Scalable Production of 2-Amino Fused Pyridines Using Cost-Effective Iron Catalysis
The pharmaceutical and fine chemical industries are constantly seeking robust, cost-effective methodologies for constructing complex heterocyclic scaffolds, particularly those serving as critical intermediates for drug discovery. Patent CN113336702A introduces a groundbreaking preparation method for 2-amino fused pyridine compounds, a structural motif prevalent in numerous bioactive molecules targeting neurodegenerative diseases, receptor antagonists, and enzyme inhibitors. This innovation represents a paradigm shift from traditional transition metal catalysis by utilizing inexpensive iron compounds to drive a multi-step free radical cascade reaction. The technology enables the one-step construction of these valuable heterocycles from readily available benzoyl oxime esters and malononitrile-derived olefins, achieving reaction yields as high as 90%. For R&D directors and procurement managers alike, this development signals a move towards more sustainable and economically viable manufacturing processes for high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-amino fused pyridines has been fraught with significant technical and economic challenges that hinder efficient commercial scale-up. Conventional routes often rely on expensive precious metal catalysts such as palladium or rhodium, which not only inflate raw material costs but also necessitate rigorous and costly post-reaction purification steps to remove trace metal residues to meet stringent pharmaceutical standards. Furthermore, many existing methods require harsh reaction conditions, including high temperatures and high pressures, which pose safety risks and limit the compatibility of sensitive functional groups. The substrates used in these traditional pathways, often derived from 2-substituted quinoline compounds, tend to have poor stability and narrow selection ranges, making the synthesis of diverse analog libraries for drug screening difficult and time-consuming. These factors collectively contribute to extended lead times and reduced overall process efficiency in the manufacturing of complex organic molecules.
The Novel Approach
In stark contrast, the methodology disclosed in CN113336702A offers a streamlined, atom-economical solution that directly addresses these pain points. By employing cheap iron compounds like ferric acetylacetonate (Fe(acac)3) or ferric chloride (FeCl3) as catalysts, the process drastically reduces catalyst costs while maintaining high catalytic activity. The reaction proceeds via a radical cascade mechanism under argon protection in 1,4-dioxane at moderate temperatures ranging from 100°C to 120°C, eliminating the need for high-pressure equipment. This approach allows for the direct use of stable, industrially commoditized raw materials that are easy to store and handle. The ability to construct the 2-amino fused pyridine core in a single step from simple precursors significantly shortens the synthetic route, thereby enhancing overall throughput and reducing waste generation, which is crucial for green chemistry initiatives in modern chemical manufacturing.
Mechanistic Insights into Fe-Catalyzed C-H Bond Activation
The core of this technological breakthrough lies in the innovative utilization of iron-catalyzed C-H bond activation of inert benzoyl oxime ester compounds. The mechanism initiates with the interaction between the iron catalyst and the oxime ester, generating an iminyl radical species through homolytic cleavage. This highly reactive intermediate then undergoes a series of intramolecular and intermolecular radical additions and cyclizations with the malononitrile-derived olefin. This cascade sequence effectively forms multiple chemical bonds, including critical C-N and C-C bonds, in a concerted fashion. The use of iron facilitates this redox cycle efficiently, allowing for the rapid assembly of the fused pyridine ring system with high regioselectivity. Understanding this mechanistic pathway is vital for process chemists aiming to optimize reaction parameters for specific substrate classes.
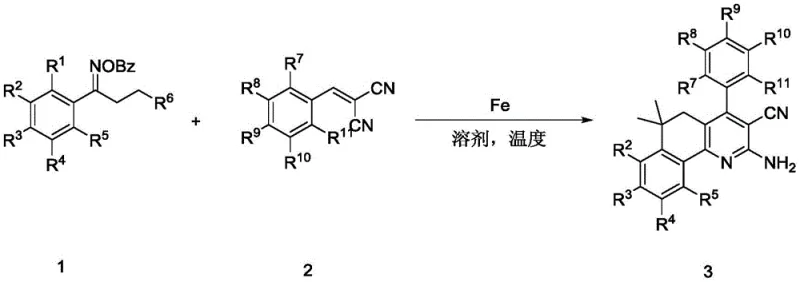
Furthermore, the reaction exhibits exceptional tolerance towards various substituents on both the oxime ester and the olefin components. The radical nature of the transformation allows it to proceed smoothly even in the presence of electron-withdrawing groups like halogens, esters, and nitriles, as well as electron-donating groups such as methoxy and methyl. This broad substrate scope is essential for medicinal chemists who need to rapidly generate diverse libraries of analogs to explore structure-activity relationships (SAR). The mechanism ensures that impurities are minimized due to the high selectivity of the radical cascade, resulting in cleaner reaction profiles that simplify downstream purification. This level of control over the chemical outcome is a key factor in ensuring the consistent quality required for reliable pharmaceutical intermediate supplier operations.
