Advanced Synthesis of Ortho-Halogenated Phenyl Pyridine Ketones for Commercial Pharmaceutical Manufacturing
The pharmaceutical industry continuously seeks robust synthetic routes for critical intermediates, particularly those serving antiviral therapies such as Hepatitis B treatments. Patent CN110563641B introduces a groundbreaking methodology for the preparation of ortho-halogenated phenyl pyridine ketone compounds, which serve as pivotal building blocks in the synthesis of Lonafanib. This innovation leverages a palladium-catalyzed selective carbon-hydrogen halogenation strategy that fundamentally alters the economic and safety profile of producing these complex molecules. By utilizing dihalohydantoin reagents instead of traditional hazardous halogen sources, the process achieves high regioselectivity while mitigating environmental impact. The structural versatility of this approach allows for the introduction of various substituents, ensuring broad applicability across different drug development pipelines. For global procurement teams, this represents a significant opportunity to secure a reliable pharmaceutical intermediate supplier capable of delivering high-purity materials with consistent quality.
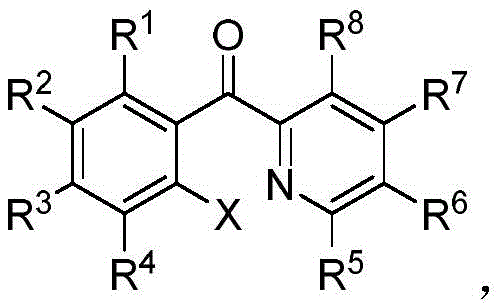
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic pathways for aromatic halides have long been plagued by significant operational hazards and inefficiencies that burden modern supply chains. Conventional methods often rely on elemental halogens, which are gaseous, highly volatile, and possess extreme toxicity, necessitating expensive containment infrastructure and rigorous safety protocols. Alternatively, the use of N-halosuccinimides suffers from poor atom economy, generating substantial stoichiometric waste that complicates waste management and increases disposal costs. Copper halide reagents introduce heavy metal contamination risks, requiring additional purification steps to meet stringent pharmaceutical purity standards. Furthermore, oxidative halogenation systems typically involve complex reaction mixtures with multiple oxidizing agents, leading to unpredictable side reactions and lower overall yields. These legacy technologies create bottlenecks in production capacity and elevate the total cost of ownership for manufacturers seeking cost reduction in API manufacturing.
The Novel Approach
The technology disclosed in patent CN110563641B presents a transformative solution by employing dibromohydantoin or dichlorohydantoin as the primary halogenating agents within a palladium-catalyzed framework. This novel approach capitalizes on the high active halogen content and exceptional storage stability of hydantoin derivatives, which are solid reagents that are far safer and easier to handle than gaseous alternatives. The reaction proceeds through a selective C-H activation mechanism that specifically targets the ortho-position of the diaryl ketone substrate, drastically reducing the formation of unwanted poly-halogenated impurities. By optimizing the catalyst loading and solvent system, the process achieves efficient conversion under moderate thermal conditions, enhancing energy efficiency. This method not only streamlines the synthetic route but also aligns with green chemistry principles, offering substantial cost savings through reduced raw material consumption and simplified downstream processing.
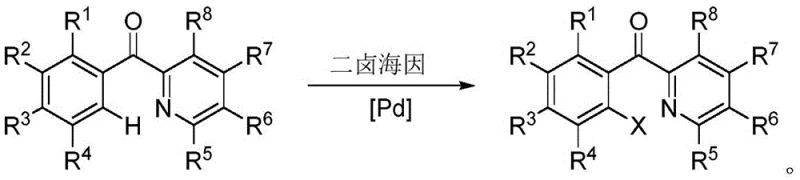
Mechanistic Insights into Pd-Catalyzed Selective C-H Halogenation
The core scientific advancement of this patent lies in its sophisticated catalytic cycle, which enables precise control over the halogenation site without requiring pre-functionalized substrates. The mechanism initiates with the coordination of the palladium catalyst to the nitrogen atom of the pyridine ring, acting as a directing group to position the metal center in proximity to the ortho-carbon hydrogen bond. This spatial arrangement facilitates the cleavage of the C-H bond through a concerted metalation-deprotonation pathway, forming a stable organopalladium intermediate. The dihalohydantoin reagent then interacts with this intermediate, transferring the halogen atom to the palladium center via an oxidative addition or ligand exchange process. Subsequent reductive elimination releases the desired ortho-halogenated product and regenerates the active palladium species, allowing the cycle to continue with high turnover numbers. This mechanistic understanding is crucial for R&D directors evaluating the feasibility of scaling this chemistry for commercial production of complex pharmaceutical intermediates.
