Advanced Aqueous-Phase Synthesis of 1,3-Substituted Thiazoles for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking more sustainable and cost-effective pathways for constructing privileged heterocyclic scaffolds. Patent CN107501204B introduces a groundbreaking methodology for synthesizing 1,3-substituted thiazole ring compounds directly from readily available styrene derivatives. This technical breakthrough addresses critical pain points in traditional heterocyclic synthesis by replacing hazardous organic solvents with water in the initial activation step. The process utilizes dibromohydantoin (DBH) as a mild brominating agent in the presence of a phase transfer catalyst, followed by a cyclization reaction with thiourea derivatives. For R&D directors and procurement managers alike, this represents a significant shift towards greener chemistry that does not compromise on yield or scalability. The ability to access diverse thiazole libraries using commodity chemicals like styrenes positions this technology as a cornerstone for next-generation API intermediate manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of thiazole derivatives has relied heavily on the Hantzsch thiazole synthesis or variations involving alpha-halo ketones, such as alpha-bromoacetophenone. While effective on a small laboratory scale, these conventional routes present substantial challenges for industrial application. The preparation of alpha-bromoacetophenone often requires the use of elemental bromine or other aggressive halogenating agents in corrosive acidic media, generating significant amounts of hazardous waste. Furthermore, the handling of these reactive intermediates poses serious safety risks during storage and transport, complicating supply chain logistics. Traditional methods also frequently suffer from poor atom economy and require rigorous purification to remove metal catalysts or toxic byproducts, driving up the overall cost of goods sold. These factors collectively create a bottleneck for manufacturers aiming to produce high-purity pharmaceutical intermediates in a compliant and economical manner.
The Novel Approach
In stark contrast, the novel approach detailed in the patent leverages the inherent reactivity of the vinyl group in styrene compounds to construct the thiazole core efficiently. By employing DBH in an aqueous medium, the process achieves selective bromination under mild conditions, avoiding the harsh environments associated with classical methods. The subsequent cyclization with thiourea proceeds smoothly in common organic solvents like ethanol, ensuring high conversion rates. This two-step sequence not only simplifies the operational workflow but also drastically reduces the environmental footprint of the synthesis. The versatility of this method is demonstrated by its tolerance to various substituents on the aromatic ring, including electron-withdrawing groups like nitro and halogens, as well as electron-donating groups like tert-butyl. 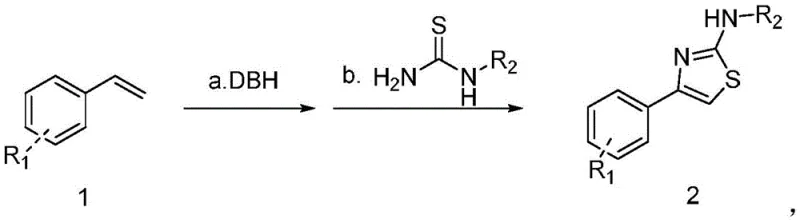
Mechanistic Insights into DBH-Mediated Bromination and Cyclization
The core of this synthetic strategy lies in the unique reactivity of dibromohydantoin (DBH) as a source of electrophilic bromine in water. In the presence of a phase transfer catalyst such as Tween or tetrabutylammonium bromide, DBH facilitates the formation of a bromonium ion intermediate across the double bond of the styrene substrate. This step is critical as it activates the alkene for nucleophilic attack without generating free radical species that could lead to polymerization or other side reactions. The aqueous environment plays a dual role: it acts as a green solvent and helps stabilize the ionic intermediates through solvation. The mild temperature of 60°C ensures that the reaction kinetics are favorable while preventing thermal degradation of sensitive functional groups. This controlled activation is key to achieving the high selectivity observed in the patent examples, where yields consistently exceed 80% across a broad substrate scope.
Following the initial bromination, the removal of water and subsequent dissolution in ethanol sets the stage for the cyclization event. The thiourea derivative acts as a bidentate nucleophile, attacking the activated carbon center to form the thiazole ring via a condensation mechanism. The use of ethanol as the solvent for this step is particularly advantageous as it dissolves both the organic intermediate and the thiourea reagent effectively, promoting homogeneous reaction conditions. The reflux conditions provide sufficient energy to drive the elimination of hydrogen bromide and finalize the aromatization of the thiazole ring. From an impurity control perspective, the mildness of the reagents minimizes the formation of poly-brominated byproducts or over-oxidized species, resulting in a cleaner crude reaction mixture. This inherent purity simplifies downstream processing, reducing the burden on purification teams and lowering solvent consumption during isolation.