How to Synthesize 2-Amino Fused Pyridines Efficiently
The practical implementation of this synthesis is straightforward and designed for reproducibility in both laboratory and pilot plant settings. The standard protocol involves charging a reaction vessel with the benzoyl oxime ester and the malononitrile olefin in a molar ratio that favors the formation of the desired product, typically using a slight excess of the oxime component. The iron catalyst is added in catalytic amounts, usually around 10 mol%, along with a stoichiometric amount of sodium pivalate (PivONa) which acts as an additive to facilitate the reaction. The mixture is then heated in 1,4-dioxane under an inert atmosphere. Detailed operational parameters, including specific temperature gradients and workup procedures, are critical for maximizing yield and purity.
- Charge a reaction vessel with benzoyl oxime ester compound, malononitrile-derived olefin, and an iron catalyst such as Fe(acac)3 under inert gas protection.
- Add 1,4-dioxane as the solvent and heat the mixture to between 100°C and 120°C for approximately 12 hours while monitoring progress via TLC.
- Upon completion, cool the reaction, filter through diatomaceous earth, wash with ethyl acetate, and purify the crude product via flash column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this iron-catalyzed technology translates into tangible strategic benefits that go beyond mere chemical efficiency. The shift from precious metals to base metals like iron represents a fundamental change in the cost structure of raw materials, decoupling production expenses from the volatile pricing of rare earth elements. Additionally, the stability and commercial availability of the starting materials ensure a resilient supply chain that is less susceptible to disruptions. The simplified reaction workflow reduces the burden on manufacturing facilities, allowing for faster turnaround times and more flexible production scheduling to meet market demands.
- Cost Reduction in Manufacturing: The replacement of expensive noble metal catalysts with inexpensive iron salts results in a drastic reduction in direct material costs. Moreover, the elimination of complex heavy metal scavenging steps simplifies the purification process, reducing solvent consumption and waste disposal fees. The high atom economy of the one-step cascade reaction means less raw material is wasted as byproducts, further driving down the cost per kilogram of the final API intermediate. These cumulative savings can be passed down the supply chain, offering a competitive pricing advantage in the global market.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, specifically benzoyl oxime esters and malononitrile derivatives, are established industrial commodities with widespread availability from multiple vendors. Their stable chemical nature allows for long-term storage without specialized conditions, reducing inventory management risks. This robustness ensures a continuous and reliable flow of materials, minimizing the risk of production stoppages due to raw material shortages. For supply chain planners, this reliability is crucial for maintaining consistent delivery schedules to downstream pharmaceutical clients.
- Scalability and Environmental Compliance: The reaction conditions are mild and do not require high-pressure reactors, making the process inherently safer and easier to scale from gram to multi-ton quantities. The use of 1,4-dioxane as a solvent is standard in the industry, and the workup involves simple filtration and chromatography, which are easily adaptable to large-scale continuous processing. Furthermore, the high efficiency and selectivity of the reaction minimize the generation of hazardous waste, aligning with increasingly strict environmental regulations and sustainability goals. This ease of scale-up facilitates the rapid commercialization of new drug candidates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from the specific experimental data and beneficial effects outlined in the patent documentation to provide clarity on process feasibility and product quality. Understanding these details helps stakeholders make informed decisions regarding the integration of this method into their existing manufacturing portfolios.
Q: What are the primary advantages of using iron catalysts over precious metals for this synthesis?
A: Iron catalysts like Fe(acac)3 are significantly cheaper and more abundant than palladium or rhodium. Furthermore, they eliminate the need for expensive and complex heavy metal removal steps, simplifying downstream purification and reducing overall manufacturing costs.
Q: Does this method support a wide range of functional groups on the substrate?
A: Yes, the protocol demonstrates excellent functional group tolerance. It successfully accommodates substrates containing halogens, esters, methoxy groups, nitriles, and even complex heterocycles like thiophenes and benzothiazoles without compromising yield.
Q: Is this process suitable for large-scale commercial production?
A: Absolutely. The reaction utilizes stable, industrially available raw materials and operates under relatively mild conditions (100-120°C) without high pressure. The simple workup procedure involving filtration and chromatography makes it highly amenable to scale-up from kilogram to multi-ton quantities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Amino Fused Pyridines Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the iron-catalyzed synthesis described in CN113336702A for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent, high-quality materials regardless of volume. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 2-amino fused pyridines adheres to the highest industry standards. We are committed to leveraging advanced catalytic technologies to deliver superior value to the global pharmaceutical community.
We invite you to collaborate with us to explore how this cost-effective synthesis route can optimize your supply chain and reduce your overall manufacturing expenses. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project needs. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, and let us help you accelerate your drug development timeline with reliable, scalable solutions.
