Scalable One-Pot Synthesis of 3,3-Disubstituted Oxindoles Using Palladium-Sodium Hydride Catalysis
Introduction to Advanced Oxindole Synthesis Technology
The pharmaceutical industry continuously seeks robust methodologies for constructing complex heterocyclic scaffolds, particularly 3,3-disubstituted oxindoles, which serve as privileged structures in numerous bioactive molecules. Patent CN109020865B introduces a groundbreaking approach utilizing a metal hydride and palladium compound system to achieve the reduction of alkenyl active methylene compounds. This technology addresses critical limitations in current synthetic routes by enabling a seamless one-pot transformation that combines reduction and functionalization. The core innovation lies in the dual functionality of sodium hydride, acting not merely as a base but as a potent reducing agent in the presence of a palladium catalyst, thereby generating a reactive nucleophilic intermediate in situ. This strategic design eliminates the need for isolating sensitive intermediates, streamlining the workflow for process chemists.
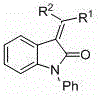
The significance of this patent extends beyond academic curiosity, offering tangible benefits for industrial manufacturing of high-purity pharmaceutical intermediates. By leveraging readily available reagents such as sodium hydride and palladium chloride, the method circumvents the reliance on hazardous high-pressure hydrogenation or costly proprietary reducing agents. The versatility of the substrate scope, accommodating various R groups including alkyl, phenyl, and heteroaryl moieties, ensures broad applicability across different drug discovery programs. Furthermore, the mild reaction conditions, typically proceeding at room temperature, underscore the potential for significant energy savings and enhanced process safety profiles in large-scale production environments.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the reduction of electron-deficient double bonds in alkenyl active methylene compounds has relied heavily on catalytic hydrogenation using hydrogen gas and palladium on carbon. While effective, this traditional approach necessitates specialized high-pressure equipment and rigorous safety protocols to mitigate the risks associated with handling explosive hydrogen gas, creating substantial barriers for smaller facilities and increasing capital expenditure. Alternative chemical reduction methods employing reagents such as the Stryker reagent, silanes, or Hantzsch esters present their own set of challenges, primarily revolving around cost and waste generation. These reagents are often prohibitively expensive for kilogram-scale synthesis and produce stoichiometric amounts of difficult-to-remove byproducts, complicating downstream purification and increasing the environmental footprint of the manufacturing process.
The Novel Approach
In stark contrast, the methodology disclosed in CN109020865B utilizes a synergistic combination of sodium hydride and a palladium catalyst to effect reduction under remarkably mild and safe conditions. This novel system operates effectively at atmospheric pressure and room temperature, completely eliminating the safety hazards associated with high-pressure hydrogenation while maintaining high reaction efficiency. The true brilliance of this approach is its ability to generate a stable sodium salt intermediate which retains nucleophilicity, allowing for immediate subsequent reaction with electrophiles in a telescoped one-pot sequence. This integration of reduction and alkylation into a single operational step drastically reduces solvent consumption, labor time, and overall processing costs, representing a paradigm shift in the synthesis of 3,3-disubstituted oxindoles.
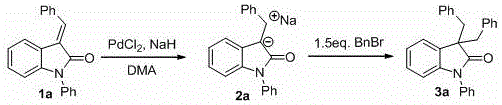
Mechanistic Insights into Pd-NaH Catalyzed Reduction and Alkylation
The mechanistic pathway of this transformation involves a sophisticated interplay between the palladium catalyst and the metal hydride reducing agent. Initially, the palladium species activates the hydride source, facilitating the transfer of hydride equivalents to the electron-deficient double bond of the alkenyl substrate. This reduction step converts the neutral alkene into a highly reactive enolate species stabilized as a sodium salt. Unlike traditional reductions that yield neutral products requiring separate activation for further functionalization, this in situ generated sodium enolate possesses inherent nucleophilicity. This unique feature allows the intermediate to directly attack electrophilic centers introduced in the second stage of the reaction, such as alkyl halides or Michael acceptors, without the need for additional bases or activation steps.
From an impurity control perspective, this mechanism offers distinct advantages by minimizing the number of unit operations and exposure of reactive intermediates to the environment. The one-pot nature of the reaction ensures that the reactive enolate is consumed immediately upon formation, reducing the likelihood of side reactions such as polymerization or hydrolysis that often plague isolated enolate chemistry. Furthermore, the use of sodium hydride results in the formation of benign sodium salts as byproducts, which are easily removed during the aqueous workup phase. This clean reaction profile simplifies the purification process, typically requiring only standard extraction and column chromatography to achieve high-purity final products, thereby ensuring a consistent and reliable quality profile suitable for pharmaceutical applications.
