Advanced One-Pot Synthesis of Polysubstituted 2-Hydroxy-1,4-Benzoxazine Derivatives for Commercial Scale-Up
The pharmaceutical and agrochemical industries are constantly seeking robust synthetic pathways to access complex heterocyclic scaffolds that serve as critical building blocks for bioactive molecules. Patent CN107382899B introduces a groundbreaking methodology for the preparation of polysubstituted 2-hydroxy-1,4-benzoxazine derivatives, a class of compounds renowned for their significant biological activities including insecticidal, bactericidal, and anti-inflammatory properties. This innovation addresses long-standing challenges in organic synthesis by providing a route that is not only chemically efficient but also operationally simple, utilizing readily available starting materials to achieve high yields under remarkably mild conditions. The strategic value of this technology lies in its ability to generate structural diversity that was previously difficult or impossible to access through conventional means, thereby opening new avenues for drug discovery and development.
From a commercial perspective, the adoption of this synthesis protocol represents a significant leap forward for manufacturers of high-purity pharmaceutical intermediates. The method eliminates the need for harsh reaction environments and complex multi-step sequences, directly translating to reduced operational costs and enhanced process safety. By leveraging hypervalent iodine chemistry, the process achieves rapid cyclization at room temperature, typically within a window of 2 to 10 hours, ensuring a streamlined workflow that is highly attractive for reliable pharmaceutical intermediate suppliers aiming to optimize their production pipelines. The broad substrate scope further ensures that this technology can be adapted for the synthesis of a wide array of analogues, supporting the diverse needs of R&D teams globally.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-hydroxy-1,4-benzoxazine derivatives has relied heavily on traditional methodologies that involve the reaction of substituted o-aminophenols or halogenated aromatic hydrocarbon derivatives with 1,2-dihalogenated alkanes and alpha-substituted halogenated acids or esters. These conventional routes are fraught with significant drawbacks that hinder their efficiency and scalability in a modern industrial setting. Primarily, these methods often require multiple synthetic steps, each necessitating separate isolation and purification procedures, which cumulatively lead to substantial material loss and decreased overall yields. Furthermore, the use of 1,2-dihalogenated alkanes introduces severe safety and environmental concerns due to their toxicity and potential carcinogenicity, complicating waste management and regulatory compliance.
In addition to safety hazards, the traditional approaches frequently demand harsh reaction conditions, such as elevated temperatures and strong bases, which can lead to the decomposition of sensitive functional groups and the formation of complex impurity profiles. This lack of chemoselectivity often results in difficult purification challenges, requiring extensive chromatographic separation that drives up production costs and extends lead times. For procurement managers and supply chain leaders, these inefficiencies translate into higher costs of goods sold (COGS) and unpredictable supply continuity. The inability of these older methods to accommodate diverse substituents also limits the chemical space available for medicinal chemists, restricting the optimization of biological activity in final drug candidates.
The Novel Approach
In stark contrast, the novel approach disclosed in patent CN107382899B utilizes a direct oxidative cyclization strategy that fundamentally reshapes the synthetic landscape for these valuable heterocycles. By employing N-(o-hydroxyaryl) enaminones as key precursors and reacting them with iodine compounds in the presence of a base, this method achieves the construction of the benzoxazine core in a single pot. The reaction proceeds smoothly at room temperature, eliminating the energy-intensive heating steps required by legacy processes. This mild condition not only preserves sensitive functional groups but also significantly simplifies the reaction setup, making it ideal for both laboratory-scale exploration and large-scale manufacturing.
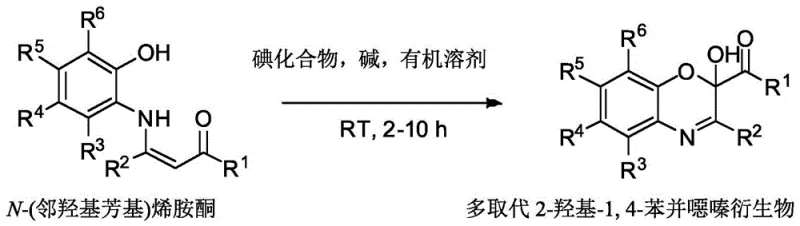
The versatility of this new methodology is exemplified by its exceptional tolerance for a wide range of substituents, allowing for the introduction of alkyl, aryl, halogen, nitro, and alkoxy groups at various positions on the molecular scaffold. As illustrated in the reaction scheme, the process accommodates diverse electronic and steric environments without compromising yield or selectivity. This capability is crucial for the commercial scale-up of complex pharmaceutical intermediates, as it enables the rapid generation of analogue libraries for structure-activity relationship (SAR) studies. Moreover, the post-treatment is remarkably straightforward, often involving simple solvent removal and washing, which drastically reduces the consumption of purification solvents and minimizes waste generation, aligning perfectly with green chemistry principles.
Mechanistic Insights into Iodine-Mediated Oxidative Cyclization
The core of this innovative synthesis lies in the mechanistic pathway facilitated by the iodine compound, likely acting as a hypervalent iodine species such as iodobenzene diacetate (PhI(OAc)2). The reaction initiates with the activation of the N-(o-hydroxyaryl) enaminone substrate, where the iodine reagent promotes the formation of a reactive intermediate that predisposes the molecule towards intramolecular cyclization. The phenolic oxygen atom acts as a nucleophile, attacking the activated carbon center adjacent to the nitrogen, leading to the formation of the C-O bond that closes the six-membered oxazine ring. This oxidative cyclization is highly efficient, driven by the thermodynamic stability of the resulting aromatic benzoxazine system and the mild oxidizing power of the iodine species.
