Scalable Copper-Catalyzed Synthesis of Benzodithiocyclopentadiene Derivatives for Industrial Applications
Scalable Copper-Catalyzed Synthesis of Benzodithiocyclopentadiene Derivatives for Industrial Applications
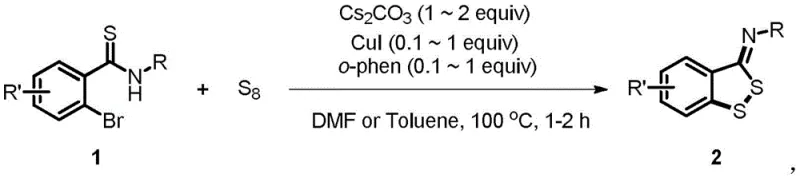
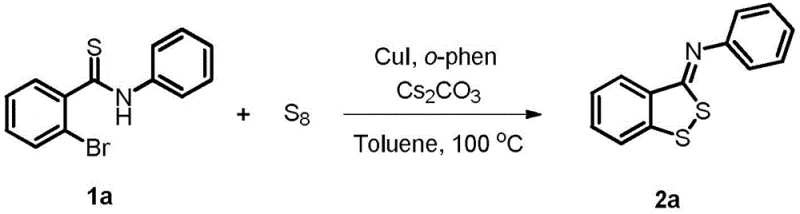
The pharmaceutical and agrochemical industries continuously demand efficient, scalable, and environmentally benign synthetic routes for complex heterocyclic scaffolds. A significant breakthrough in this domain is detailed in Chinese Patent CN109503547B, which discloses a novel preparation method for benzodithiocyclopentadiene derivatives. This technology leverages elemental sulfur (S8) as a sustainable sulfur source to react with 2-bromothioamides, catalyzed by a copper system. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediates supplier, this methodology represents a paradigm shift away from traditional, hazardous thiol-based chemistries. The process not only ensures high purity and yield but also aligns with modern green chemistry principles, making it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of benzodithio-cyclopentadiene cores has relied heavily on the use of thiophenols or their derivatives as the sulfur source. While chemically feasible, these conventional pathways suffer from severe practical drawbacks that hinder large-scale adoption. Thiophenols are notorious for their extremely pungent, unpleasant odor, which poses significant challenges for worker safety and requires specialized containment infrastructure to prevent environmental contamination. Furthermore, these reagents are often expensive and exhibit narrow substrate tolerance, frequently leading to low product yields and difficult purification processes. The harsh reaction conditions often associated with these older methods can also result in the formation of complex impurity profiles, necessitating costly downstream processing steps that erode profit margins and extend lead times for high-purity intermediates.
The Novel Approach
In stark contrast, the methodology described in patent CN109503547B utilizes elemental sulfur (S8), a cheap, odorless, and abundantly available industrial commodity, effectively bypassing the hazards associated with thiols. This innovative route employs a copper-catalyzed cyclization strategy that operates under relatively mild conditions, typically around 100°C, using robust bases like cesium carbonate. The versatility of this approach is demonstrated by its compatibility with a wide range of solvents, including polar aprotic solvents like DMF and non-polar options like toluene, allowing process chemists to optimize for solubility and workup efficiency. By replacing toxic thiols with S8, the process drastically simplifies the operational workflow and enhances the overall safety profile of the manufacturing facility.
Mechanistic Insights into Copper-Catalyzed Cyclization with Elemental Sulfur
The core of this transformative synthesis lies in the efficient activation of the carbon-sulfur bond formation using a copper catalytic cycle. The reaction mechanism likely involves the oxidative addition of the copper(I) species into the carbon-bromine bond of the 2-bromothioamide substrate, generating an organocopper intermediate. Subsequently, the elemental sulfur (S8) acts as an electrophilic sulfur source, inserting into the copper-carbon bond or reacting with the thioamide functionality to facilitate ring closure. The presence of 1,10-phenanthroline as a bidentate ligand is crucial, as it stabilizes the copper center, prevents aggregation, and enhances the catalytic turnover number. This ligand-accelerated catalysis ensures that the reaction proceeds rapidly, often reaching completion within just 1 to 2 hours, which is a significant improvement over multi-day protocols seen in legacy literature.
From an impurity control perspective, the use of S8 minimizes the formation of disulfide byproducts that are common when using thiols, which can oxidize easily. The reaction conditions are sufficiently mild to preserve sensitive functional groups such as halogens (chlorine, bromine) and ethers (methoxy), which are often present in advanced drug candidates. This chemoselectivity is vital for maintaining the integrity of the molecular scaffold during the construction of the benzodithiolane ring. The resulting products, as evidenced by the spectral data in the patent, are obtained as high-purity yellow solids after simple extraction and chromatography, indicating a clean reaction profile that reduces the burden on quality control laboratories.
