Scalable Synthesis of Epoxy-fused 2-methylene Pyrrolidines for Advanced Pharmaceutical Intermediates
The pharmaceutical industry continuously seeks efficient pathways to construct complex nitrogen-containing heterocycles, which serve as the structural backbone for numerous bioactive molecules. Patent CN112500419A introduces a groundbreaking methodology for the preparation of epoxy-fused 2-methylene pyrrolidine compounds, a privileged scaffold found in potent natural products and drug candidates. This innovation addresses critical bottlenecks in synthetic organic chemistry by providing a route that is not only operationally simple but also exhibits exceptional functional group tolerance. The significance of this structural motif is underscored by its presence in biologically active agents such as (-)-slaframine and ABBV-3221, highlighting the urgent demand for reliable access to these intermediates.
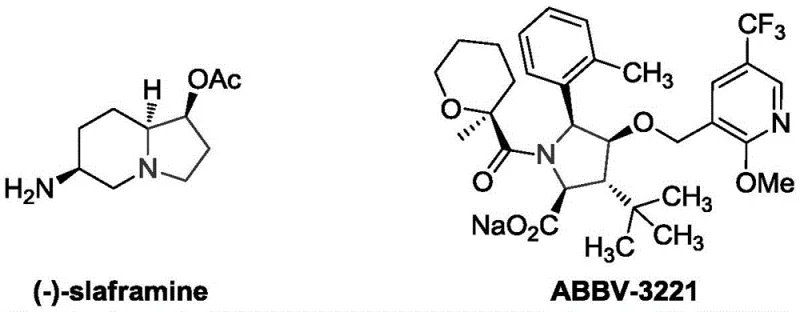
As a leading entity in the fine chemical sector, we recognize that the ability to rapidly construct these five-membered nitrogen heterocycles is paramount for accelerating drug discovery pipelines. The disclosed technology leverages a unique tandem reaction strategy involving sulfonyl-protected beta-aminoketone derivatives and prop-2-alkynyl sulfonium salts. By shifting away from hazardous and expensive reagents towards stable, commercially accessible starting materials, this patent lays the foundation for a more sustainable and cost-effective supply chain for high-purity pharmaceutical intermediates. The following analysis details how this technological leap translates into tangible value for R&D and procurement stakeholders.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of epoxy-fused 2-methylene pyrrolidine units has been fraught with synthetic challenges that hinder scalability and increase production costs. Prior art, such as the strategy reported by the Borhan group, relied on a one-pot tandem aza-Payne/hydroamination reaction starting from aziridinol. While chemically elegant, this conventional approach necessitates the use of Grignard reagents, which are notoriously sensitive to moisture and require stringent anhydrous conditions. Furthermore, the preparation of the initial aziridinol raw materials is relatively complex, often demanding precise stereochemical control that limits substrate scope.
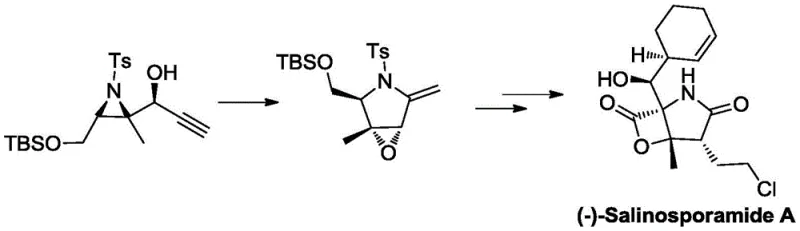
The reliance on such reactive organometallic species introduces significant safety risks and operational complexities, particularly when attempting to transition from milligram-scale laboratory synthesis to kilogram-scale commercial manufacturing. Additionally, the post-reaction treatment processes associated with these traditional methods are complicated, often requiring extensive workup procedures to remove metal residues and byproducts. These factors collectively contribute to longer lead times and higher manufacturing expenses, creating a substantial barrier for pharmaceutical companies seeking reliable suppliers for these critical building blocks.
The Novel Approach
In stark contrast, the methodology disclosed in CN112500419A offers a streamlined and robust alternative that circumvents the pitfalls of organometallic chemistry. The core of this innovation lies in the reaction between a sulfonyl-protected beta-aminoketone derivative and a prop-2-alkynyl sulfonium salt compound under the influence of a mild inorganic base. This transformation proceeds efficiently in common organic solvents like dichloromethane at temperatures ranging from 0°C to 100°C, with optimal results observed at near-ambient or cooled conditions. The reaction mechanism facilitates the rapid assembly of the bicyclic framework without the need for transition metal catalysts or pyrophoric reagents.
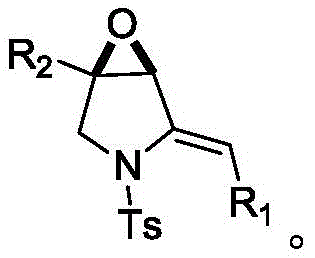
This novel approach dramatically simplifies the synthetic workflow, allowing for the direct conversion of readily available precursors into the target epoxy-fused structures with excellent yields. The versatility of this method is evidenced by its compatibility with a wide array of substituents, including various substituted phenyl groups, alkyl chains, and heterocyclic aryl moieties. By eliminating the need for complex protecting group manipulations and harsh reaction environments, this process not only enhances safety but also significantly reduces the environmental footprint of the manufacturing process, aligning perfectly with modern green chemistry principles.
