Scalable Synthesis of Chiral (1S,5R)-Lactone Intermediates via Novel Organocatalytic Oxidation
Scalable Synthesis of Chiral (1S,5R)-Lactone Intermediates via Novel Organocatalytic Oxidation
The pharmaceutical industry constantly seeks robust, scalable, and cost-effective routes for synthesizing complex chiral building blocks, particularly those serving as critical precursors for bioactive molecules like prostaglandins. Patent CN108084129B introduces a groundbreaking methodology for the synthesis of (1S,5R)-lactone, a pivotal intermediate in the prostaglandin value chain, utilizing an enantioselective Baeyer-Villiger oxidation. This technology leverages a novel chiral spirocyclic phosphonic acid catalyst to transform substituted bicyclo[3.2.0]-hept-2-en-6-one substrates into high-value lactones with exceptional stereocontrol. By shifting away from traditional transition metal catalysis or cumbersome biological resolutions, this process offers a streamlined pathway that aligns perfectly with modern green chemistry principles and industrial efficiency standards. For R&D directors and procurement specialists alike, this patent represents a significant opportunity to optimize supply chains for high-purity pharmaceutical intermediates while mitigating the risks associated with heavy metal residues and complex purification workflows.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of (1S,5R)-lactone has been plagued by significant technical and economic hurdles that hindered its widespread industrial adoption. Early approaches relied heavily on classical resolution techniques, such as diastereomeric crystallization using chiral amines, which inherently suffer from a maximum theoretical yield of 50% and require multiple recycling steps to improve overall efficiency. Furthermore, biological methods employing microorganisms or enzymes, while capable of high enantioselectivity, often struggle with scalability, substrate specificity, and difficult downstream processing due to the presence of biological impurities. Transition metal-catalyzed routes, including those utilizing Zirconium-Salen or Rhodium complexes, have demonstrated moderate success but are frequently limited by low turnover numbers, expensive ligand systems, and the persistent challenge of removing trace metal contaminants to meet stringent pharmaceutical regulatory limits. These legacy methods collectively result in elevated production costs, extended lead times, and inconsistent batch-to-batch quality, creating a pressing need for a more reliable and economically viable synthetic strategy.
The Novel Approach
The methodology disclosed in CN108084129B fundamentally reimagines this chemical transformation by employing an organocatalytic system that bypasses the limitations of both biological and metallic catalysts. By utilizing a specifically designed chiral spirocyclic phosphonic acid, the process achieves an enantiomeric excess (ee) exceeding 95% under mild reaction conditions, effectively eliminating the need for energy-intensive cryogenic environments or high-pressure equipment. The use of aqueous hydrogen peroxide as the terminal oxidant not only serves as a cost-effective and atom-economical reagent but also ensures that the only byproduct generated is water, thereby simplifying waste management and enhancing the environmental profile of the manufacturing process. This approach allows for the direct conversion of readily available bicyclic ketones into the desired lactone architecture with high fidelity, offering a robust platform that can be easily adapted for the synthesis of various derivatives by modifying the substituent groups on the starting material. The operational simplicity and high selectivity of this new route position it as a superior alternative for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Chiral Spirocyclic Phosphonic Acid Catalyzed Baeyer-Villiger Oxidation
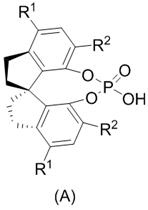
The core innovation of this technology lies in the unique activation mode provided by the chiral spirocyclic phosphonic acid catalyst, which facilitates the asymmetric oxygen insertion into the carbon-carbon bond adjacent to the carbonyl group. Unlike traditional Lewis acid catalysts that coordinate directly to the carbonyl oxygen, this Brønsted acid catalyst likely operates through a hydrogen-bonding network that activates the hydrogen peroxide oxidant while simultaneously organizing the substrate within a chiral pocket. This dual activation mechanism ensures that the nucleophilic attack of the peroxide occurs exclusively from one face of the planar ketone intermediate, thereby dictating the absolute stereochemistry of the resulting lactone ring. The rigid spirocyclic backbone of the catalyst provides the necessary steric bulk to differentiate between the enantiotopic faces of the substrate, leading to the observed high levels of enantioselectivity (>95% ee) even at relatively low catalyst loadings. Understanding this mechanistic nuance is crucial for R&D teams aiming to further optimize reaction parameters or adapt the protocol for structurally related analogs within the prostaglandin family.
![Reaction scheme showing the enantioselective Baeyer-Villiger oxidation of substituted bicyclo[3.2.0]-hept-2-en-6-one to (1S,5R)-lactone using H2O2 and chiral catalyst](/insights/img/1s5r-lactone-synthesis-pharma-intermediate-supplier-20260304122755-01.png)
Furthermore, the structural integrity of the catalyst plays a pivotal role in maintaining consistent performance throughout the reaction lifecycle. The specific substitution pattern on the aromatic rings of the catalyst, typically featuring bulky groups like triisopropylphenyl moieties, prevents non-productive aggregation and enhances solubility in common organic solvents such as chloroform. This structural design not only maximizes the catalytic turnover frequency but also facilitates the recovery and reuse of the catalyst, a feature that is often overlooked in academic studies but is paramount for industrial viability. The ability to recover the catalyst quantitatively means that the effective cost per kilogram of the final product is significantly reduced, as the expensive chiral information does not need to be replenished for every batch. For technical teams evaluating this process, the combination of high stereoselectivity and catalyst recyclability offers a compelling argument for adopting this technology over existing metal-based alternatives that require costly scavenging steps.
