Scalable Catalyst-Free Synthesis of 2-Phenyl-3-Methylbenzofuran Pharmaceutical Intermediates
Scalable Catalyst-Free Synthesis of 2-Phenyl-3-Methylbenzofuran Pharmaceutical Intermediates
The landscape of medicinal chemistry is constantly evolving, driven by the urgent need for more sustainable and economically viable synthetic routes for complex heterocyclic scaffolds. A significant breakthrough in this domain is documented in Chinese Patent CN109384753B, which details a novel method for synthesizing 2-phenyl-3-methylbenzofuran compounds. These benzofuran derivatives are highly valued in the pharmaceutical industry due to their diverse biological activities, including potent antitumor, antidiabetic, and antioxidant properties. The patent introduces a groundbreaking approach that utilizes a thermal rearrangement of 3-benzofuran methyl phenyl ethers under catalyst-free and solvent-free conditions. This innovation addresses critical pain points in modern API manufacturing, specifically the reliance on scarce precious metals and the generation of hazardous waste, positioning it as a superior alternative for reliable pharmaceutical intermediate supplier networks seeking green chemistry solutions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 2-phenyl-3-methylbenzofuran scaffold has relied heavily on transition metal-mediated cross-coupling strategies, which present substantial logistical and financial burdens for large-scale production. For instance, earlier methodologies reported by the Gillmore group utilized palladium catalysts such as Pd2(dba)3 alongside specialized ligands like DPEphos to facilitate intramolecular C-O bond formation. As illustrated in the reaction scheme below, these processes typically require rigorous exclusion of air and moisture, expensive reagents, and generate significant amounts of metal-contaminated waste that necessitates costly purification steps.
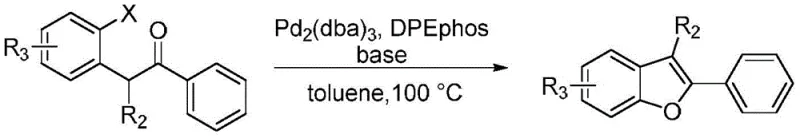
Furthermore, other reported methods involving rhodium-catalyzed C-H activation or titanium-mediated cyclization suffer from similar drawbacks, including narrow substrate scope, moderate yields, and the use of toxic chlorinated solvents. For a procurement manager, these factors translate into volatile supply chains and unpredictable cost structures, as the price of precious metals like palladium and rhodium fluctuates wildly on the global market. Additionally, the regulatory pressure to reduce residual heavy metals in final drug substances adds another layer of complexity and expense to these traditional routes, making them less attractive for commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
In stark contrast, the method disclosed in CN109384753B offers a streamlined, atom-economical pathway that bypasses the need for exogenous catalytic systems entirely. The core innovation lies in the thermal rearrangement of readily available 3-benzofuran methyl phenyl ether substrates. By simply heating the substrate, potentially with silica gel as a mild additive, the molecule undergoes a structural reorganization to form the desired 2-phenyl-3-methylbenzofuran core. This approach not only drastically simplifies the operational procedure but also aligns perfectly with the principles of green chemistry by eliminating solvent usage during the critical reaction phase.
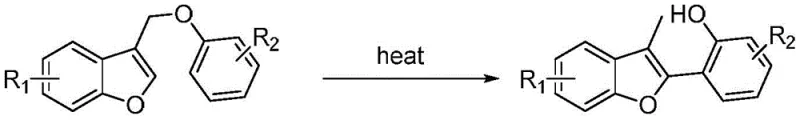
The versatility of this novel approach is evident in its ability to accommodate various substituents on the aromatic rings, allowing for the rapid generation of diverse analog libraries essential for drug discovery programs. For supply chain heads, this translates to a robust process that relies on stable, commodity-grade starting materials rather than sensitive, custom-synthesized catalysts. The elimination of complex catalytic cycles reduces the risk of batch failure due to catalyst deactivation, thereby enhancing the overall reliability and continuity of supply for high-purity pharmaceutical intermediates.
Mechanistic Insights into Thermal Rearrangement
The underlying mechanism of this transformation is believed to proceed through a concerted pericyclic rearrangement, akin to a Claisen rearrangement, followed by tautomerization or aromatization to restore the benzofuran system. In the absence of strong Lewis acids or transition metals, the reaction is driven purely by thermal energy, which overcomes the activation barrier for the migration of the benzyl group from the oxygen atom to the adjacent carbon position on the furan ring. The addition of silica gel, as demonstrated in the optimized examples, likely acts as a solid support that provides a large surface area for heat transfer and may offer weak Lewis acidic sites on its surface to slightly lower the activation energy, thereby improving reaction rates and yields without introducing metallic contaminants.
