Advanced Green Synthesis of Polysubstituted Nicotinic Acid Esters for Commercial Scale-up
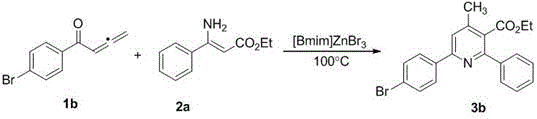
The pharmaceutical and fine chemical industries are constantly seeking more efficient and environmentally benign pathways to construct complex heterocyclic scaffolds, which serve as the backbone for numerous bioactive molecules. Patent CN107721917B introduces a groundbreaking green synthesis method for polysubstituted nicotinic acid ester compounds, addressing critical challenges in modern organic synthesis. This technology leverages the unique properties of acidic ionic liquids, specifically [Bmim]ZnBr3 (1-methyl-4-butylimidazolium tribromide zinc salt), to facilitate a tandem cyclization reaction between substituted enamine esters and 1,2-allenone compounds. Unlike traditional methods that often rely on toxic volatile organic solvents and harsh Lewis acids, this novel approach operates under mild conditions at 100°C, offering a sustainable alternative for the production of high-value pharmaceutical intermediates. The significance of this innovation lies not only in its environmental profile but also in its operational simplicity and broad substrate compatibility, making it a highly attractive candidate for industrial adoption by forward-thinking chemical manufacturers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of polysubstituted nicotinic acid esters has been plagued by several inherent drawbacks that hinder large-scale commercial viability. Conventional protocols frequently necessitate the use of stoichiometric amounts of hazardous reagents and strong acids, which generate substantial quantities of corrosive waste streams requiring expensive neutralization and disposal procedures. Furthermore, many existing methods suffer from limited substrate scope, often failing to tolerate sensitive functional groups such as halogens or trifluoromethyl moieties that are crucial for modulating the pharmacokinetic properties of drug candidates. Reaction conditions in traditional processes are often extreme, involving high temperatures or prolonged reaction times that can lead to thermal decomposition of sensitive intermediates and reduced overall yields. Additionally, the reliance on volatile organic solvents poses significant safety risks regarding flammability and operator exposure, while the difficulty in separating catalysts from the final product often results in metal contamination, necessitating complex and costly purification steps to meet stringent pharmaceutical purity standards.
The Novel Approach
In stark contrast to these legacy issues, the methodology disclosed in patent CN107721917B represents a paradigm shift towards greener and more efficient manufacturing. By utilizing the room-temperature acidic ionic liquid [Bmim]ZnBr3 as a dual-function medium, this process eliminates the need for additional organic solvents during the reaction phase, thereby drastically reducing the environmental footprint. The reaction proceeds smoothly at a moderate temperature of 100°C within a short timeframe of 1.5 hours, demonstrating exceptional energy efficiency compared to conventional heating methods. A key advantage of this system is the recyclability of the ionic liquid catalyst; experimental data indicates that the catalyst can be recovered and reused multiple times with only marginal decreases in yield, translating to significant long-term cost savings. Moreover, the workup procedure is remarkably simple, involving basic extraction and washing steps that allow for the easy isolation of the target polysubstituted nicotinic acid esters with high purity, streamlining the downstream processing workflow for commercial production facilities.
Mechanistic Insights into [Bmim]ZnBr3-Catalyzed Tandem Cyclization
The core of this technological advancement lies in the unique catalytic mechanism facilitated by the zinc-based ionic liquid. The [Bmim]ZnBr3 species acts as a potent Lewis acid, activating the carbonyl group of the 1,2-allenone substrate towards nucleophilic attack by the amino group of the enamine ester. This initial interaction triggers a cascade of intramolecular cyclization events that efficiently construct the pyridine ring system in a single pot. The ionic nature of the solvent stabilizes charged intermediates throughout the reaction pathway, lowering the activation energy barrier and allowing the transformation to proceed under milder thermal conditions than would be possible in non-polar solvents. This stabilization effect is particularly beneficial for maintaining the integrity of sensitive functional groups, ensuring that the structural diversity of the starting materials is preserved in the final product. The tandem nature of the reaction means that multiple bond-forming steps occur sequentially without the need to isolate unstable intermediates, which minimizes material loss and maximizes atom economy, a critical metric for sustainable chemical manufacturing.
![General reaction scheme for the green synthesis of polysubstituted nicotinic acid esters using [Bmim]ZnBr3](/insights/img/polysubstituted-nicotinic-acid-ester-synthesis-pharma-supplier-20260303052649-01.png)
Furthermore, the robustness of this catalytic system is evidenced by its impressive tolerance to a wide array of substituents on both the allenone and enamine ester components. As illustrated in the specific examples provided within the patent documentation, the reaction successfully accommodates electron-withdrawing groups such as bromo, fluoro, chloro, and trifluoromethyl at various positions on the aromatic rings without compromising yield. For instance, substrates bearing 4-bromophenyl or 3-fluorophenyl groups react efficiently to produce the corresponding nicotinic acid derivatives in yields ranging from 60% to over 80%. This broad functional group compatibility is essential for medicinal chemists who require diverse libraries of analogs for structure-activity relationship (SAR) studies. The ability to introduce halogen atoms directly onto the heterocyclic core also provides valuable handles for subsequent cross-coupling reactions, enabling the rapid elaboration of these intermediates into more complex drug-like molecules. The mechanistic efficiency combined with this versatility makes this ionic liquid-catalyzed route a superior choice for the synthesis of high-purity pharmaceutical intermediates.
