Advanced Copper-Catalyzed Synthesis of E-Fluoroalkenyl Silanes for Commercial Scale-up
The landscape of organosilicon chemistry is undergoing a significant transformation driven by the urgent demand for efficient fluorination strategies in modern drug discovery. As detailed in the recent patent CN116284100A, a groundbreaking preparation method for monofluoroolefin silanes has been developed that addresses long-standing synthetic bottlenecks. Fluoroalkenyl silanes serve as critical building blocks, acting as electronic isosteres for amide bonds to enhance metabolic stability and conformational rigidity in bioactive molecules. This new technology leverages a copper-catalyzed decarboxylative silylation strategy, utilizing readily available fluoroacrylic acids and commercial hydrosilanes. For R&D directors and procurement specialists alike, this represents a paradigm shift from complex, multi-step sequences to a streamlined, one-pot operation that delivers high-purity E-configured products with exceptional stereoselectivity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of fluoroalkenyl silanes has been plagued by severe operational constraints and prohibitive costs that hinder large-scale adoption. Early methodologies, such as those reported in 2000, relied heavily on olefination reactions requiring harsh cryogenic conditions down to -78°C and the use of hazardous strong bases like butyllithium. 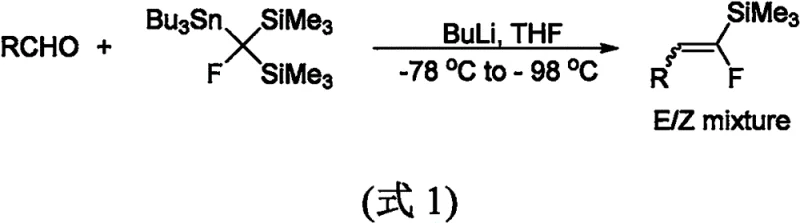 Furthermore, these legacy routes necessitated the pre-synthesis of specialized fluorine and silicon reagents, which are not only expensive but also difficult to store and handle safely. A more recent approach from 2018 attempted to improve this via defluorosilylation of gem-difluoroalkenes; however, it introduced new dependencies on costly iridium catalysts and required three equivalents of lithium tert-butoxide.
Furthermore, these legacy routes necessitated the pre-synthesis of specialized fluorine and silicon reagents, which are not only expensive but also difficult to store and handle safely. A more recent approach from 2018 attempted to improve this via defluorosilylation of gem-difluoroalkenes; however, it introduced new dependencies on costly iridium catalysts and required three equivalents of lithium tert-butoxide. 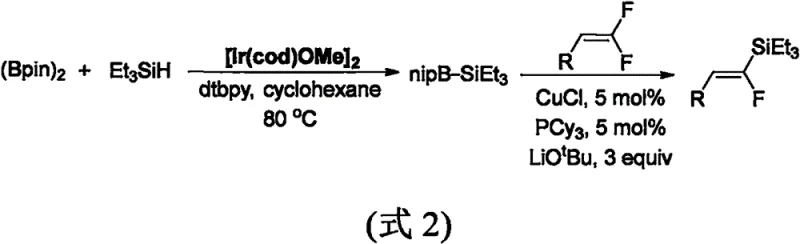 These conventional pathways often resulted in poor stereoselectivity, producing inseparable mixtures of E and Z isomers, thereby complicating purification and drastically reducing overall process efficiency for commercial manufacturing.
These conventional pathways often resulted in poor stereoselectivity, producing inseparable mixtures of E and Z isomers, thereby complicating purification and drastically reducing overall process efficiency for commercial manufacturing.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN116284100A offers a robust and economically viable alternative that bypasses these historical hurdles. By employing cuprous oxide as an inexpensive catalyst and di-tert-butyl peroxide as a radical initiator, the reaction proceeds smoothly in tert-butanol at a moderate temperature of 100°C. 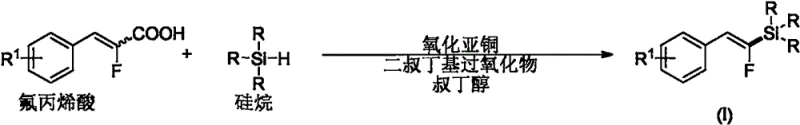 This novel approach eliminates the need for cryogenic cooling and avoids the use of pyrophoric bases entirely, significantly enhancing operational safety. Crucially, the reaction utilizes direct decarboxylative silylation of fluoroacrylic acids, meaning the starting materials are commodity chemicals rather than bespoke synthetic intermediates. This simplification of the supply chain, coupled with the ability to achieve high E-selectivity without complex chiral ligands, positions this technology as a superior choice for cost reduction in pharmaceutical intermediates manufacturing.
This novel approach eliminates the need for cryogenic cooling and avoids the use of pyrophoric bases entirely, significantly enhancing operational safety. Crucially, the reaction utilizes direct decarboxylative silylation of fluoroacrylic acids, meaning the starting materials are commodity chemicals rather than bespoke synthetic intermediates. This simplification of the supply chain, coupled with the ability to achieve high E-selectivity without complex chiral ligands, positions this technology as a superior choice for cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Cu-Catalyzed Decarboxylative Silylation
The core of this technological advancement lies in its unique radical-mediated mechanism, which diverges sharply from traditional ionic pathways. The reaction is initiated by the thermal decomposition of di-tert-butyl peroxide, generating tert-butoxy radicals that abstract hydrogen from the hydrosilane to produce silyl radicals. Concurrently, the cuprous oxide catalyst facilitates the single-electron oxidation of the fluoroacrylic acid substrate, promoting decarboxylation to generate a transient vinyl radical species. The convergence of these radical intermediates leads to the formation of the carbon-silicon bond with remarkable regio- and stereocontrol. Understanding this mechanism is vital for R&D teams, as it explains the tolerance for diverse functional groups, including esters, halides, and even complex vitamin E derivatives, without the side reactions typical of nucleophilic attacks.
