Advanced Green Synthesis of 1-(4-Chlorophenyl)-2-Cyclopropyl-1-Propanone for Scalable Agrochemical Production
Advanced Green Synthesis of 1-(4-Chlorophenyl)-2-Cyclopropyl-1-Propanone for Scalable Agrochemical Production
The global demand for high-efficiency triazole fungicides continues to surge, driving the need for robust and environmentally compliant synthetic routes for their key intermediates. Patent CN107721834B, published in early 2021, introduces a groundbreaking preparation method for 1-(4-chlorophenyl)-2-cyclopropyl-1-propanone, a critical building block in the manufacture of Cyproconazole. This advanced protocol addresses long-standing industry challenges by replacing hazardous reagents with safer alternatives and eliminating toxic wastewater streams. The technology represents a significant leap forward in green chemistry for the agrochemical sector, offering a pathway to high-purity intermediates that align with modern sustainability goals.

Cyproconazole is a potent ergosterol demethylation inhibitor widely used to protect cereal crops, coffee, and fruits from devastating fungal pathogens. The efficiency of the final active pharmaceutical ingredient (API) relies heavily on the quality and cost-effectiveness of its precursors. The patented method described in CN107721834B utilizes a novel coupling reaction between a specific sulfonylhydrazone derivative and p-chlorobenzaldehyde. This approach not only simplifies the synthetic sequence but also drastically improves the overall atom economy. For R&D directors and process chemists, this patent offers a viable alternative to legacy methods that have plagued the industry with safety hazards and complex waste management issues for decades.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1-(4-chlorophenyl)-2-cyclopropyl-1-propanone has relied heavily on the Horner-Wadsworth-Emmons (HWE) reaction, as disclosed in various prior art documents such as US4664696 and WO2014079344A. While effective in forming the carbon-carbon double bond, these traditional routes suffer from severe drawbacks that hinder modern industrial application. The process typically requires the conversion of raw materials into alkyl phosphite esters, which then react with cyclopropylmethyl ketone under the influence of extremely strong bases like sodium amide or sodium hydride. These reagents pose significant safety risks during transportation, storage, and handling due to their pyrophoric nature and reactivity with moisture.
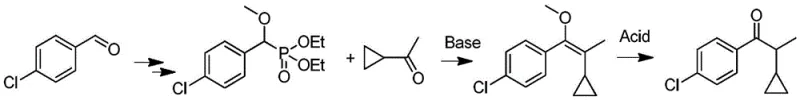
Furthermore, the post-reaction treatment in conventional HWE methods generates substantial amounts of phosphorus-containing wastewater, which is notoriously difficult and expensive to treat to meet environmental discharge standards. The subsequent acidolysis step required to obtain the final ketone produces large volumes of acidic wastewater, compounding the environmental burden. From a supply chain perspective, the reliance on specialized phosphorus reagents and hazardous bases increases raw material costs and introduces volatility into the procurement process. The long synthetic route also results in lower overall yields and higher energy consumption, making these legacy processes economically uncompetitive in a market increasingly driven by green chemistry mandates.
The Novel Approach
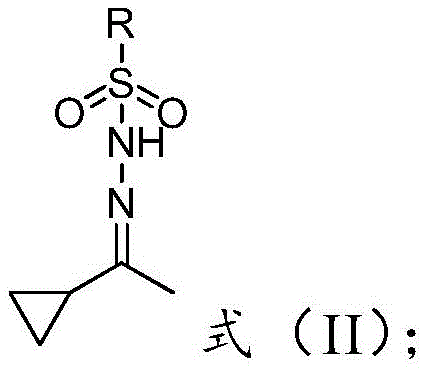
In stark contrast to the cumbersome traditional methods, the novel approach disclosed in patent CN107721834B streamlines the synthesis into a more direct and benign process. The core innovation lies in the use of an acetyl cyclopropane sulfonylhydrazone derivative (Formula II) as the key nucleophilic partner. This intermediate reacts directly with p-chlorobenzaldehyde under mild basic conditions to yield the target ketone. The elimination of phosphorus reagents entirely removes the associated wastewater treatment costs and environmental liabilities. Moreover, the reaction can be catalyzed by inexpensive halogenated metal salts such as cuprous iodide or simple phase transfer catalysts, avoiding the need for precious metal catalysts often found in cross-coupling reactions.
The operational simplicity of this new route is a major advantage for manufacturing teams. The reaction proceeds efficiently in common organic solvents like dioxane, methanol, or tert-butanol at temperatures ranging from 50°C to 150°C. The use of milder bases such as potassium carbonate, sodium carbonate, or alkoxides significantly reduces the safety risks associated with exothermic runaway reactions. Experimental examples in the patent demonstrate that this method consistently delivers high yields, often exceeding 80%, with product purities above 95% as determined by HPLC. This robustness makes the process highly attractive for commercial scale-up, offering a reliable supply of high-quality intermediates for the production of Cyproconazole and related agrochemicals.
Mechanistic Insights into Sulfonylhydrazone-Mediated Coupling
The chemical transformation at the heart of this patent involves the base-mediated decomposition of the sulfonylhydrazone to generate a reactive diazo species in situ. Upon treatment with a base, the sulfonylhydrazone derivative undergoes deprotonation and subsequent elimination of the sulfinic acid moiety, forming a diazo compound. This highly reactive intermediate is then poised to react with the electrophilic carbonyl group of p-chlorobenzaldehyde. The presence of a promoter, such as a copper(I) salt or a halide source, plays a crucial role in facilitating this coupling. It is hypothesized that the metal catalyst assists in the decomposition of the diazo species or stabilizes the transition state, leading to the formation of the desired ketone functionality while preserving the sensitive cyclopropyl ring.