How to Synthesize Polysubstituted Nicotinic Acid Esters Efficiently
Implementing this green synthesis protocol in a laboratory or pilot plant setting requires adherence to specific operational parameters to ensure optimal results and reproducibility. The process begins with the precise weighing of the substituted enamine ester and 1,2-allenone compound, typically in a 1:1 molar ratio, which are then dissolved directly into the dried acidic ionic liquid [Bmim]ZnBr3. The mixture is initially stirred at room temperature for approximately 10 minutes to ensure homogeneity before being heated to the reaction temperature of 100°C. Monitoring the reaction progress via thin-layer chromatography (TLC) is recommended to confirm the complete consumption of starting materials, which usually occurs within 1.5 hours. Upon completion, the reaction mixture is cooled to room temperature, and the product is extracted using diethyl ether, followed by washing with saturated brine to remove any residual ionic species. The organic layer is then dried over anhydrous sodium sulfate, filtered, and concentrated under reduced pressure. The crude residue is finally purified by column chromatography using a mixture of petroleum ether and ethyl acetate to afford the pure polysubstituted nicotinic acid ester derivative. For detailed standardized synthesis steps, please refer to the guide below.
- Dissolve substituted enamine ester and 1,2-allenone compound in acidic ionic liquid [Bmim]ZnBr3 at room temperature.
- Heat the mixture to 100°C and stir for 1.5 hours to complete the tandem cyclization reaction.
- Extract the product with ether, wash with brine, dry, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this ionic liquid-catalyzed synthesis offers tangible strategic benefits that extend beyond mere technical feasibility. The primary value proposition lies in the significant reduction of operational costs associated with solvent procurement, waste disposal, and catalyst replacement. By eliminating the need for large volumes of volatile organic solvents during the reaction phase, facilities can lower their expenditure on raw materials and reduce the regulatory burden associated with solvent emissions and storage. The recyclability of the [Bmim]ZnBr3 catalyst further enhances cost efficiency, as the same batch of ionic liquid can be utilized for multiple reaction cycles, effectively amortizing the initial investment over a larger production volume. This leads to a more predictable and stable cost structure for the manufacturing of these critical pharmaceutical intermediates, allowing for better budget forecasting and margin protection in competitive markets.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the reduction in solvent usage directly translate to lower variable costs per kilogram of product. Since the ionic liquid serves as both solvent and catalyst, the process simplifies the bill of materials and reduces the complexity of the supply chain. The simplified workup procedure, which avoids complex distillation or specialized filtration equipment for catalyst removal, also lowers utility consumption and labor hours required for purification. Consequently, this method enables cost reduction in pharmaceutical intermediate manufacturing by streamlining the entire production workflow and minimizing waste generation fees.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, namely substituted enamine esters and 1,2-allenone compounds, are either commercially available or easily prepared from common precursors, ensuring a robust and resilient supply chain. The mild reaction conditions reduce the risk of equipment failure or safety incidents that could disrupt production schedules, thereby enhancing supply continuity. Furthermore, the high functional group tolerance means that a single standardized protocol can be used to produce a wide variety of derivatives, reducing the need for multiple specialized production lines and increasing overall facility flexibility. This reliability is crucial for reducing lead time for high-purity pharmaceutical intermediates, ensuring that downstream drug development projects remain on schedule.
- Scalability and Environmental Compliance: The green nature of this process aligns perfectly with increasingly stringent global environmental regulations regarding chemical manufacturing. The absence of volatile organic solvents and the ability to recycle the catalyst significantly reduce the environmental footprint, facilitating easier permitting and compliance with local environmental protection laws. The mild thermal requirements (100°C) make the reaction inherently safer and easier to scale up from gram to ton quantities without the need for specialized high-pressure or high-temperature reactors. This scalability supports the commercial scale-up of complex heterocyclic compounds, allowing manufacturers to respond quickly to market demand surges while maintaining a commitment to sustainable and responsible chemistry practices.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of this synthesis route for their specific applications, we have compiled answers to common inquiries based on the patent data. These questions address key concerns regarding catalyst performance, substrate compatibility, and product isolation, providing a clear understanding of the technology's capabilities. The responses are derived directly from the experimental results and technical specifications outlined in the intellectual property documentation, ensuring accuracy and relevance for process development decisions. Understanding these details is essential for integrating this green method into existing manufacturing workflows effectively.
Q: What are the advantages of using [Bmim]ZnBr3 ionic liquid in this synthesis?
A: The acidic ionic liquid [Bmim]ZnBr3 serves a dual role as both catalyst and solvent, eliminating the need for volatile organic solvents. It allows for mild reaction conditions at 100°C and can be recycled multiple times without significant loss of catalytic activity, reducing waste and cost.
Q: What is the substrate scope for this nicotinic acid ester synthesis?
A: The method exhibits excellent functional group tolerance. It accommodates various substituents on the aryl rings, including electron-withdrawing groups like bromo, fluoro, chloro, and trifluoromethyl, as well as electron-donating groups like methyl and benzyl, yielding products with good to high efficiency.
Q: How is the product isolated and purified after the reaction?
A: The isolation process is straightforward and environmentally friendly. After the reaction, the mixture is cooled and extracted with ether. The organic phase is washed with saturated brine, dried over anhydrous sodium sulfate, and the target product is purified using standard column chromatography with petroleum ether and ethyl acetate.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted Nicotinic Acid Esters Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of green chemistry technologies like the ionic liquid-catalyzed synthesis of polysubstituted nicotinic acid esters. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications required by global regulatory bodies. We are committed to delivering high-quality intermediates that empower our clients to accelerate their drug discovery and development pipelines with confidence and efficiency.
We invite you to collaborate with us to leverage this advanced synthesis technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can optimize your supply chain and drive value for your organization.