From an impurity control perspective, the high stereoselectivity observed (E/Z > 30:1) is a direct result of the thermodynamic stability of the transition state during the radical coupling process. Unlike base-mediated eliminations that often yield thermodynamic mixtures, this radical pathway favors the formation of the sterically less hindered E-isomer. This intrinsic selectivity is a massive advantage for quality control, as it minimizes the burden on downstream purification units. For process chemists, this means that the crude reaction profile is much cleaner, reducing the load on silica gel columns and minimizing product loss during isolation. The ability to consistently produce high-purity fluoroalkenyl silanes ensures that downstream coupling reactions, such as Hiyama couplings, proceed with predictable kinetics and yields.
How to Synthesize E-Fluoroalkenyl Silanes Efficiently
The practical implementation of this synthesis is designed for ease of execution, making it accessible for both laboratory discovery and pilot plant operations. The protocol involves a straightforward charging sequence where the catalyst and substrate are mixed under an inert atmosphere, followed by the addition of solvent and silane. The reaction is then heated to reflux, allowing the radical cascade to proceed to completion over a standard 20-hour period. This simplicity removes the need for specialized low-temperature reactors or glovebox techniques, lowering the barrier to entry for contract manufacturing organizations.
- Load cuprous oxide catalyst and fluoroacrylic acid substrate into a sealed reaction vessel under inert atmosphere.
- Add tert-butanol solvent and hydrosilane reagent, then heat the mixture to 100°C for 20 hours.
- Remove solvent under reduced pressure and purify the crude residue via silica gel chromatography to isolate the E-isomer.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the economic implications of adopting this copper-catalyzed route are profound, offering substantial cost savings and risk mitigation. The shift from precious metal catalysts like iridium to abundant copper salts represents a direct reduction in raw material costs, while the elimination of cryogenic reagents lowers energy expenditures and safety compliance overheads. Furthermore, the reliance on commercially available fluoroacrylic acids and hydrosilanes ensures a stable and resilient supply chain, free from the bottlenecks associated with custom-synthesized reagents. This robustness is essential for maintaining continuity in the production of high-value active pharmaceutical ingredients.
- Cost Reduction in Manufacturing: The replacement of expensive iridium catalysts and specialized organolithium reagents with inexpensive cuprous oxide and commercial silanes drives down the Bill of Materials significantly. Additionally, the simplified workup procedure, which avoids complex quenching of strong bases, reduces labor hours and waste disposal costs associated with hazardous byproducts. The high atom economy of the decarboxylative process further contributes to overall process efficiency, ensuring that every kilogram of input translates effectively into valuable output.
- Enhanced Supply Chain Reliability: By utilizing commodity chemicals such as triethylsilane and substituted fluoroacrylic acids, manufacturers can source materials from multiple global suppliers, mitigating the risk of single-source dependency. The operational simplicity of running reactions at 100°C rather than -78°C allows for the use of standard glass-lined steel reactors, increasing equipment availability and reducing turnaround time between batches. This flexibility enables faster response to market demands and shorter lead times for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The absence of pyrophoric reagents and the use of tert-butanol as a relatively green solvent align with modern environmental, health, and safety (EH&S) standards. Scaling this process from grams to tons does not require exponential increases in safety infrastructure, as the thermal hazards are manageable with standard engineering controls. The high selectivity reduces solvent consumption during purification, lowering the facility's overall environmental footprint and simplifying regulatory filings for commercial scale-up of complex polymer additives or drug candidates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, aiming to clarify the operational benefits for potential partners.
Q: What are the primary advantages of this copper-catalyzed method over traditional lithiation routes?
A: Unlike traditional methods requiring cryogenic conditions (-78°C) and hazardous strong bases like butyllithium, this novel protocol operates at a mild 100°C using inexpensive cuprous oxide. This significantly reduces energy consumption and safety risks associated with handling pyrophoric reagents.
Q: How does this process impact the stereoselectivity of the final fluoroalkenyl silane product?
A: The reaction demonstrates exceptional stereocontrol, consistently yielding the E-configuration isomer with ratios exceeding 30:1. This high selectivity eliminates the need for difficult and yield-lossing separation of Z-isomers, streamlining downstream purification.
Q: Is this synthesis method scalable for industrial production of pharmaceutical intermediates?
A: Yes, the use of commercially available hydrosilanes and fluoroacrylic acids, combined with the absence of sensitive pre-formed reagents, makes this route highly amenable to scale-up. The simple operational parameters facilitate transition from gram-scale discovery to multi-kilogram manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fluoroalkenyl Silane Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this copper-catalyzed decarboxylative silylation technology for the next generation of fluorinated drugs and materials. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to market is seamless. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of fluoroalkenyl silane meets the exacting standards required for clinical and commercial applications.
We invite you to collaborate with us to leverage this cost-effective and scalable synthesis route for your specific project needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your target molecule, demonstrating exactly how this method can optimize your budget. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, and let us help you secure a competitive advantage in the global marketplace.