Mechanistic Insights into Base-Mediated Cyclization
The success of this synthesis hinges on the precise interplay between the nucleophilic nitrogen center and the electrophilic sulfonium salt. The reaction initiates with the deprotonation of the sulfonyl-protected beta-aminoketone by the base, generating a reactive nucleophile that attacks the propargylic position of the sulfonium salt. This step is critical as it establishes the carbon-nitrogen bond necessary for ring closure. The use of cesium carbonate (Cs2CO3) is particularly advantageous due to its optimal basicity and solubility profile in organic media, which promotes the reaction kinetics without inducing unwanted side reactions or decomposition of the sensitive epoxide moiety.
Following the initial alkylation, an intramolecular cyclization occurs, driven by the nucleophilic attack of the oxygen atom on the activated alkyne or allene intermediate, ultimately forming the strained oxirane ring fused to the pyrrolidine core. The mild reaction conditions, specifically the preference for temperatures between 0°C and 10°C, are crucial for controlling the stereoselectivity and preventing the polymerization of the reactive methylene group. This mechanistic pathway ensures high purity of the final product, as the absence of heavy metal catalysts eliminates the risk of metal contamination, a critical quality attribute for pharmaceutical intermediates intended for clinical use.
How to Synthesize Epoxy-fused 2-methylene Pyrrolidine Efficiently
To implement this cutting-edge synthesis in a laboratory or pilot plant setting, operators must adhere to specific protocol parameters to maximize yield and reproducibility. The process involves dissolving the key reactants in a suitable solvent system and maintaining strict temperature control throughout the reaction duration. Detailed standard operating procedures regarding stoichiometry, mixing rates, and purification techniques are essential for consistent outcomes. For a comprehensive breakdown of the standardized synthesis steps, please refer to the technical guide below.
- Dissolve the sulfonyl-protected beta-aminoketone derivative, cesium carbonate base, and prop-2-alkynyl sulfonium salt compound in dichloromethane solvent.
- Stir the reaction mixture overnight at a controlled temperature range of 0-10°C until the starting materials are fully consumed.
- Filter the reaction system, concentrate the filtrate under reduced pressure, and purify the residue via silica gel column chromatography to isolate the target compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers profound strategic benefits that extend beyond mere chemical efficiency. The shift from complex, multi-step organometallic sequences to a direct, one-pot cyclization fundamentally alters the cost structure of producing these valuable intermediates. By utilizing commercially available sulfonium salts and simple ketone derivatives, manufacturers can secure a more stable and predictable supply of raw materials, mitigating the risks associated with sourcing specialized or custom-synthesized reagents.
- Cost Reduction in Manufacturing: The elimination of expensive noble metal catalysts and pyrophoric Grignard reagents results in substantial cost savings on raw material procurement. Furthermore, the simplified post-reaction workup, which avoids complex metal scavenging steps, reduces the consumption of auxiliary chemicals and lowers waste disposal costs. This streamlined process inherently lowers the overall cost of goods sold (COGS), allowing for more competitive pricing in the global market for high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The robustness of this chemistry against variations in reaction conditions ensures high batch-to-b consistency, which is vital for maintaining continuous supply lines. Since the raw materials are commodity chemicals rather than bespoke synthons, the risk of supply disruption due to vendor capacity issues is significantly minimized. This reliability enables pharmaceutical partners to plan their production schedules with greater confidence, reducing the need for excessive safety stock and optimizing inventory management.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of hazardous heavy metals make this process highly amenable to scale-up from gram to ton quantities without requiring specialized containment infrastructure. The reduced generation of toxic waste streams simplifies regulatory compliance and environmental permitting, facilitating faster approval for commercial production facilities. This scalability ensures that the supply chain can flexibly respond to increasing market demand for API intermediates without compromising on quality or safety standards.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is crucial for stakeholders evaluating its integration into their existing workflows. The following questions address common inquiries regarding the practical application and scope of this technology, drawing directly from the experimental data provided in the patent documentation. These insights are designed to clarify the operational feasibility and potential limitations of the method.
Q: What are the key advantages of this new synthesis method over traditional routes?
A: Unlike traditional methods requiring complex Grignard reagents and strict stereochemical control, this novel approach utilizes commercially available sulfonium salts and mild base conditions, eliminating the need for heavy metal catalysts and simplifying post-reaction purification.
Q: What represents the optimal reaction condition for maximum yield?
A: Experimental data indicates that using dichloromethane as the solvent with cesium carbonate as the base at a temperature range of 0-10°C provides the highest yields, often exceeding 80% for various substituted substrates.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process features simple operation, readily available raw materials, and excellent functional group tolerance, making it highly amenable to commercial scale-up without the safety hazards associated with pyrophoric reagents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Epoxy-fused 2-methylene Pyrrolidine Supplier
The technological advancements detailed in patent CN112500419A represent a significant opportunity for the pharmaceutical industry to access complex heterocyclic building blocks more efficiently. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to leverage such innovative synthetic routes for the benefit of our global clients. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of large-scale API manufacturing. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs and advanced analytical capabilities.
We invite forward-thinking pharmaceutical companies to collaborate with us to explore the full potential of this synthesis technology. By partnering with our technical procurement team, you can obtain a Customized Cost-Saving Analysis tailored to your specific project needs. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our optimized manufacturing processes can drive value and accelerate your drug development timelines.
