Advanced Base-Mediated Synthesis Of Alkyl Trimethyl Tin Compounds For Commercial Scale-Up
Advanced Base-Mediated Synthesis Of Alkyl Trimethyl Tin Compounds For Commercial Scale-Up
The landscape of organotin chemistry is undergoing a significant transformation driven by the need for safer, more cost-effective, and scalable synthetic routes. A pivotal advancement in this field is detailed in Chinese patent CN112979693A, which discloses a novel series of alkyl trimethyl tin compounds and their preparation methods. This technology represents a major departure from conventional reliance on hazardous Grignard reagents or expensive transition metal catalysts. By utilizing a mild base-mediated transmetallation strategy involving alkyl iodides and hexamethylditin, the process achieves high yields under remarkably gentle conditions, specifically at 30 °C over 24 hours. For R&D directors and procurement managers in the fine chemical sector, this innovation offers a robust pathway to access critical building blocks for Stille coupling reactions, liquid crystal synthesis, and pharmaceutical intermediate manufacturing with enhanced safety profiles and reduced operational costs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of alkyl trimethyl tin compounds has been fraught with significant technical and economic challenges that hinder efficient commercial scale-up. Traditional methodologies predominantly rely on the preparation of corresponding alkyl Grignard reagents or alkyl lithium reagents from alkyl halides, followed by reaction with trimethyl tin chloride. This approach is inherently problematic due to the extreme sensitivity of Grignard and organolithium reagents to moisture and oxygen, necessitating rigorous anhydrous conditions and specialized equipment that drive up capital expenditure. Furthermore, alternative routes involving the reaction of alkyl iodides or bromides with hexamethylditin often require the presence of expensive palladium catalysts, which not only inflate raw material costs but also introduce complexities regarding heavy metal removal and residual catalyst control in the final product. These conventional methods are often unfriendly to the environment, involve complex operations, and require harsh reaction conditions that limit substrate scope and pose safety risks in large-scale manufacturing environments.
The Novel Approach
In stark contrast to these legacy techniques, the method disclosed in patent CN112979693A introduces a streamlined and economically superior protocol that eliminates the need for transition metal catalysts and pyrophoric reagents. The core innovation lies in the direct reaction of widely available and inexpensive alkyl iodides with hexamethylditin in the presence of a strong base, such as lithium tert-butoxide, within a polar aprotic solvent like dimethyl sulfoxide (DMSO). This system operates effectively at room temperature (30 °C), drastically reducing energy consumption compared to refluxing conditions often required in older methods. The simplicity of mixing alkyl iodide, base, and hexamethylditin uniformly allows for a straightforward reaction setup that is amenable to automation and continuous processing. By avoiding the preparation of intermediate organometallic species like Grignard reagents, the process shortens the synthetic timeline and minimizes waste generation, presenting a compelling value proposition for cost reduction in pharmaceutical intermediates manufacturing and specialty chemical production.
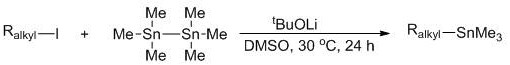
Mechanistic Insights into Base-Mediated Transmetallation
The mechanistic elegance of this synthesis lies in the efficient transmetallation facilitated by the alkali metal alkoxide base. In this system, the base, preferably lithium tert-butoxide (tBuOLi), acts as a crucial activator that promotes the cleavage of the tin-tin bond in hexamethylditin and facilitates the transfer of the trimethylstannyl group to the alkyl moiety derived from the alkyl iodide. The use of DMSO as a solvent is particularly strategic, as its high polarity stabilizes the ionic intermediates and enhances the solubility of the inorganic base, thereby accelerating the reaction kinetics without the need for external heating. This mechanism ensures that the reaction proceeds smoothly even with sterically hindered substrates or those containing sensitive functional groups. The absence of transition metals means there is no risk of catalyst poisoning by heteroatoms, a common issue in palladium-catalyzed stannylation, thus broadening the scope of compatible substrates significantly.
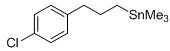
Furthermore, the exceptional functional group tolerance observed in this method is a critical advantage for synthesizing complex pharmaceutical intermediates. The reaction conditions are mild enough to preserve labile groups such as esters, nitriles, halides (chloro, bromo, fluoro), and sulfones, which might otherwise decompose under the harsh conditions of Grignard formation or high-temperature catalysis. For instance, substrates like 1-chloro-6-iodohexane or 7-iodoheptanitrile can be converted to their corresponding trimethyl tin derivatives with high fidelity, maintaining the integrity of the chloro and cyano functionalities for downstream cross-coupling applications. This chemoselectivity allows medicinal chemists to design convergent synthetic routes where the organotin moiety is introduced late in the sequence, minimizing protection-deprotection steps and improving overall process efficiency. The structural versatility is further evidenced by the successful synthesis of aryl-containing alkyl tin compounds, such as 3-(4-chlorophenyl)propyltrimethyltin, which serves as a key precursor for biaryl liquid crystal materials.