From an impurity control perspective, this mechanism is exceptionally clean. Traditional metal-catalyzed reactions often suffer from side reactions such as homocoupling, beta-hydride elimination, or incomplete conversion leading to difficult-to-remove metal-organic complexes. In this thermal rearrangement, the primary by-products are typically limited to unreacted starting material or minor decomposition products resulting from excessive heating, both of which are easily separated via standard chromatographic techniques. This inherent cleanliness ensures that the final product meets stringent purity specifications required for GMP manufacturing, reducing the burden on quality control labs and minimizing the risk of batch rejection due to out-of-specification impurity profiles.
How to Synthesize 2-Phenyl-3-Methylbenzofuran Efficiently
The practical implementation of this synthesis is remarkably straightforward, designed to be accessible even in facilities with limited specialized equipment. The process begins with the preparation of the ether substrate, followed by a simple thermal treatment. The patent highlights that while the reaction can proceed with just heat, the inclusion of silica gel significantly enhances the outcome, pushing yields from moderate levels up to nearly 80% in optimized cases. This simplicity makes it an ideal candidate for technology transfer and rapid scale-up.
- Dissolve the 3-benzofuran methyl phenyl ether substrate in a volatile solvent like dichloromethane, mix with silica gel, and evaporate to dryness.
- Heat the solid mixture to 120-160°C under stirring for 3-5 hours to promote the rearrangement reaction.
- Purify the cooled reaction mixture directly via flash column chromatography using petroleum ether and ethyl acetate to isolate the target product.
Commercial Advantages for Procurement and Supply Chain Teams
For decision-makers focused on the bottom line and operational efficiency, the shift from metal-catalyzed coupling to thermal rearrangement offers profound economic benefits. The most immediate impact is seen in the raw material costs, where the removal of palladium, rhodium, or titanium salts eliminates a major variable expense. Furthermore, the simplified workup procedure reduces the consumption of auxiliary chemicals such as scavengers, filtration aids, and extensive solvent volumes required for extraction and washing, leading to substantial cost savings in manufacturing overheads.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and specialized ligands directly lowers the Bill of Materials (BOM) cost per kilogram of product. Additionally, the solvent-free nature of the reaction step reduces the energy load associated with solvent recovery and distillation, while the simplified purification process minimizes silica gel and solvent usage during chromatography, collectively driving down the total cost of goods sold (COGS) for these valuable intermediates.
- Enhanced Supply Chain Reliability: By relying on thermally stable organic substrates rather than air-sensitive metal catalysts, the process becomes far more resilient to supply chain disruptions. There is no dependency on the volatile market for precious metals, and the starting materials are derived from common phenols and benzofuran methanols, which are widely available from multiple global vendors, ensuring a continuous and secure supply of high-purity intermediates for downstream API synthesis.
- Scalability and Environmental Compliance: The absence of toxic solvents and heavy metals during the reaction phase significantly eases the burden on waste treatment facilities. This green profile facilitates easier regulatory approval and reduces the environmental footprint of the manufacturing site. The process is inherently scalable, as the thermal rearrangement can be performed in standard heated reactors without the need for specialized high-pressure or inert atmosphere equipment, allowing for seamless expansion from pilot plant to commercial production scales.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthetic route. These insights are derived directly from the experimental data and technical disclosures within the patent documentation, providing a clear understanding of the process capabilities and limitations for potential partners and licensees.
Q: What are the primary advantages of this catalyst-free method over traditional palladium-catalyzed routes?
A: The primary advantage is the complete elimination of expensive transition metal catalysts (like Pd, Rh, or Ti) and toxic ligands, which significantly reduces raw material costs and simplifies the purification process by removing the need for heavy metal scavenging steps.
Q: Does this rearrangement reaction require hazardous organic solvents?
A: No, the core rearrangement step is conducted under solvent-free conditions. While a volatile solvent like dichloromethane is used initially to mix the substrate with silica gel, it is evaporated prior to heating, making the actual reaction phase green and environmentally benign.
Q: What is the typical yield range for this synthetic method?
A: According to the patent data, the method achieves yields ranging from 35% to 79% depending on the specific substrate substitution and reaction conditions, with optimized silica-gel promoted conditions reaching up to 79% yield.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Phenyl-3-Methylbenzofuran Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this catalyst-free technology in streamlining the production of complex heterocyclic building blocks. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 2-phenyl-3-methylbenzofuran delivered meets the highest standards of quality and consistency required by the global pharmaceutical industry.
We invite forward-thinking R&D and procurement leaders to collaborate with us to leverage this efficient synthetic route for your next drug development project. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and comprehensive route feasibility assessments, ensuring that your supply chain is optimized for both performance and profitability in the competitive landscape of API manufacturing.