How to Synthesize 3,3-Disubstituted Oxindoles Efficiently
The practical execution of this synthesis involves a straightforward protocol that is highly amenable to scale-up. The process begins with the suspension of the palladium catalyst and sodium hydride in a polar aprotic solvent such as DMA under an inert nitrogen atmosphere to prevent moisture interference. Once the catalytic system is activated, the alkenyl substrate is introduced, and the mixture is stirred at room temperature to allow complete reduction to the sodium salt intermediate. Following confirmation of reduction completion, the electrophilic reagent is added directly to the reaction vessel to effect the substitution, culminating in the formation of the target 3,3-disubstituted oxindole. Detailed standardized synthesis steps are provided in the guide below.
- Suspend a palladium compound (e.g., PdCl2) and a metal hydride (e.g., NaH) in a polar aprotic solvent like DMA under nitrogen atmosphere.
- Add the alkenyl active methylene substrate to the mixture and stir at room temperature to effect reduction to the sodium salt intermediate.
- Introduce an electrophilic reagent (e.g., benzyl bromide, methyl iodide) directly to the reaction mixture to form the 3,3-disubstituted product, followed by aqueous quench and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this Pd-NaH catalytic system presents a compelling value proposition centered on cost efficiency and operational reliability. The replacement of expensive and hazardous reagents with commodity chemicals like sodium hydride and palladium chloride significantly lowers the raw material cost base. Moreover, the elimination of high-pressure hydrogenation equipment reduces capital investment requirements and maintenance overheads, while the one-pot procedure minimizes solvent usage and waste disposal costs. These factors collectively contribute to a more lean and cost-effective manufacturing process that enhances the overall competitiveness of the supply chain for critical pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The economic benefits of this technology are driven by the substitution of high-cost reducing agents with inexpensive sodium hydride, which is widely available and cost-effective compared to specialized reagents like the Stryker reagent or silanes. Additionally, the telescoped one-pot process eliminates the need for intermediate isolation and purification steps, resulting in substantial savings in labor, solvent, and time. The avoidance of high-pressure hydrogenation equipment further reduces capital expenditure and operational safety costs, leading to a significantly lower cost of goods sold for the final active pharmaceutical ingredient intermediates.
- Enhanced Supply Chain Reliability: The reliance on readily available, commodity-grade reagents such as sodium hydride and palladium salts ensures a robust and resilient supply chain that is less susceptible to disruptions compared to processes dependent on specialized or imported catalysts. The mild reaction conditions and operational simplicity facilitate easier technology transfer between manufacturing sites, ensuring consistent production capacity and continuity of supply. This reliability is crucial for meeting the stringent delivery schedules of global pharmaceutical clients and maintaining inventory levels without the risk of production delays caused by complex or hazardous processing requirements.
- Scalability and Environmental Compliance: The process demonstrates excellent scalability due to its mild thermal profile and lack of exothermic hazards associated with high-pressure hydrogenation, making it ideal for transition from laboratory to commercial scale production. From an environmental standpoint, the generation of harmless sodium salt byproducts and the high atom economy of the one-pot reaction align perfectly with green chemistry principles, simplifying wastewater treatment and regulatory compliance. This eco-friendly profile not only reduces the environmental footprint but also mitigates regulatory risks, ensuring long-term sustainability for the manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on the operational parameters and scope of the technology. Understanding these details is essential for evaluating the feasibility of integrating this method into existing manufacturing workflows.
Q: What are the safety advantages of using NaH/Pd over traditional hydrogenation?
A: Traditional hydrogenation requires high-pressure hydrogen gas, posing explosion risks. The NaH/Pd system operates at atmospheric pressure and room temperature, significantly enhancing operational safety while avoiding expensive specialized equipment.
Q: How does this method improve atom economy compared to silane reductions?
A: Unlike silane reductions which generate stoichiometric silicon waste, the NaH/Pd system produces harmless sodium salts as byproducts. Furthermore, the intermediate sodium salt is directly utilized in the subsequent alkylation step, maximizing atom utilization.
Q: Can this one-pot protocol accommodate diverse electrophiles?
A: Yes, the patent demonstrates high versatility. The reactive sodium enolate intermediate successfully reacts with various electrophiles including alkyl halides (benzyl bromide, methyl iodide), Michael acceptors (methyl acrylate), and oxidants (hydrogen peroxide).
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,3-Disubstituted Oxindoles Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the Pd-NaH catalytic system for the efficient production of 3,3-disubstituted oxindoles and other complex pharmaceutical intermediates. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive materials that meet the most stringent purity specifications. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical capabilities to guarantee the quality and consistency of every batch, adhering to international regulatory standards for pharmaceutical manufacturing.
We invite potential partners to engage with our technical procurement team to discuss how this innovative technology can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits this route offers for your specific supply chain. We encourage you to contact us today to obtain specific COA data and comprehensive route feasibility assessments, allowing you to make informed decisions that drive value and efficiency in your drug development pipeline.