From an impurity control perspective, this mechanism offers distinct advantages over carbene-based or organometallic alternatives. The specificity of the reaction minimizes the formation of side products such as polymerization byproducts or over-alkylated species that are common in less controlled environments. The patent data indicates that by optimizing the molar ratio of the promoter to the hydrazone (preferably between 0.05:1 and 1:1), the generation of byproducts is effectively suppressed. This precise control over the reaction pathway ensures that the crude product requires minimal purification, thereby reducing solvent usage and processing time. For quality assurance teams, understanding this mechanism is vital for setting appropriate in-process control (IPC) parameters to monitor the consumption of the aldehyde and the formation of the diazo intermediate.
How to Synthesize 1-(4-Chlorophenyl)-2-Cyclopropyl-1-Propanone Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production. The process begins with the preparation of the sulfonylhydrazone precursor, which is achieved by condensing cyclopropyl methyl ketone with a substituted sulfonyl hydrazide in an alcoholic solvent at room temperature. This step is straightforward and high-yielding, providing a stable intermediate that can be stored or used directly in the next stage. The subsequent coupling reaction with p-chlorobenzaldehyde is the critical step where process parameters such as temperature, base selection, and catalyst loading must be strictly controlled to maximize efficiency. Detailed standard operating procedures for this synthesis are essential for ensuring batch-to-batch consistency.
- Prepare the acetyl cyclopropane sulfonylhydrazone derivative by reacting cyclopropyl methyl ketone with a substituted sulfonyl hydrazide in an alcoholic solvent at room temperature.
- React the resulting sulfonylhydrazone with p-chlorobenzaldehyde in the presence of a base (e.g., potassium carbonate) and a promoter (e.g., cuprous iodide) in a solvent like dioxane.
- Heat the mixture to 50-150°C under nitrogen protection, filter the reaction solution, and purify the crude product to obtain the target ketone with high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the technology described in CN107721834B presents a compelling value proposition centered on cost stability and operational reliability. The shift away from hazardous and regulated reagents towards commodity chemicals fundamentally alters the cost structure of manufacturing this key agrochemical intermediate. By eliminating the need for specialized phosphorus compounds and dangerous strong bases, manufacturers can secure raw materials from a broader supplier base, reducing the risk of supply disruptions. Furthermore, the simplified workflow reduces the number of unit operations required, leading to faster throughput times and lower labor costs per kilogram of product.
- Cost Reduction in Manufacturing: The most significant economic driver of this new method is the drastic reduction in waste treatment expenses. Traditional routes generate phosphorus-containing and acidic wastewater that requires complex neutralization and precipitation processes before discharge. By completely avoiding these pollutants, the new process lowers the operational expenditure (OPEX) associated with environmental compliance. Additionally, the use of inexpensive catalysts like cuprous iodide or simple halide salts instead of precious metals further drives down the direct material costs. The higher yields reported in the patent examples also mean less raw material is wasted, improving the overall mass balance and profitability of the production line.
- Enhanced Supply Chain Reliability: The raw materials for this synthesis, including p-chlorobenzaldehyde and cyclopropyl methyl ketone, are widely produced commodity chemicals with stable global supply chains. This contrasts sharply with the specialized reagents required for the Horner-Wadsworth-Emmons reaction, which may be subject to availability fluctuations. The robustness of the reaction conditions, which tolerate a range of solvents and bases, provides manufacturing flexibility. If a specific solvent becomes scarce or expensive, the process can be adapted to use alternatives like methanol or dioxane without compromising the outcome, ensuring continuous production even during market volatility.
- Scalability and Environmental Compliance: As regulatory pressures on chemical manufacturing intensify globally, the ability to demonstrate a "green" synthesis route is a strategic asset. This method generates virtually no wastewater during the key coupling step, aligning perfectly with zero-discharge initiatives. The absence of pyrophoric reagents simplifies the safety infrastructure required for the plant, potentially lowering insurance premiums and capital investment in specialized containment systems. The process is inherently scalable, having been demonstrated to work efficiently with standard heating and stirring equipment, making the transition from pilot plant to multi-ton commercial production seamless and low-risk.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These insights are derived directly from the experimental data and claims presented in patent CN107721834B, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this technology into their existing manufacturing portfolios.
Q: What are the primary advantages of the sulfonylhydrazone route over the Horner-Wadsworth-Emmons method?
A: The sulfonylhydrazone route eliminates the use of hazardous strong bases like sodium hydride and avoids the generation of phosphorus-containing wastewater, significantly reducing environmental compliance costs and safety risks associated with traditional methods.
Q: What yields and purity levels can be expected from this patented process?
A: Experimental data from the patent indicates that the process consistently achieves yields ranging from 70% to 88%, with HPLC purity levels exceeding 95%, ensuring high-quality material for downstream fungicide synthesis.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the process utilizes cheap and readily available raw materials, operates under relatively mild conditions (50-150°C), and generates minimal wastewater, making it highly suitable for commercial scale-up and continuous manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-(4-Chlorophenyl)-2-Cyclopropyl-1-Propanone Supplier
The technological advancements detailed in patent CN107721834B underscore the potential for more sustainable and efficient agrochemical manufacturing. At NINGBO INNO PHARMCHEM, we specialize in translating such innovative patent technologies into commercial reality. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, ensuring that every batch of 1-(4-chlorophenyl)-2-cyclopropyl-1-propanone meets the exacting standards required for high-performance fungicide formulation.
We invite global partners to collaborate with us to leverage this green synthesis route for their supply chains. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you optimize your production of Cyproconazole intermediates with superior quality and sustainability.