How to Synthesize 1,3-Substituted Thiazoles Efficiently
Implementing this synthesis route requires careful attention to the stoichiometry of the reagents and the selection of the appropriate phase transfer catalyst. The patent specifies a molar ratio of styrene to DBH of approximately 1:1.5, ensuring complete conversion of the starting material while minimizing excess reagent waste. The reaction time is notably short, with the bromination step completing within 1 hour at 60°C, followed by a 1-hour reflux for cyclization. This rapid turnover time is highly beneficial for increasing throughput in a manufacturing setting. Operators should ensure efficient removal of water between steps to prevent hydrolysis of the intermediate or dilution of the ethanol solvent. Detailed standardized synthetic steps see the guide below.
- Dissolve styrene derivatives and a phase transfer catalyst in water, then add DBH and react at 60°C for 1 hour.
- Remove water under reduced pressure, dissolve the residue in an organic solvent like ethanol, and add thiourea derivatives.
- Reflux the mixture for 1 hour, then perform standard workup including extraction and column chromatography to isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this aqueous-based synthesis protocol offers tangible benefits in terms of cost structure and operational reliability. The primary driver of cost reduction is the substitution of expensive and hazardous specialty reagents with commodity chemicals. Styrene derivatives are produced on a massive global scale for the polymer industry, making them significantly cheaper and more readily available than specialized alpha-halo ketones. Additionally, the use of water as the solvent for the first reaction step eliminates the need for large volumes of volatile organic compounds (VOCs), leading to substantial savings in solvent procurement and waste disposal costs. The simplified workup procedure, which involves basic extraction and concentration, further reduces labor hours and equipment occupancy time, enhancing overall plant efficiency.
- Cost Reduction in Manufacturing: The elimination of heavy metal catalysts and corrosive acids from the process flow removes the necessity for expensive scavenging resins and specialized corrosion-resistant equipment. This shift allows manufacturers to utilize standard glass-lined or stainless steel reactors, significantly lowering capital expenditure requirements. Furthermore, the high atom economy of the reaction means that a greater proportion of the raw material mass is converted into the final product, reducing the effective cost per kilogram of the API intermediate. The reduction in hazardous waste generation also translates to lower environmental compliance fees and insurance premiums, contributing to a leaner overall cost base.
- Enhanced Supply Chain Reliability: Relying on widely available starting materials like styrenes and thioureas mitigates the risk of supply disruptions that often plague niche chemical markets. Since these feedstocks are produced by multiple vendors globally, procurement teams can negotiate better pricing and secure long-term contracts with greater confidence. The robustness of the reaction conditions, which tolerate a wide range of temperatures and minor variations in reagent quality, ensures consistent batch-to-batch performance. This reliability is crucial for maintaining continuous production schedules and meeting the just-in-time delivery expectations of downstream pharmaceutical clients.
- Scalability and Environmental Compliance: The inherent safety of using water and ethanol as solvents makes this process exceptionally easy to scale from pilot plant to commercial production. There are no exothermic runaway risks associated with elemental bromine handling, allowing for larger batch sizes without compromising safety protocols. The reduction in toxic emissions aligns perfectly with increasingly stringent global environmental regulations, future-proofing the manufacturing site against regulatory changes. This green chemistry approach not only satisfies corporate sustainability goals but also enhances the marketability of the final product to eco-conscious partners in the value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a reliable basis for feasibility assessments. Understanding these details is essential for R&D teams evaluating the route for potential adoption and for supply chain planners estimating production timelines. The clarity provided here aims to bridge the gap between academic innovation and industrial application.
Q: What are the primary advantages of using DBH over traditional brominating agents?
A: DBH (dibromohydantoin) offers a controlled release of bromine in aqueous media, eliminating the need for hazardous elemental bromine and reducing corrosive waste streams significantly.
Q: Can this synthesis method be scaled for industrial production?
A: Yes, the use of water as the primary solvent in the first step and mild reaction temperatures (60°C) makes this process highly suitable for large-scale commercial manufacturing with minimal safety risks.
Q: What is the typical purity profile of the resulting thiazole derivatives?
A: The method yields high-purity products with isolated yields often exceeding 80%, minimizing the need for complex purification steps compared to traditional routes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,3-Substituted Thiazoles Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this green synthesis pathway for the production of high-value thiazole intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the bench to the plant. Our facilities are equipped with state-of-the-art reactors capable of handling aqueous-organic biphasic systems efficiently, and our stringent purity specifications guarantee that every batch meets the rigorous demands of the pharmaceutical industry. With our rigorous QC labs and commitment to continuous improvement, we are positioned to deliver this advanced chemistry with the highest levels of quality and consistency.
We invite you to collaborate with us to leverage this cost-effective and environmentally friendly technology for your upcoming projects. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and target specifications. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can optimize your supply chain and accelerate your time to market.