How to Synthesize Alkyl Trimethyl Tin Efficiently
The practical implementation of this synthesis route is designed for ease of operation, making it accessible for both laboratory-scale optimization and industrial production. The standard protocol involves charging a reaction vessel with the specific alkyl iodide substrate, the base (lithium tert-butoxide being preferred for optimal yield), and hexamethylditin in DMSO. The mixture is stirred at 30 °C for 24 hours, after which the solvent is removed via concentration, typically using rotary evaporation under reduced pressure. The crude residue is then subjected to column chromatography, utilizing petroleum ether or a mixture of petroleum ether and ethyl acetate as the mobile phase, to isolate the pure alkyl trimethyl tin product. Yields reported in the patent range broadly from 38% to 94%, depending on the substrate structure, demonstrating the robustness of the method across diverse chemical spaces. Detailed standardized synthesis steps for specific high-value derivatives are outlined in the guide below.
- Mix alkyl iodide, lithium tert-butoxide, and hexamethylditin in dimethyl sulfoxide (DMSO) solvent.
- React the mixture uniformly at 30°C for 24 hours under mild conditions without transition metal catalysts.
- Concentrate the reaction mixture and purify the crude product via column chromatography to obtain high-purity alkyl trimethyl tin.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this base-mediated synthesis technology translates into tangible strategic benefits regarding cost stability and supply security. The primary driver of cost efficiency is the substitution of expensive and hazardous reagents with commodity chemicals. Alkyl iodides are generally more stable and easier to source in bulk quantities compared to the precursors required for Grignard reagent preparation, and the elimination of palladium catalysts removes a significant variable cost associated with noble metal price volatility. Moreover, the mild reaction conditions reduce the energy load on manufacturing facilities, as there is no need for cryogenic cooling or high-temperature heating, leading to lower utility costs per kilogram of product. The simplified workup procedure, which avoids complex quenching of reactive organometallics, also reduces the consumption of auxiliary materials and waste treatment expenses, contributing to a leaner and more sustainable manufacturing process.
- Cost Reduction in Manufacturing: The economic impact of this method is profound due to the drastic simplification of the reagent profile. By eliminating the need for equivalent amounts of alkyl metal reagents and expensive transition metal catalysts, the direct material cost is significantly lowered. The process utilizes hexamethylditin and alkali bases which are cost-effective at scale, and the absence of heavy metals simplifies the purification workflow, reducing the need for specialized scavenging resins or extensive washing protocols. This streamlined approach ensures that the production of high-purity alkyl trimethyl tin compounds can be achieved with substantially lower operational expenditures, enhancing the margin potential for downstream API and intermediate manufacturers.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available and stable raw materials. Unlike pyrophoric organolithium or Grignard reagents which require specialized logistics and storage infrastructure, alkyl iodides and solid bases can be transported and stored with standard safety measures, reducing the risk of supply disruptions due to regulatory or logistical constraints. The robustness of the reaction against functional group variations means that a single manufacturing line can be adapted to produce a wide array of tin derivatives without extensive retooling, allowing for flexible response to market demand fluctuations. This adaptability ensures a consistent supply of critical intermediates for clients in the pharmaceutical and agrochemical sectors, mitigating the risk of production delays.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this method aligns well with modern green chemistry principles. The reaction generates less hazardous waste compared to traditional methods that produce stoichiometric amounts of magnesium or lithium salts alongside heavy metal residues. The mild conditions minimize the formation of side products and decomposition byproducts, leading to higher atom economy and easier waste stream management. Scaling this process from 100 kgs to multi-tonne production is straightforward because the exotherm is manageable and the reagents are not shock-sensitive, ensuring safe operation in large reactors. This compliance with stringent environmental standards facilitates smoother regulatory approvals and supports the long-term sustainability goals of global chemical enterprises.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this alkyl trimethyl tin synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on process capabilities and product quality. Understanding these aspects is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the advantages of this synthesis method over traditional Grignard routes?
A: Unlike traditional methods requiring complex Grignard reagent preparation or expensive palladium catalysts, this method uses readily available alkyl iodides and operates under mild conditions (30°C) without transition metals, significantly reducing operational complexity and cost.
Q: Does this method tolerate sensitive functional groups?
A: Yes, the process demonstrates excellent functional group tolerance, successfully accommodating substituents such as chloro, bromo, fluoro, cyano, ester, and sulfone groups without degradation.
Q: Is this process suitable for large-scale industrial production?
A: Absolutely. The reaction avoids pyrophoric reagents and harsh conditions, utilizing stable raw materials and simple workup procedures like distillation and chromatography, making it highly scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alkyl Trimethyl Tin Supplier
As the demand for high-quality organotin intermediates grows in the pharmaceutical and electronic materials sectors, partnering with an experienced CDMO is essential for successful commercialization. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. Our facility is equipped with rigorous QC labs capable of meeting stringent purity specifications required for GMP-grade intermediates, utilizing advanced analytical techniques to verify the absence of heavy metal contaminants and ensure consistent batch-to-batch quality. We understand the critical nature of supply continuity for your drug development pipelines and are committed to delivering reliable alkyl trimethyl tin supplier services that meet your exacting standards.
We invite you to collaborate with us to leverage this innovative synthesis technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your target molecules, demonstrating how this base-mediated route can optimize your budget without compromising quality. Please contact us to request specific COA data for our catalog compounds or to discuss route feasibility assessments for custom synthesis projects. Let us help you accelerate your development timeline with our proven expertise in organometallic chemistry and commitment to excellence.