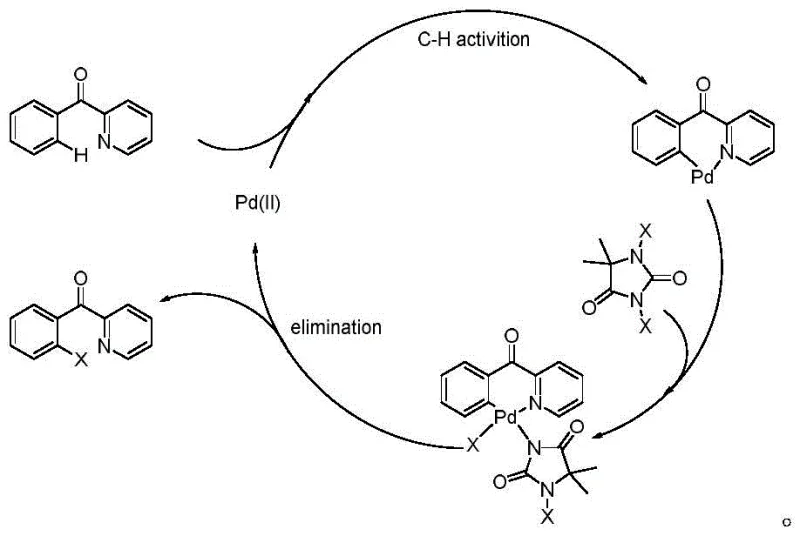
Impurity control is inherently built into the reaction design through the steric and electronic properties of the hydantoin reagent and the palladium catalyst system. Unlike free radical halogenation which often leads to random substitution patterns, this coordinated pathway ensures that halogenation occurs exclusively at the activated ortho-position directed by the pyridine nitrogen. The mild reaction conditions prevent thermal degradation of sensitive functional groups that might be present on the aromatic rings, preserving the integrity of the molecular scaffold. Furthermore, the use of dichloroethane as a preferred solvent enhances the solubility of both organic substrates and inorganic catalyst species, promoting homogeneous reaction kinetics. The result is a crude reaction mixture with a significantly cleaner profile, reducing the burden on purification units and minimizing product loss during isolation. This level of precision is essential for maintaining the stringent purity specifications required for intermediates used in antiviral drug synthesis.
How to Synthesize Ortho-Halogenated Phenyl Pyridine Ketone Efficiently
Implementing this synthesis route requires careful attention to reaction parameters to maximize yield and reproducibility on a manufacturing scale. The process begins with the precise weighing of phenylpyridyl ketone substrate and the dihalohydantoin reagent, ensuring the molar ratios align with the optimized conditions described in the patent examples. The selection of the solvent is critical, with dichloroethane demonstrating superior performance in terms of solubility and reaction rate compared to other polar or non-polar alternatives. Operators must maintain the reaction temperature within the specified range of 90-120°C to balance reaction kinetics with thermal stability, avoiding excessive heat that could degrade the catalyst. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction mixture by combining phenylpyridyl ketone substrate, dihalohydantoin halogenating agent, and Pd(OAc)2 catalyst in a suitable solvent such as dichloroethane.
- Heat the reaction mixture to a temperature range of 90-120°C and maintain stirring for a duration of 10 to 20 hours to ensure complete conversion.