How to Synthesize (1S,5R)-Lactone Efficiently
Implementing this synthesis route requires careful attention to reaction conditions to maximize both yield and optical purity, although the protocol is designed to be operationally simple. The process begins with the dissolution of the substituted bicyclic ketone substrate and the chiral spirocyclic phosphonic acid catalyst in a suitable solvent, with chloroform identified as the optimal medium for balancing solubility and reaction rate. The addition of the oxidant, typically a 10-80% aqueous hydrogen peroxide solution, must be controlled to manage the exotherm and maintain the reaction temperature within the preferred range of -20°C to 0°C. Detailed standard operating procedures regarding stoichiometry, mixing rates, and workup protocols are essential for ensuring reproducible results on a manufacturing scale. The following guide outlines the critical steps for executing this transformation effectively, ensuring that the high standards required for pharmaceutical intermediate production are met consistently.
- Prepare the reaction mixture by combining substituted bicyclo[3.2.0]-hept-2-en-6-one, chiral spirocyclic phosphonic acid catalyst, and chloroform solvent in a dry reaction flask.
- Add 10-80% hydrogen peroxide solution slowly while maintaining the reaction temperature between -40°C and 25°C, preferably -20°C to 0°C.
- Stir the mixture for 24 to 72 hours, then quench with hydrochloric acid, extract with ether, and purify via recrystallization to obtain the high-purity lactone.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this organocatalytic Baeyer-Villiger oxidation offers transformative benefits that extend far beyond simple yield improvements. The elimination of precious metal catalysts such as rhodium or zirconium removes a significant variable cost component and mitigates the supply risk associated with fluctuating prices of rare earth elements. Moreover, the avoidance of heavy metals simplifies the regulatory compliance landscape, as there is no need for expensive and time-consuming metal scavenging resins or additional purification stages to meet ICH Q3D guidelines for elemental impurities. This streamlining of the downstream processing directly translates to reduced manufacturing cycle times and lower capital expenditure on specialized filtration equipment. For supply chain heads, the robustness of the reaction conditions and the stability of the reagents ensure a reliable flow of materials, minimizing the risk of production delays caused by sensitive catalyst deactivation or strict storage requirements.
- Cost Reduction in Manufacturing: The economic model of this process is strengthened by the quantitative recovery of the chiral catalyst, which allows the initial investment in the organocatalyst to be amortized over multiple production batches. Additionally, the use of hydrogen peroxide as a green oxidant replaces more hazardous and expensive peroxyacids like m-CPBA, which generate stoichiometric amounts of chlorobenzoic acid waste that requires disposal. By switching to a system where water is the primary byproduct, the facility can achieve substantial cost savings in waste treatment and solvent recovery operations. The overall reduction in unit operations, combined with the high atom economy of the oxidation step, drives down the total cost of goods sold, making the final API intermediate more competitive in the global market without compromising on quality standards.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as hydrogen peroxide and chloroform, rather than bespoke ligands or specialized biological cultures, ensures that the raw material supply chain is resilient and less prone to disruption. The synthetic route is insensitive to minor variations in moisture or oxygen levels compared to air-sensitive metal catalysts, allowing for more flexible manufacturing scheduling and reduced downtime. This operational robustness is critical for maintaining continuous supply to downstream API manufacturers, particularly during periods of high demand for cardiovascular or anti-inflammatory medications that utilize prostaglandin derivatives. The ability to source starting materials and reagents from multiple qualified vendors further de-risks the procurement strategy, ensuring long-term continuity of supply for this critical chiral building block.
- Scalability and Environmental Compliance: Scaling this reaction from laboratory to commercial production is facilitated by the mild thermal conditions and the absence of highly exothermic runaway risks often associated with peracid oxidations. The process generates minimal hazardous waste, aligning with increasingly stringent environmental regulations and corporate sustainability goals. The simplicity of the workup procedure, involving basic extraction and crystallization, allows for easy adaptation to large-scale reactor systems without the need for complex chromatography or specialized handling equipment. This ease of scale-up ensures that production volumes can be rapidly increased to meet market demands, from pilot plant quantities of 100 kgs to full commercial campaigns of 100 MT, while maintaining the high purity and stereochemical integrity required for pharmaceutical applications.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical aspects of adopting this technology. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers highlight the specific advantages in terms of purity, cost, and operational safety that distinguish this method from conventional alternatives.
Q: What are the key advantages of this organocatalytic method over traditional metal-catalyzed routes?
A: This method eliminates the need for expensive transition metals like Zirconium or Rhodium, significantly reducing heavy metal contamination risks and downstream purification costs while achieving superior enantioselectivity (>95% ee).
Q: Is the chiral spirocyclic phosphonic acid catalyst recoverable for industrial reuse?
A: Yes, the patent explicitly states that the chiral catalyst can be quantitatively recovered after the reaction, which drastically lowers the cost of goods sold (COGS) for large-scale manufacturing.
Q: What represents the primary safety benefit of using hydrogen peroxide in this oxidation process?
A: Using aqueous hydrogen peroxide replaces hazardous oxidants like m-CPBA, generating water as the only byproduct and simplifying waste treatment protocols for environmentally compliant production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (1S,5R)-Lactone Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of high-quality chiral intermediates in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. By leveraging advanced organocatalytic technologies like the one described in CN108084129B, we can offer our partners a supply of (1S,5R)-lactone that is not only chemically superior but also economically optimized for large-scale pharmaceutical manufacturing.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project requirements. Whether you are looking to reduce lead time for high-purity pharmaceutical intermediates or seeking a **Customized Cost-Saving Analysis** for your current supply chain, we are ready to provide the expertise and capacity you need. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to technical excellence can drive value for your organization.
