Advanced Four-Step Synthesis Protocol For High-Purity Proline Compound Intermediates And Commercial Scalability
The pharmaceutical and agrochemical industries continuously demand more efficient pathways for constructing complex chiral building blocks, particularly proline derivatives which serve as critical scaffolds in drug discovery. Patent CN103619815A introduces a robust manufacturing method for producing proline compounds represented by formula (1) or their salts with exceptional yield and purity. This technology addresses the longstanding challenges associated with multi-step syntheses by streamlining the process into four distinct and highly controllable stages labeled A through D. By leveraging specific reducing agents and precise temperature controls, this method ensures the preservation of optical purity while minimizing the formation of difficult-to-remove impurities. For R&D directors and procurement specialists, understanding this protocol offers a strategic advantage in securing reliable supply chains for high-value intermediates used in the production of active pharmaceutical ingredients and agricultural chemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of proline compounds similar to formula (1a) has relied heavily on routes starting from succinic anhydride derivatives, as documented in various prior art references. These traditional methodologies are frequently criticized for their excessive operational complexity, often requiring a large number of reaction steps to achieve the desired molecular architecture. Furthermore, the cumulative yield across these numerous steps is not necessarily sufficient, leading to significant material loss and increased production costs. Another conventional approach involves the use of enzymes such as oxidases to convert pyrrolidine derivatives, which can introduce limitations regarding substrate specificity and reaction conditions. These inefficiencies create bottlenecks in the supply chain, making it difficult to scale up production to meet the rigorous demands of commercial pharmaceutical intermediate manufacturing without incurring prohibitive expenses.
The Novel Approach
In stark contrast, the present invention provides a streamlined four-step sequence that dramatically improves efficiency and output. The novel approach initiates with a highly selective reduction of a pyrrolidone compound, followed by cyanation, hydrolysis or alcoholysis, and finally deprotection. This logical progression allows for the direct construction of the target proline skeleton with minimal side reactions. By utilizing specific aluminum-based reducing agents in the presence of bases, the method achieves conversion rates exceeding 99% in optimal conditions, vastly outperforming older techniques. The ability to maintain optical purity throughout the sequence ensures that the final product meets the stringent quality specifications required for chiral drug synthesis. This technological leap represents a significant advancement in cost reduction in pharmaceutical intermediate manufacturing by reducing waste and maximizing throughput.
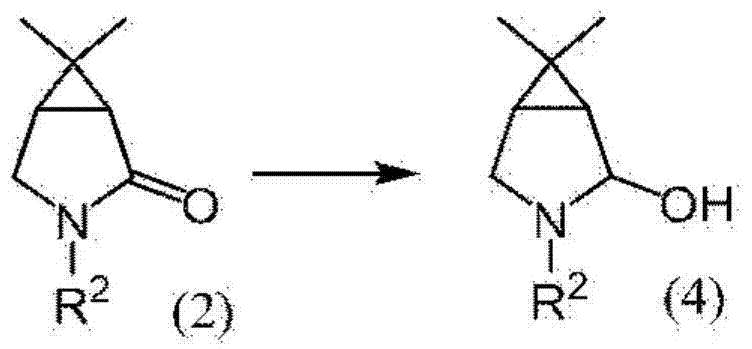
Mechanistic Insights into Selective Aluminum Hydride Reduction
The cornerstone of this synthesis lies in Step A, where the pyrrolidone compound (2) is converted to the pyrrolidinol compound (4) using a specialized reducing agent. The patent specifies the use of reducing agents represented by formula (3), such as sodium bis(2-methoxyethoxy)aluminum hydride, in the presence of bases like potassium tert-butoxide or sodium methoxide. This specific chemical environment is crucial because standard reducing agents like diisobutylaluminum hydride often lead to over-reduction, producing unwanted per-reduced by-products that complicate purification. The mechanism involves the careful delivery of hydride ions to the carbonyl group of the lactam ring without attacking other sensitive functionalities. The reaction is typically conducted at low temperatures, preferably between -20°C and 20°C, to kinetically favor the formation of the alcohol over the amine. This precise control over reaction thermodynamics is what enables the high selectivity observed in the experimental data.
Impurity control is further enhanced by the choice of solvent and workup procedures. The use of coordinating solvents such as tetrahydrofuran or diethylene glycol dimethyl ether helps stabilize the intermediate species and suppress side reactions. Following the reduction, the reaction mixture is treated with aqueous solutions containing chelating agents like sodium gluconate. This step is vital for sequestering metal ions and facilitating clean phase separation, ensuring that inorganic residues do not carry over into subsequent steps. The result is a pyrrolidinol intermediate of high purity, which sets the foundation for the high overall yield of the final proline compound. For quality assurance teams, this level of mechanistic detail underscores the reproducibility and robustness of the process when transferred to larger scales.