- Purify the resulting ortho-halogenated diaryl ketone product using thin layer chromatography or standard industrial purification techniques to achieve high purity specifications.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented methodology addresses several critical pain points that traditionally impact the procurement of specialized chemical intermediates. The shift from hazardous gaseous halogens to stable solid hydantoin reagents significantly reduces the logistical complexities and regulatory burdens associated with transporting and storing dangerous goods. This enhancement in safety profile translates directly into lower insurance premiums and reduced need for specialized containment facilities, contributing to overall cost reduction in pharmaceutical intermediate manufacturing. Additionally, the high selectivity of the reaction minimizes the generation of difficult-to-separate by-products, which streamlines the purification process and improves overall material throughput. For supply chain heads, the robustness of this chemistry ensures consistent batch-to-batch quality, reducing the risk of production delays caused by out-of-specification materials. These factors collectively strengthen the resilience of the supply chain for critical antiviral medication components.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous halogen gases removes the need for complex gas handling infrastructure and scrubbing systems, leading to significant capital expenditure savings. Furthermore, the high atom economy of the dihalohydantoin reagents ensures that a greater proportion of the raw material mass is incorporated into the final product, reducing waste disposal costs. The simplified purification process resulting from high regioselectivity also lowers solvent consumption and energy usage during isolation. These cumulative efficiencies drive down the unit cost of production without compromising on the quality of the final intermediate.
- Enhanced Supply Chain Reliability: Dihalohydantoin reagents possess excellent storage stability and are readily available from multiple global chemical suppliers, mitigating the risk of raw material shortages. The solid nature of these reagents simplifies inventory management and reduces the shelf-life concerns associated with volatile liquid halogenating agents. By adopting a process that relies on stable and common chemicals, manufacturers can secure a more predictable supply timeline for their production schedules. This reliability is paramount for maintaining continuous manufacturing operations and meeting delivery commitments to downstream pharmaceutical clients.
- Scalability and Environmental Compliance: The reaction conditions are mild enough to be safely scaled from laboratory benchtop to multi-ton industrial reactors without requiring exotic high-pressure equipment. The reduced toxicity of the reagents and the minimized generation of heavy metal waste align with increasingly strict environmental regulations governing chemical manufacturing. This compliance reduces the administrative burden of environmental reporting and permits, facilitating faster approval for production scale-up. Consequently, the process supports sustainable manufacturing practices while maintaining high productivity levels for commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this halogenation technology. These answers are derived directly from the experimental data and beneficial effects documented in the patent specification to ensure accuracy. Understanding these details helps stakeholders assess the fit of this technology within their existing manufacturing frameworks. The responses cover aspects ranging from reagent selection to scalability potential.
Q: What are the advantages of using dihalohydantoin over traditional halogenating reagents?
A: Dihalohydantoin reagents offer superior storage stability, lower toxicity, and higher active halogen content compared to hazardous halogen gases or expensive N-halosuccinimides, significantly reducing safety risks and raw material costs.
Q: How does this patent address the issue of reaction selectivity in ortho-halogenation?
A: The method utilizes a palladium-catalyzed C-H activation mechanism that specifically targets the ortho-position relative to the carbonyl group, minimizing the formation of poly-halogenated by-products and simplifying downstream purification.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the process operates under relatively mild thermal conditions and uses commercially available solvents and catalysts, making it highly scalable for commercial manufacturing of hepatitis B drug intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ortho-Halogenated Phenyl Pyridine Ketone Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating advanced patent technologies like CN110563641B into commercial reality for our global partners. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab to plant is seamless and efficient. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of ortho-halogenated intermediates meets the exacting standards required for Hepatitis B drug synthesis. Our commitment to quality and safety makes us a trusted partner for multinational corporations seeking to optimize their supply chains.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into the potential economic advantages of switching to this greener, more efficient methodology. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your production needs. Let us collaborate to enhance the efficiency and sustainability of your pharmaceutical manufacturing operations.