How to Synthesize Benzodithiolane Derivatives Efficiently
To implement this robust synthetic route in a laboratory or pilot plant setting, operators must adhere to precise stoichiometric ratios and thermal parameters to maximize yield. The process begins with the careful weighing of the 2-bromothioamide substrate and elemental sulfur, followed by the addition of the copper catalyst system. The choice of solvent plays a pivotal role; while DMF offers excellent solubility for polar substrates, toluene may be preferred for easier solvent recovery and lower boiling point workups. The reaction is heated to reflux or maintained at 100°C, monitored closely by TLC or HPLC to ensure complete conversion before proceeding to the isolation phase. The detailed standardized synthesis steps see the guide below.
- Combine 2-bromothioamide substrate, elemental sulfur (S8), cuprous iodide catalyst, 1,10-phenanthroline ligand, and cesium carbonate base in a suitable solvent like toluene or DMF.
- Heat the reaction mixture to 100°C under stirring for 1 to 2 hours until TLC indicates complete consumption of the starting material.
- Quench the reaction with saturated brine, extract with dichloromethane, dry the organic layer, and purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this S8-based copper catalysis offers tangible strategic benefits beyond mere chemical elegance. The shift from specialized, odorous thiols to bulk commodity sulfur fundamentally alters the cost structure of the supply chain. By utilizing raw materials that are globally sourced and inexpensive, manufacturers can achieve significant cost reduction in pharmaceutical intermediates manufacturing without compromising on quality. Furthermore, the simplified reaction workup, which avoids the complex scrubbing systems required for thiol off-gassing, reduces capital expenditure on environmental control equipment and lowers the total cost of ownership for the production line.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous thiophenol reagents directly lowers the bill of materials (BOM) for every batch produced. Additionally, the high efficiency of the copper catalyst, used in relatively low loadings (0.1 to 1 equivalent), combined with the short reaction time of 1-2 hours, increases reactor throughput and reduces utility costs associated with prolonged heating and stirring. This operational efficiency translates into substantial cost savings that can be passed down the supply chain or reinvested into further R&D initiatives.
- Enhanced Supply Chain Reliability: Relying on elemental sulfur, a byproduct of petroleum refining with massive global availability, mitigates the risk of raw material shortages that often plague specialty chemical markets. Unlike custom-synthesized thiols which may have long lead times and single-source dependencies, S8 is a stable, storable commodity. This ensures continuous production capability and reduces the risk of supply disruptions, allowing supply chain planners to maintain leaner inventory levels while guaranteeing on-time delivery to downstream API manufacturers.
- Scalability and Environmental Compliance: The green nature of this process aligns perfectly with increasingly stringent environmental regulations. The absence of volatile sulfur compounds in the off-gas simplifies waste treatment and reduces the environmental footprint of the facility. The reaction's robustness in common solvents like toluene and DMF facilitates straightforward scale-up from gram to ton scales, ensuring that the process remains viable and compliant as production volumes increase to meet commercial demand.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity for technical teams evaluating this route for potential licensing or contract manufacturing opportunities. Understanding these nuances is critical for assessing the feasibility of integrating this chemistry into existing production portfolios.
Q: What is the primary advantage of using S8 over thiophenols in this synthesis?
A: Using elemental sulfur (S8) eliminates the need for malodorous and toxic thiophenols, significantly improving the environmental profile and operator safety of the manufacturing process while reducing raw material costs.
Q: Can this copper-catalyzed method tolerate diverse functional groups?
A: Yes, the protocol demonstrates excellent substrate scope, successfully accommodating electron-donating groups like methyl and methoxy, as well as electron-withdrawing halogens such as chlorine, on both the thioamide and the aniline moieties.
Q: What are the typical reaction conditions for scaling this process?
A: The reaction operates under mild thermal conditions at 100°C with a short reaction time of 1-2 hours, utilizing common solvents like DMF or toluene, which facilitates straightforward scale-up in standard industrial reactors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzodithiocyclopentadiene Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the copper-catalyzed S8 insertion strategy for producing high-value heterocycles. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to market is seamless. Our state-of-the-art facilities are equipped to handle the specific requirements of sulfur chemistry safely and efficiently, adhering to stringent purity specifications and operating rigorous QC labs to guarantee that every batch meets the highest international standards for pharmaceutical and agrochemical applications.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific molecule, demonstrating how this green chemistry approach can optimize your budget. Please contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary targets, ensuring a secure and competitive supply chain for your critical intermediates.