Crucially, the choice of base plays a pivotal role in deprotonating the phenolic hydroxyl group, enhancing its nucleophilicity and accelerating the ring-closing step. Common bases such as triethylamine, potassium carbonate, or DBU are effective in this regard, ensuring that the reaction proceeds rapidly even at ambient temperatures. The mechanism is designed to be highly chemoselective, minimizing side reactions such as over-oxidation or polymerization that often plague oxidative transformations. This precision in reaction control is vital for maintaining stringent purity specifications required in API manufacturing, as it limits the formation of structurally related impurities that are difficult to remove. The result is a clean reaction profile that supports high isolated yields, often exceeding 80% for optimized substrates.
How to Synthesize Polysubstituted 2-Hydroxy-1,4-Benzoxazine Derivatives Efficiently
The practical implementation of this synthesis route is designed for ease of execution, requiring standard laboratory equipment and commonly available reagents. The process begins by charging a reaction vessel with the N-(o-hydroxyaryl) enaminone precursor, the chosen iodine oxidant, and an appropriate organic base. Solvents such as ethanol, methanol, acetonitrile, or toluene are suitable media, providing excellent solubility for the reactants while maintaining a benign environmental profile. The molar ratios are flexible, typically ranging from 1:0.3:1 to 1:2:3 for the enaminone, iodine compound, and base respectively, allowing for fine-tuning based on specific substrate reactivity. Detailed standardized synthesis steps are provided in the guide below.
- Combine N-(o-hydroxyaryl) enaminone, an iodine compound (such as iodobenzene acetate), and a base in an organic solvent like ethanol or acetonitrile.
- Stir the reaction mixture at room temperature for a duration of 2 to 10 hours to allow complete cyclization.
- Remove the organic solvent, dissolve the residue in ethyl acetate, wash with water, dry over anhydrous sodium sulfate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis method offers compelling economic and logistical benefits that directly impact the bottom line. The shift from multi-step, hazardous traditional routes to a streamlined one-pot process significantly reduces the complexity of the manufacturing workflow. This simplification translates into lower labor costs, reduced equipment occupancy time, and minimized utility consumption, as the reaction does not require prolonged heating or cryogenic cooling. The elimination of toxic 1,2-dihalogenated alkanes also removes the need for specialized containment and disposal protocols, further driving down operational expenditures and reducing the environmental footprint of the production facility.
- Cost Reduction in Manufacturing: The economic advantages of this method are substantial, primarily driven by the high atom economy and the use of inexpensive, commercially available starting materials. By avoiding expensive transition metal catalysts and complex ligand systems often required in cross-coupling alternatives, the raw material costs are kept low. Furthermore, the simplified workup procedure, which often bypasses the need for extensive column chromatography in favor of crystallization or simple washing, drastically reduces solvent usage and waste disposal fees. These factors combine to deliver a cost-effective manufacturing solution that enhances profit margins for cost reduction in pharmaceutical intermediate manufacturing.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness of the reaction conditions and the accessibility of the reagents. Since the synthesis operates at room temperature and tolerates a wide variety of substrates, it is less susceptible to batch-to-batch variability caused by minor fluctuations in process parameters. The use of stable iodine compounds and common organic bases ensures that raw material sourcing is reliable and not subject to the volatility associated with specialized or scarce reagents. This stability allows for consistent production scheduling and reducing lead time for high-purity pharmaceutical intermediates, ensuring that downstream customers receive their materials on time.
- Scalability and Environmental Compliance: Scaling this process from gram to ton quantities is straightforward due to the absence of exothermic hazards and the use of standard solvents. The mild conditions mean that heat transfer limitations, which often bottleneck scale-up efforts, are virtually non-existent. Additionally, the green nature of the process, characterized by reduced waste generation and the avoidance of heavy metals, aligns with increasingly stringent global environmental regulations. This compliance reduces the risk of regulatory shutdowns and facilitates smoother audits, making it a sustainable choice for long-term commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these benzoxazine derivatives. The answers are derived directly from the technical specifications and experimental data provided in the patent literature, ensuring accuracy and relevance for industry professionals. Understanding these details is essential for evaluating the feasibility of integrating this technology into existing production lines or R&D pipelines.
Q: What are the primary advantages of this synthesis method over conventional routes?
A: Unlike traditional methods that rely on hazardous 1,2-dihalogenated alkanes and multi-step sequences with low yields, this patented approach utilizes a one-pot oxidative cyclization at room temperature. It features readily available raw materials, short reaction times, and a simple, green post-treatment process.
Q: What is the substrate scope for this benzoxazine synthesis?
A: The method demonstrates exceptional versatility, accommodating a wide range of substituents including alkyl, aryl, halogens, nitro, and alkoxy groups on both the enaminone and the phenolic ring. This broad tolerance allows for the creation of diverse libraries of bioactive analogues.
Q: Is this process suitable for industrial scale-up?
A: Yes, the process is highly scalable due to its mild reaction conditions (room temperature), which minimize energy consumption and safety risks associated with exothermic heating. The simple workup procedure further enhances its feasibility for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted 2-Hydroxy-1,4-Benzoxazine Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced synthetic methodologies like the one described in CN107382899B for accelerating drug discovery and development. As a leading CDMO partner, we possess the technical expertise and infrastructure to seamlessly translate this laboratory-scale innovation into robust, commercial-grade manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify identity and potency.
We invite you to collaborate with us to leverage this cutting-edge chemistry for your next project. Whether you require custom synthesis of specific analogues or bulk supply of key intermediates, our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact our technical procurement team today to request specific COA data and discuss route feasibility assessments that can optimize your supply chain and reduce your time to market.
