Advanced Manufacturing of N-benzyl-3-pyrrolidone: A Technical Breakthrough for Global Supply Chains
The global demand for high-quality heterocyclic intermediates continues to surge, driven by the expanding pipeline of novel pharmaceutical and agrochemical active ingredients. In this context, patent CN102060743B represents a pivotal advancement in the efficient manufacturing of N-benzyl-3-pyrrolidone, a critical building block for various bioactive molecules. This technical disclosure outlines a streamlined four-step synthetic pathway that addresses historical bottlenecks associated with yield loss and operational complexity. By optimizing reaction parameters such as temperature control and molar ratios, the disclosed method achieves superior conversion rates compared to legacy technologies. For R&D directors and procurement strategists, understanding the nuances of this patent is essential for securing a competitive edge in the supply of reliable pharmaceutical intermediate supplier networks. The following analysis dissects the chemical ingenuity behind this process and its profound implications for cost-effective, large-scale production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-benzyl-3-pyrrolidone has been plagued by inefficient multi-step sequences that erode overall process economics. As referenced in prior art, such as patent 200710011673.7, traditional routes often involve the condensation of benzylamine with chloroacetic acid followed by esterification and subsequent reactions with acrylic acid. This convoluted pathway not only extends the production timeline but also suffers from suboptimal yields at critical junctures. Specifically, the ring-closing step in conventional methods typically achieves a yield of merely 65%, while the subsequent decarboxylation step reaches only about 85%. When compounded, these inefficiencies result in a cumulative yield for these two stages of approximately 55.3%, representing a massive loss of raw material value. Furthermore, the requirement for multiple isolation and purification steps between these transformations increases solvent consumption and waste generation, creating significant environmental and disposal cost burdens for manufacturing facilities.
The Novel Approach
In stark contrast, the methodology presented in CN102060743B introduces a robust and linear synthetic strategy that dramatically enhances material throughput. The innovation lies in the strategic reordering of bond formations, initiating with a highly efficient Michael addition followed by a selective alkylation. This approach bypasses the low-yielding bottlenecks of earlier techniques. For instance, the initial formation of ethyl 3-benzylaminopropionate proceeds with exceptional efficiency, yielding up to 96.4% under optimized thermal conditions. Similarly, the subsequent alkylation step to introduce the ester side chain maintains a high yield of roughly 94.3%. By securing such high conversion rates in the early stages, the process ensures that the maximum amount of valuable starting material flows into the final cyclization steps. This structural optimization not only simplifies the operational workflow but also fundamentally alters the cost basis of production, making it a preferred choice for cost reduction in fine chemical manufacturing.
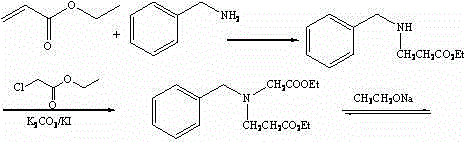
Mechanistic Insights into Base-Catalyzed Cyclization and Hydrolysis
The core chemical transformation in this synthesis relies on a sophisticated sequence of nucleophilic attacks and intramolecular condensations. The process begins with the Michael addition of benzylamine to ethyl acrylate, a reaction driven by the nucleophilicity of the amine nitrogen attacking the beta-carbon of the alpha,beta-unsaturated ester. This step is meticulously controlled at temperatures below 30°C during addition and maintained between 30-40°C during reaction to prevent polymerization of the acrylate while ensuring complete conversion. Following this, the secondary amine undergoes N-alkylation with ethyl chloroacetate in the presence of potassium carbonate and a catalytic amount of potassium iodide. The iodide ion acts as a nucleophilic catalyst, facilitating the displacement of the chloride leaving group through an SN2 mechanism, which is crucial for achieving the reported 94%+ yields without excessive heating that could degrade the sensitive ester functionalities.
The final construction of the pyrrolidone ring involves an intramolecular Claisen condensation, often referred to as a Dieckmann condensation in cyclic systems. Upon treatment with sodium ethoxide in anhydrous toluene, the alpha-proton of the newly introduced ethyl acetate moiety is abstracted to form an enolate. This enolate then attacks the carbonyl carbon of the adjacent propionate ester chain, closing the five-membered ring to form N-benzyl-4-ethoxycarbonyl-3-pyrrolidone. The subsequent hydrolysis and decarboxylation steps are equally critical; acidic hydrolysis cleaves the ester groups, and thermal treatment facilitates the loss of carbon dioxide to yield the final ketone. Throughout this sequence, impurity control is paramount. The patent specifies the use of GC and LC-MS monitoring to track reaction progress, ensuring that side products such as dialkylated amines or hydrolyzed acids are minimized before they can contaminate the final high-purity N-benzyl-3-pyrrolidone product.
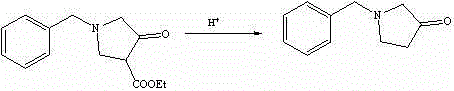
How to Synthesize N-benzyl-3-pyrrolidone Efficiently
Implementing this synthesis route requires precise adherence to the stoichiometric ratios and thermal profiles defined in the patent to replicate the high yields observed in the examples. The process is designed to be operationally simple, utilizing standard reactor equipment capable of handling vacuum distillation and exothermic additions. The key to success lies in the careful management of the exotherm during the initial amine-acrylate mixing and the strict exclusion of moisture during the sodium ethoxide cyclization step. Operators must ensure that the molar ratio of benzylamine to ethyl acrylate is maintained between 1:1.5 and 1:2.0 to drive the equilibrium forward while allowing for the recovery of excess amine. Detailed standardized operating procedures for each of the four distinct chemical transformations are outlined below to guide process engineers in scaling this technology.
