raw material sourcing and waste disposal logistics. The ability to recycle the catalyst directly translates to a lower cost of goods sold (COGS) over the lifecycle of the product, while the simplified purification train reduces energy consumption and processing time. This technological upgrade mitigates supply chain risks associated with environmental regulatory crackdowns on heavy metal discharge, ensuring uninterrupted production schedules for downstream API manufacturers.
- Cost Reduction in Manufacturing: The elimination of stoichiometric aluminum chloride removes the necessity for expensive quenching and neutralization steps, which traditionally consume large volumes of water and base. Additionally, the recovery and reuse of the HZSM-5 molecular sieve catalyst mean that the effective catalyst cost per kilogram of product approaches zero after the initial investment, driving substantial cost savings in pharmaceutical intermediates manufacturing. The high yield of the precursor synthesis in water further reduces solvent purchase and recovery costs, creating a leaner and more economical production model.
- Enhanced Supply Chain Reliability: Utilizing water as the primary solvent for the precursor step and minimizing organic solvent use in the cyclization step reduces dependency on volatile petrochemical-derived solvents, whose prices can fluctuate wildly. The robustness of the solid acid catalyst against moisture and air simplifies storage and handling requirements, reducing the risk of production delays due to reagent degradation. This stability ensures a consistent supply of high-quality intermediates, allowing partners to maintain tighter inventory controls and meet just-in-time delivery commitments with greater confidence.
- Scalability and Environmental Compliance: The process is inherently scalable because the exothermic nature of the Friedel-Crafts reaction is easier to manage in a melt or low-solvent system compared to large volume solution reactions. The drastic reduction in three-waste discharge, particularly the absence of aluminum-containing wastewater, simplifies the permitting process for new production lines and ensures compliance with increasingly strict global environmental standards. This eco-friendly profile enhances the brand reputation of the supplier and future-proofs the supply chain against tightening environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing clarity on how this technology compares to legacy methods. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this process into their existing manufacturing infrastructure.
Q: What are the primary advantages of using HZSM-5 molecular sieves over traditional AlCl3 catalysts?
A: The use of HZSM-5 molecular sieves eliminates the generation of massive amounts of acidic wastewater associated with stoichiometric aluminum chloride usage. Furthermore, the solid acid catalyst can be easily filtered, regenerated, and reused multiple times, significantly reducing raw material costs and environmental disposal fees.
Q: How does this process improve the regioselectivity for the 7-hydroxy isomer?
A: The solid acid catalyst provides a specific acidic environment that favors the intramolecular cyclization at the position ortho to the hydroxyl group, ensuring high selectivity for the 7-hydroxy-3,4-dihydroquinolone structure required for Aripiprazole synthesis, thereby minimizing difficult-to-remove isomeric impurities.
Q: Is the precursor synthesis step environmentally sustainable?
A: Yes, the precursor N-(3-chloropropionyl)-3-hydroxyaniline is synthesized in an aqueous medium using sodium bicarbonate as an acid binding agent, avoiding the use of volatile organic solvents and reducing the overall carbon footprint of the manufacturing process.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 7-Hydroxy-3,4-dihydroquinolone Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient intermediate synthesis in the global pharmaceutical supply chain. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and robust. We are committed to delivering high-purity materials that adhere to stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. By leveraging advanced catalytic technologies like the one described in CN101302195B, we empower our clients to accelerate their drug development timelines while maintaining the highest standards of quality and sustainability.
We invite you to collaborate with us to explore how this optimized synthetic route can benefit your specific project requirements. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can drive value and efficiency for your organization.