How to Synthesize 3-Azabicyclo[3.1.0]Hexane-2-Carboxylate Efficiently
Implementing this synthesis route requires adherence to the specific parameters outlined in the patent to ensure optimal results. The process begins with the preparation of the pyrrolidone starting material, which can be derived from commercially available lactones via reaction with amines. Once the starting material is secured, the four-step sequence A through D is executed with careful monitoring of temperature and stoichiometry. The detailed standardized synthesis steps see the guide below for a comprehensive breakdown of reagent quantities and reaction times. Adhering to these protocols allows manufacturers to replicate the high yields reported in the examples, such as the 93.9% yield achieved in the initial reduction step under optimized conditions. This structured approach minimizes trial and error, accelerating the timeline from laboratory development to commercial production.
- Step A: Selective reduction of pyrrolidone compound using aluminum hydride reducing agent in the presence of a base.
- Step B: Reaction of the resulting pyrrolidinol compound with a cyanating agent to form cyanopyrrolidine.
- Step C: Hydrolysis or alcoholysis of the cyanopyrrolidine compound to obtain the protected proline compound.
- Step D: Deprotection of the nitrogen substituent to yield the final proline compound or salt.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented methodology offers substantial benefits that directly impact the bottom line and supply chain stability. The primary advantage stems from the significant simplification of the synthetic route, which inherently reduces the consumption of raw materials and solvents. By eliminating the need for excessive purification steps associated with low-yield conventional methods, manufacturers can achieve drastic cost savings in production. The high conversion rates mean that less starting material is wasted, improving the overall atom economy of the process. Furthermore, the use of readily available reagents and common industrial solvents ensures that the supply chain remains resilient against market fluctuations. This reliability is critical for maintaining continuous production schedules and meeting delivery commitments to downstream pharmaceutical clients.
- Cost Reduction in Manufacturing: The elimination of complex multi-step sequences found in traditional succinic anhydride routes leads to a substantial decrease in operational expenditures. By consolidating the synthesis into four high-yielding steps, the process reduces labor hours, energy consumption, and waste disposal costs. The high selectivity of the reduction step minimizes the need for expensive chromatographic purifications, allowing for more economical crystallization or extraction techniques. Consequently, the overall cost of goods sold for the proline intermediate is significantly lowered, providing a competitive edge in pricing negotiations.
- Enhanced Supply Chain Reliability: The reliance on commercially available starting materials and standard reagents mitigates the risk of supply disruptions. Unlike processes that depend on specialized enzymes or rare catalysts, this method utilizes robust chemical transformations that can be sourced from multiple vendors. The scalability of the reaction conditions, which operate effectively in common solvents like toluene and tetrahydrofuran, ensures that production can be ramped up quickly to meet surges in demand. This flexibility enhances the reliability of the supply chain, ensuring consistent availability of high-purity intermediates for critical drug manufacturing programs.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction conditions that are easily transferable from laboratory to pilot and commercial scales. The controlled temperature ranges and manageable pressure requirements reduce the engineering complexity of the reactor systems. Additionally, the high yields and efficient workup procedures result in reduced waste generation, aligning with modern environmental compliance standards. The ability to recycle solvents and minimize hazardous by-products further supports sustainable manufacturing practices, making this route attractive for companies focused on green chemistry initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this proline synthesis technology. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation. Understanding these details helps stakeholders assess the feasibility and benefits of adopting this method for their specific production needs. The information covers aspects ranging from reaction mechanics to scalability considerations, providing a comprehensive overview for decision-makers.
Q: What distinguishes this proline synthesis method from conventional succinic anhydride routes?
A: Conventional methods starting from succinic anhydride derivatives often involve a large number of reaction steps and suffer from insufficient yields. This patented method utilizes a streamlined four-step sequence (A to D) that significantly reduces process complexity while achieving excellent yields, such as over 93% in the initial reduction step.
Q: How does the process control over-reduction impurities during the initial synthesis stage?
A: The process employs specific aluminum hydride reducing agents, such as sodium bis(2-methoxyethoxy)aluminum hydride, in the presence of bases like potassium tert-butoxide. This combination, along with controlled low-temperature conditions ranging from -20°C to 20°C, effectively suppresses the formation of per-reduced by-products that commonly plague standard reduction protocols.
Q: Is this synthesis route suitable for large-scale commercial production of pharmaceutical intermediates?
A: Yes, the method is designed for scalability. It uses commercially available starting materials and common organic solvents like toluene and tetrahydrofuran. The reaction conditions are manageable within standard industrial reactors, and the high conversion rates minimize waste, making it highly viable for cost reduction in pharmaceutical intermediate manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Azabicyclo[3.1.0]Hexane-2-Carboxylate Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis and manufacturing, possessing the technical expertise to translate complex patent methodologies into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We understand the critical importance of stringent purity specifications and rigorous QC labs in the pharmaceutical sector, and our facilities are equipped to deliver products that exceed these demanding standards. By partnering with us, you gain access to a wealth of process knowledge that optimizes yield and minimizes risk.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can benefit your specific projects. Request a Customized Cost-Saving Analysis to understand the potential economic impact of switching to this more efficient method. Our experts are ready to provide specific COA data and route feasibility assessments tailored to your requirements. Let us help you secure a stable and cost-effective supply of high-quality proline intermediates for your next generation of therapeutic agents.