- Perform Michael addition of benzylamine with ethyl acrylate at controlled temperatures (≤30°C to 40°C) to synthesize ethyl 3-benzylaminopropionate.
- Conduct N-alkylation using ethyl chloroacetate with potassium carbonate and potassium iodide catalyst at room temperature for 48-50 hours.
- Execute intramolecular cyclization using sodium ethoxide in anhydrous toluene, followed by acidic hydrolysis and decarboxylation to finalize the pyrrolidone ring.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers tangible strategic benefits beyond mere chemical elegance. The primary advantage is the substantial reduction in raw material waste, which directly correlates to lower variable costs per kilogram of finished product. By achieving yields in the mid-90% range for the first two steps, the process minimizes the volume of unreacted starting materials that must be separated, recycled, or disposed of. This efficiency translates into a leaner manufacturing footprint, requiring smaller reactor volumes to produce the same output tonnage compared to older, less efficient routes. Additionally, the use of commodity chemicals such as ethyl acrylate, benzylamine, and ethyl chloroacetate ensures that the supply chain remains resilient against shortages of exotic or specialized reagents. This reliance on widely available feedstocks significantly mitigates the risk of production delays caused by upstream supply disruptions.
- Cost Reduction in Manufacturing: The economic impact of this synthesis route is driven by the elimination of low-yielding steps that traditionally act as profit drains. By avoiding the 55% cumulative yield bottleneck found in prior art, the new method effectively nearly doubles the output from the same mass of input precursors in the critical ring-forming stages. This drastic improvement in atom economy means that the cost of goods sold (COGS) is significantly lowered, allowing for more competitive pricing in the global market. Furthermore, the simplified workup procedures, which rely on standard distillation and extraction rather than complex chromatography, reduce labor hours and utility consumption, contributing to overall operational expenditure savings.
- Enhanced Supply Chain Reliability: Stability in supply is often compromised by complex syntheses that are prone to failure or batch-to-batch variability. This robust four-step process enhances reliability by utilizing well-understood reaction types that are easy to control on a large scale. The mild reaction conditions, mostly operating below 40°C, reduce the stress on equipment and lower the probability of thermal runaways or safety incidents that could halt production lines. Moreover, the ability to recover and reuse excess benzylamine and ethyl chloroacetate through distillation creates a closed-loop system that further insulates the manufacturing process from raw material price volatility, ensuring consistent delivery schedules for downstream clients.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges regarding heat transfer and mixing, but this route is inherently scalable due to its homogeneous reaction phases and manageable exotherms. The use of toluene and ethyl acetate as solvents aligns with standard industrial practices, facilitating easy solvent recovery and recycling systems that meet modern environmental regulations. The reduction in waste generation, stemming from higher yields and fewer purification steps, lowers the burden on wastewater treatment facilities and reduces the carbon footprint of the manufacturing site. This alignment with green chemistry principles not only ensures regulatory compliance but also enhances the corporate sustainability profile for companies adopting this technology.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims within patent CN102060743B, providing a factual basis for decision-making. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing production portfolios. The clarity provided here aims to bridge the gap between laboratory-scale success and industrial-scale application.
Q: What is the primary advantage of the new synthesis route over conventional methods?
A: The new route described in patent CN102060743B significantly reduces the number of synthesis steps and improves overall yield. Specifically, the initial Michael addition step achieves yields exceeding 96%, and the alkylation step reaches over 94%, contrasting sharply with the lower cumulative yields of older multi-step processes.
Q: How does this process ensure high purity for pharmaceutical applications?
A: The process utilizes rigorous monitoring via Gas Chromatography (GC) and LC-MS at each stage. Furthermore, the final purification involves vacuum distillation collecting fractions at specific boiling points (145-150°C / 6mmHg), ensuring the removal of volatile impurities and unreacted starting materials to meet stringent pharmaceutical standards.
Q: Is this synthesis route scalable for industrial production?
A: Yes, the reaction conditions are mild and highly scalable. Key steps operate at moderate temperatures (30-40°C) and atmospheric or reduced pressure, utilizing common solvents like toluene and ethyl acetate. The absence of extreme cryogenic conditions or exotic catalysts facilitates straightforward commercial scale-up from pilot plants to multi-ton manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-benzyl-3-pyrrolidone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires deep technical expertise and robust infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this high-yield synthesis are fully realized in practice. We are committed to delivering high-purity N-benzyl-3-pyrrolidone that meets stringent purity specifications required by the pharmaceutical industry. Our rigorous QC labs employ advanced analytical techniques to verify the identity and purity of every batch, guaranteeing that our customers receive a product that is consistent, safe, and ready for immediate use in downstream API synthesis.
We invite global partners to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging our optimized manufacturing capabilities, we can help you reduce lead time for high-purity pharmaceutical intermediates while maintaining the highest standards of quality. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. Let us collaborate to secure your supply chain and drive innovation in your drug development pipeline through superior chemical manufacturing solutions.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
